
VOLUME 3
|
ISSUE 1
|
JANUARY 2013
http://www.kidney-international.org
Official JOurnal Of the internatiOnal SOciety Of nephrOlOgy
KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of
Chronic Kidney Disease

KDIGO 2012 Clinical Practice Guideline
for the Evaluation and Management of
Chronic Kidney Disease
KDIGO gratefully acknowledges the following consortium of sponsors that make our initiatives possible: Abbott, Amgen,
Bayer Schering Pharma, Belo Foundation, Bristol-Myers Squibb, Chugai Pharmaceutical, Coca-Cola Company, Dole Food
Company, Fresenius Medical Care, Genzyme, Hoffmann-LaRoche, JC Penney, Kyowa Hakko Kirin, NATCO—The
Organization for Transplant Professionals, NKF-Board of Directors, Novartis, Pharmacosmos, PUMC Pharmaceutical,
Robert and Jane Cizik Foundation, Shire, Takeda Pharmaceutical, Transwestern Commercial Services, Vifor Pharma,
and Wyeth.
Sponsorship Statement: KDIGO is supported by a consor tium of sponsors and no funding is accepted for the development
of specific guidelines.

KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management
of Chronic Kidney Disease
Tables and Figuresv
KDIGO Board Membersvii
Reference Keysviii
CKD Nomenclaturex
Conversion Factors & HbA
1c
Conversionxi
Abbreviations and Acronymsxii
Notice1
Foreword2
Work Group Membership3
Abstract4
Summary of Recommendation Statements5
Introduction: The case for updating and context15
Chapter 1: Definition, and classification of CKD19
Chapter 2: Definition, identification, and prediction of CKD progression63
Chapter 3: Management of progression and complications of CKD73
Chapter 4: Other complications of CKD: CVD, medication dosage, patient safety, infections,
hospitalizations, and caveats for investigating complications of CKD
91
Chapter 5: Referral to specialists and models of care112
Methods for Guideline Development120
Biographic and Disclosure Information128
Acknowledgments134
References136
http://www.kidney-international.org contents
& 2013 KDIGO
VOL 3 | ISSUE 1 | JANUARY (1) 2013
This journal is a member of, and subscribes to the principles of, the Committee on Publication Ethics
(COPE) www.publicationethics.org

TABLES
Table 1. KDIGO nomenclature and description for grading recommendations18
Table 2. Criteria for CKD20
Table 3. Criteria for definition of CKD20
Table 4. Classification of CKD based on presence or absence of systemic disease and location within the kidney of
pathologic-anatomic findings
27
Table 5. GFR categories in CKD27
Table 6. Albuminuria categories in CKD28
Table 7. Relationship among categories for albuminuria and proteinuria31
Table 8. CGA staging of CKD: examples of nomenclature and comments32
Table 9. Prognosis of CKD: Relationship of outcomes and strength of relationship to Cause (C), GFR (G), Albuminuria (A)
and other measures
33
Table 10. Annual percentage change in GFR across diagnosis categories35
Table 11. Sources of error in GFR estimating using creatinine39
Table 12. Equations based on serum creatinine assays in adults that are traceable to the standard reference material41
Table 13. Performance comparison of creatinine-based GFR estimating equations in North America, Europe, and Australia44
Table 14. Performance comparison of creatinine- based GFR estimating equations outside of North America, Europe, and Australia48
Table 15. Sources of error in GFR estimating using cystatin C50
Table 16. Equations based on IDMS traceable creatinine and IFCC traceable cystatin C assays52
Table 17. Performance comparison of cystatin C-based estimating equations in North American and European populations54
Table 18. Strengths and limitations of GFR measurement methods and markers55
Table 19. Factors affecting urinary ACR59
Table 20. Decline in kidney function in various populations (longitudinal studies only)64
Table 21. Decline in kidney function in CKD populations65
Table 22. Studies evaluating rapid progression (general population studies only)67
Table 23. CKD progression and risk of all-cause mortality and ESRD using baseline (first) eGFR68
Table 24. Association between absolute and percentage change in kidney function and risk of ESRD, based on adjustment
for eGFR at the first and last measurement
68
Table 25. Intensive versus normal glycemic control and albuminuria outcome77
Table 26. Recommended Daily Intake of sodium for healthy children78
Table 27. Prevalence of CKD complications by GFR category derived from CKD cohorts81
Table 28. Hemoglobin cutoffs for people living at sea level81
Table 29. Phosphate binding agents in routine clinical practice and their ranked cost87
Table 30. Summary data for bisphosphonates and CKD89
Table 31. Peripheral arterial disease and CKD100
Table 32. Cautionary notes for prescribing in people with CKD103
Table 33. Risk factors for infection in people with CKD107
Table 34. Components of community CKD management programs110
Table 35. Early versus late referral: consequences and benefits114
Table 36. Outcomes of early versus late referral114
Table 37. Topics of interest for the management of CKD guideline122
Table 38. Literature yield of primary articles for all topics123
Table 39. Classification of study quality123
Table 40. GRADE system for grading quality of evidence124
Table 41. Final grade for overall quality of evidence124
Table 42. Balance of benefits and harm124
Table 43. KDIGO nomenclature and description for grading recommendations125
Table 44. Determinants of strength of recommendation125
Table 45. The Conference on Guideline Standardization checklist for reporting clinical practice guidelines125
http://www.kidney-international.org contents
& 2013 KDIGO
Kidney International Supplements (2013) 3,v v

FIGURES
Figure 1. Conceptual model of CKD16
Figure 2. Normal values for GFR by age22
Figure 3. Relationship of eGFR with mortality23
Figure 4. Relationship of albuminuria with mortality23
Figure 5. Age-standardized rates of death from any cause, cardiovascular events, and hospitalization, according to the
eGFR among 1,120,295 ambulatory adults
28
Figure 6. Summary of continuous meta-analysis for general population cohorts with ACR29
Figure 7. Summary of categorical meta-analysis for general population cohorts with ACR30
Figure 8. Prevalence of CKD in the USA by GFR and albuminuria31
Figure 9. Prognosis of CKD by GFR and albuminuria category34
Figure 10. Determinants of the serum level of endogenous filtration markers39
Figure 11. Performance of the CKD-EPI and MDRD Study equations in estimating measured GFR in the external validation
data set
43
Figure 12. Comparison of distribution of GFR and CKD prevalence by age (NHANES 1999-2004)46
Figure 13. Meta-analysis of NRI for all-cause mortality, CVD mortality, and ESRD47
Figure 14. Association of CKD definitions with all-cause mortality and ESRD50
Figure 15. Performance of three equations for estimating GFR53
Figure 16. Suggested protocol for the further investigation of an individual demonstrating a positive reagent strip test for
albuminuria/proteinuria or quantitative albuminuria/proteinuria test
57
Figure 17. GFR and albuminuria grid to reflect the risk of progression63
Figure 18. Distribution of the probability of nonlinearity with three example trajectories demonstrating different probabilities
of nonlinearity
69
Figure 19. Summary estimates for risks of all-cause mortality and cardiovascular mortality associated with levels of serum
phosphorus, PTH, and calcium
86
Figure 20. Prevalence of deficiency of 1,25(OH)
2
D
3
, 25(OH)D
3
, and secondary hyperparathyroidism by GFR intervals88
Figure 21. Referral decision making by GFR and albuminuria113
Figure 22. The CKD chronic care model116
Additional information in the form of supplementary materials can be found online at http://www.kdigo.org/clinical_practice_guidelin es/ckd.php
vi
Kidney International Supplements (2013) 3,vi
contents http://www.kidney-international.org
& 2013 KDIGO

KDIGO Board Members
Garabed Eknoyan, MD
Norbert Lameire, MD, PhD
Founding KDIGO Co-Chairs
Kai-Uwe Eckardt, MD
Immediate Past Co-Chair
Bertram L Kasiske, MD
KDIGO Co-Chair
David C Wheeler, MD, FRCP
KDIGO Co-Chair
Omar I Abboud, MD, FRCP
Sharon Adler, MD, FASN
Rajiv Agarwal, MD
Sharon P Andreoli, MD
Gavin J Becker, MD, FRACP
Fred Brown, MBA, FACHE
Daniel C Cattran, MD, FRCPC
Allan J Collins, MD, FACP
Rosanna Coppo, MD
Josef Coresh, MD, PhD
Ricardo Correa-Rotter, MD
Adrian Covic, MD, PhD
Jonathan C Craig, MBChB, MM (Clin Epi), DCH, FRA CP, PhD
Angel LM de Francisco, MD
Paul E de Jong, MD, PhD
Ana Figueiredo, RN, MSc, PhD
Mohammed Benghanem Gharbi, MD
Gordon Guyatt, MD, MSc, BSc, FRCPC
David Harris, MD
Lai Seong Hooi, MD
Enyu Imai, MD, PhD
Lesley A Inker, MD, MS, FRCP
Michel Jadoul, MD
Simon Jenkins, MBE, FRCGP
Suhnggwon Kim, MD, PhD
Martin K Kuhlmann, MD
Nathan W Levin, MD, FACP
Philip K-T Li, MD, FRCP, FACP
Zhi-Hong Liu, MD
Pablo Massari, MD
Peter A McCullough, MD, MPH, FACC, FACP
Rafique Moosa, MD
Miguel C Riella, MD
Adibul Hasan Rizvi, MBBS, FRCP
Bernardo Rodriquez-Iturbe, MD
Robert Schrier, MD
Justin Silver, MD, PhD
Marcello Tonelli, MD, SM, FRCPC
Yusuke Tsukamoto, MD
Theodor Vogels, MSW
Angela Yee-Moon Wang, MD, PhD, FRCP
Christoph Wanner, MD
Elena Zakharova, MD, PhD
NKF-KDIGO GUIDELINE DEVELOPMENT STAFF
Kerry Willis, PhD, Senior Vice-President for Scientific Activities
Michael Cheung, MA, Guideline Development Director
Sean Slifer, BA, Guideline Development Manager
Kidney International Supplements (2013) 3, vii vii
http://www.kidney-international.org
& 2013 KDIGO

ADULT GFR ESTIMATING EQUATIONS
2009 CKD-EPI creatinine equation: 141 min(SCr/k,1)
a
max(SCr/k,1)
1.209
0.993
Age
[ 1.018 if female] [ 1.159 if black], where
SCr is serum creatinine (in mg/dl), k is 0.7 for females and 0.9 for males, a is 0.329 for females and 0.411 for males, min is the
minimum of SCr/k or 1, and max is the maximum of SCr/k or 1.
2012 CKD-EPI cystatin C equation: 133 min(SCysC/0.8, 1)
0.499
max(SCysC/0.8, 1)
1.328
0.996
Age
[ 0.932 if female], where
SCysC is serum cystatin C (in mg/l), min indicates the minimum of SCysC/0.8 or 1, and max indicates the maximum of SCysC/0.8 or 1.
Grade Quality of evidence Meaning
A High We are confident that the true effect lies close to that of the estimate of the effect.
B Moderate The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
C Low The true effect may be substantially different from the estimate of the effect.
D Very low The estimate of effect is very uncertain, and often will be far from the truth.
Implications
Grade* Patients Clinicians Policy
Level 1
‘We recommend’
Most people in your situation would want
the recommended course of action and only
a small proportion would not.
Most patients should receive the
recommended course of action.
The recommendation can be evaluated as a
candidate for developing a policy or a
performance measure.
Level 2
‘We suggest’
The majority of people in your situation
would want the recommended course of
action, but many would not.
Different choices will be appropriate for
different patients. Each patient needs help to
arrive at a management decision consistent
with her or his values and preferences.
The recommendation is likely to require
substantial debate and involvement of
stakeholders before policy can be
determined.
*The additional category ‘Not Graded’ was used, typically, to provide guidance based on common sense or where the topic does not allow adequate application of evidence.
The most common examples include recommendations regarding monitoring intervals, counseling, and referral to other clinical specialists. The ungraded recommendations
are generally written as simple declarative statements, but are not meant to be interpreted as being stronger recommendations than Level 1 or 2 recommendations.
NOMENCLATURE AND DESCRIPTION FOR RATING GUIDELINE RECOMMENDATIONS
Within each recommendation, the strength of recommendation is indicated as Level 1, Level 2,orNot Graded, and the quality of the
supporting evidence is shown as A, B, C,orD.
Reference Keys
Equations expressed for specified sex and serum creatinine level
Gender Serum creatinine Equation for estimating GFR
Female p0.7 mg/dl (p62 mmol/l) 144 (SCr/0.7)
0.329
0.993
Age
[ 1.159 if black]
Female 40.7 mg/dl (462 mmol/l) 144 (SCr/0.7)
1.209
0.993
Age
[ 1.159 if black]
Male p0.9 mg/dl (p80 mmol/ l) 141 (SCr/0.9)
0.411
0.993
Age
[ 1.159 if black]
Male 40.9 mg/dl (480 mmol/l) 141 (SCr/0.9)
1.209
0.993
Age
[ 1.159 if black]
Equations expressed for serum cystatin C level
Serum cystatin C Equation for estimating GFR
Female or male p0.8 mg/l 133 (SCysC/0.8)
0.499
0.996
Age
[ 0.932 if female]
Female or male 40.8 mg/l 133 (SCysC/0.8)
1.328
0.996
Age
[ 0.932 if female]
viii Kidney International Supplements (2013) 3, viii
http://www.kidney-international.org
& 2013 KDIGO

2012 CKD-EPI creatinine–cystatin C equation: 135 min(SCr/k,1)
a
max(SCr/k,1)
0.601
min(SCysC/0.8, 1)
0.375
max(SCysC/
0.8, 1)
0.711
0.995
Age
[ 0.969 if female] [ 1.08 if black], where SCr is serum creatinine (in mg/dl), SCysC is serum cystatin C (in mg/l),
k is 0.7 for females and 0.9 for males, a is 0.248 for females and 0.207 for males, min(SCr/k, 1) indicates the minimum of SCr/k or 1,
and max(SCr/k, 1) indicates the maximum of SCr/k or 1; min(SCysC/0.8, 1) indicates the minimum of SCysC/0.8 or 1 and max(SCysC/
0.8, 1) indicates the maximum of SCysC/0.8 or 1.
PEDIATRIC GFR ESTIMATING EQUATIONS
http://www.kidney-international.org
& 2013 KDIGO
Equations expressed for specified sex, serum creatinine, and serum cystatin C level
Gender Serum creatinine Serum cystatin C Equation for estimating GFR
Female p0.7 mg/ dl (p62 mmol/l) p0.8 mg/l
40.8 mg/l
130 (SCr/0.7)
0.248
(SCysC/0.8)
0.375
0.995
Age
[ 1.08 if black]
130 (SCr/0.7)
0.248
(SCysC/0.8)
0.711
0.995
Age
[ 1.08 if black]
Female 40.7 mg/dl (462 mmol/l) p0.8 mg/l
40.8 mg/l
130 (SCr/0.7)
0.601
(SCysC/0.8)
0.375
0.995
Age
[ 1.08 if black]
130 (SCr/0.7)
0.601
(SCysC/0.8)
0.711
0.995
Age
[ 1.08 if black]
Male p0.9 mg/ dl (p80 mmol/l) p0.8 mg/l
40.8 mg/l
135 (SCr/0.9)
0.207
(SCysC/0.8)
0.375
0.995
Age
[ 1.08 if black]
135 (SCr/0.9)
0.207
(SCysC/0.8)
0.711
0.995
Age
[ 1.08 if black]
Male 40.9 mg/dl (480 mmol/l) p0.8 mg/l
40.8 mg/l
135 (SCr/0.9)
0.601
(SCysC/0.8)
0.375
0.995
Age
[ 1.08 if black]
135 (SCr/0.9)
0.601
(SCysC/0.8)
0.711
0.995
Age
[ 1.08 if black]
Creatinine-based equations
41.3 (height/SCr)
40.7 (height/SCr)
0.64
(30/BUN)
0.202
BUN, blood urea nitrogen in mg/dl; height in meters; SCr, serum creatinine in mg/dl
Cystatin C-based equations
70.69 (SCysC)
0.931
SCysC, serum cystatin C in mg/l.
Kidney International Supplements (2013) 3,ix
ix

CURRENT CHRONIC KIDNEY DISEASE (CKD) NOMENCLATURE
USED BY KDIGO
CKD is defined as abnormalities of kidney structure or function, present for 43 months, with implications for
health and CKD is
classified based on cause, GFR category, and albuminuria category (CGA).
Persistent albuminuria categories
Description and range
A1 A2 A3
Normal to
mildly
increased
Moderately
increased
Severely
increased
<30 mg/g
<3 mg/mmol
30-300 mg/g
3-30 mg/mmol
>300 mg/g
>30 mg/mmol
GFR categories (ml/min/ 1.73 m
2
)
Description and range
G1 Normal or high ≥90
G2 Mildly decreased 60-89
G3a
Mildly to moderately
decreased
45-59
G3b
Moderately to
severely decreased
30-44
G4 Severely decreased 15-29
G5 Kidney failure <15
Prognosis of CKD by GFR
and Albuminuria Categories:
KDIGO 2012
Prognosis of CKD by GFR and albuminuria category
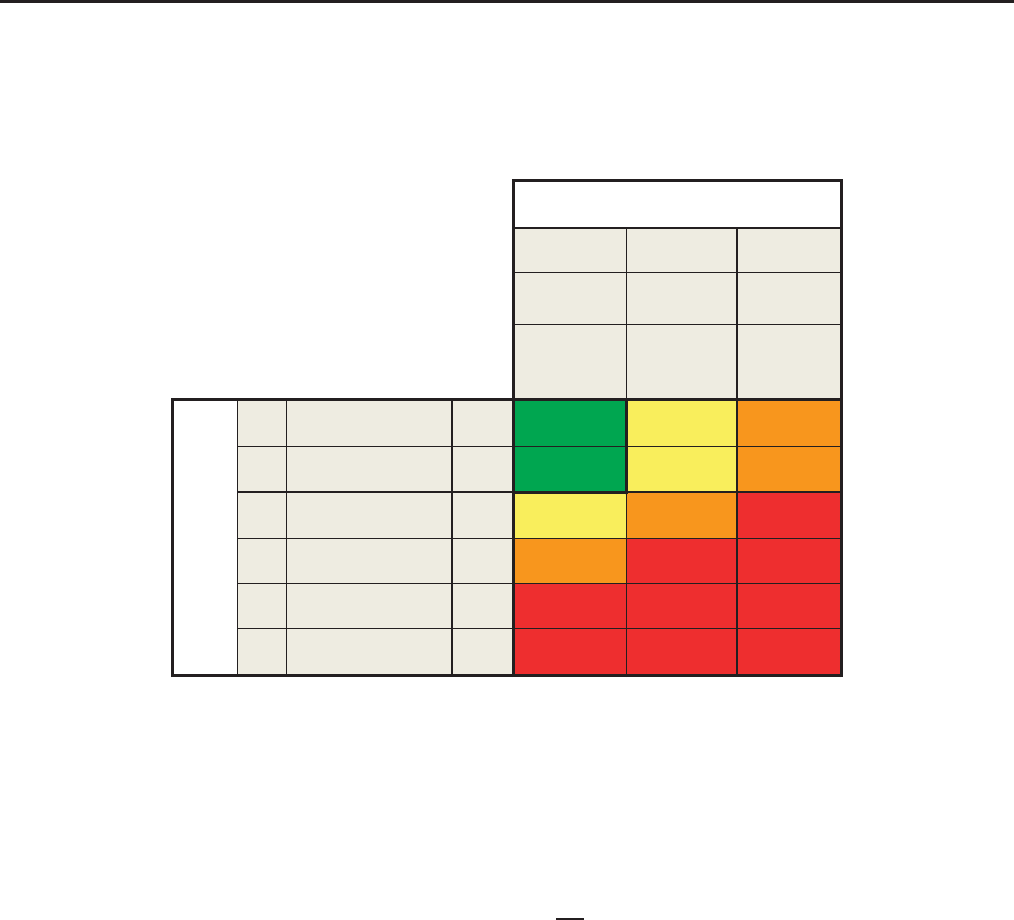
Green: low risk (if no other markers of kidney disease, no CKD); Yellow: moderately increased risk;
Orange: high risk; Red, very high risk.
http://www.kidney-international.org
& 2013 KDIGO
x
Kidney International Supplements (2013) 3,x

CONVERSION FACTORS OF METRIC UNITS TO SI UNITS
HbA
1C
CONVERSION CHART
DCCT
(%)
IFCC
(mmol/mol)
DCCT
(%)
IFCC
(mmol/mol)
DCCT
(%)
IFCC
(mmol/mol)
DCCT
(%)
IFCC
(mmol/mol)
DCCT
(%)
IFCC
(mmol/mol)
5.0 31 6.0 42 7.0 53 8.0 64 9.0 75
5.1 32 6.1 43 7.1 54 8.1 65 9.1 76
5.2 33 6.2 44 7.2 55 8.2 66 9.2 77
5.3 34 6.3 45 7.3 56 8.3 67 9.3 78
5.4 36 6.4 46 7.4 57 8.4 68 9.4 79
5.5 37 6.5 48 7.5 58 8.5 69 9.5 80
5.6 38 6.6 49 7.6 60 8.6 70 9.6 81
5.7 39 6.7 50 7.7 61 8.7 72 9.7 83
5.8 40 6.8 51 7.8 62 8.8 73 9.8 84
5.9 41 6.9 52 7.9 63 8.9 74 9.9 85
DCCT
(%)
IFCC
(mmol/mol)
DCCT
(%)
IFCC
(mmol/mol)
DCCT
(%)
IFCC
(mmol/mol)
DCCT
(%)
IFCC
(mmol/mol)
DCCT
(%)
IFCC
(mmol/mol)
10.0 86 11.0 97 12.0 108 13.0 119 14.0 130
10.1 87 11.1 98 12.1 109 13.1 120 14.1 131
10.2 88 11.2 99 12.2 110 13.2 121 14.2 132
10.3 89 11.3 100 12.3 111 13.3 122 14.3 133
10.4 90 11.4 101 12.4 112 13.4 123 14.4 134
10.5 91 11.5 102 12.5 113 13.5 124 14.5 135
10.6 92 11.6 103 12.6 114 13.6 125 14.6 136
10.7 93 11.7 104 12.7 115 13.7 126 14.7 137
10.8 95 11.8 105 12.8 116 13.8 127 14.8 138
10.9 96 11.9 107 12.9 117 13.9 128 14.9 139
IFCC-HbA
1c
(mmol/mol) ¼ [DCCT-HbA
1c
(%)2.15] 10.929
Abbreviations: DCCT, Diabetes Control and Complications Trial; IFCC, International Federation of Clinical Chemistry and Laboratory Medicine.
Source: Diabetes UK, www.diabetes.org.uk .
Parameter Metric units Conversion factor SI units
Albumin (serum) g/dl 10 g/l
Blood urea nitrogen (BUN) mg/dl 0.357 mmol/l
Creatinine (serum) mg/dl 88.4 mmol/l
Creatinine clearance ml/min 0.01667 ml/s
Hemoglobin g/dl 10 g/l
Phosphate (serum) mg/dl 0.323 mmol/l
PTH (parathyroid hormone, serum) pg/ml 0.106 pmol/l
Urea (plasma) mg/dl 0.167 mmol/l
Uric acid mg/dl 59.485 mmol/l
Vitamin D, 25-hydroxyvitamin D ng/ml 2.496 nmol/l
Note: Metric unit conversion factor ¼ SI unit.
Kidney International Supplements (2013) 3,xi
xi
http://www.kidney-international.org
& 2013 KDIGO

Kidney International Supplements (2013) 3, xii xii
Abbreviations and Acronyms
4C Cardio vascular Comorbidity in Children with CKD
AASK African American Study of Kidney Disease and
Hypertension
ABPM Ambulatory blood pressure monitoring
ACCORD Action to Control Cardiovascular Risk in Diabetes
ACE-I Angiotensin-converting enzyme inhibitor
ACR Albumin-to-creatinine ratio
ACS Acute coronary syndrome
ADVANCE Action in Diabetes and Vascular Disease:
Preterax and Diamicron Modified Release
Controlled Evaluation
AER Albumin excretion rate
AGREE Appraisal of Guidelines for Research and
Evaluation
AKD Acute kidney disease
AKDN Alberta Kidney Disease Network
AKI Acute kidney injury
ANP Atrial natriuretic peptide
APPROACH Alberta Provincial Project for Outcome Assess-
ment in Coronary Heart Disease
AusDiab Australian Diabetes, Obesity and Lifestyle study
ARB Angiotensin-receptor blocker
BMD Bone mineral density
BMI Body mass index
BNP B-type natriuretic peptide
BP Blood pressure
BSA Body surface area
BUN Blood urea nitrogen
CAD Coronary artery disease
CAPRICORN Carvedilol Post-Infarct Survival Control in Left
Ventricular Dysfunction
CGA
Cause, GFR and Albuminuria categories
CHARM Candesartan in Heart Failure-Assessment of
Reduction in Mortality and Morbidity
CHF Congestive heart failure
CHS Cardiovascular Health Study
CI Confidence interval
CIBIS II Cardiac Insufficiency Bisoprolol Study II
CKD Chronic kidney disease
CKD-EPI CKD Epidemiology Collaboration
CKD-MBD Chronic Kidney Disease-Mineral and Bone
Disorder
CKiD Chronic Kidney Disease in Children
COGS Conference on Guideline Standardization
COPERNICUS Carvedilol Prospective Randomized Cumulative
Survival
COX-2 Cyclooxygenase-2
Cr-EDTA Chromium-ethylenediamine tetraacetic acid
CREATE Cardiovascular Risk Reduction by Early Anemia
Treatment with Epoetin Beta Trial
CREDO Clopidogrel for Reduction of Events During
Observation
CrCl Creatinine clearance
CRIC Chronic Renal Insufficiency Cohort
CRP C-reactive protein
cTnI Cardiac troponin I
cTnT Cardiac troponin T
CVD Cardiovascular disease
DCCT/EDIC Diabetes Control and Complications Trial/
Epidemiology of Diabetes Interventions and
Complications
DIG Digitalis Intervention Group
DPI Dietary protein intake
DXA Dual-energ y x-ray absorptiometry
ECG Electrocardiography
eGFR Estimated GFR
EMU Early morning urine
ERT Evidence review team
ESA Erythropoiesis-stimulating agent
ESCAPE Effect of Strict Blood Pressure Control and
ACE-Inhibition on Progression of Chronic
Renal Failure in Pediatric Patients
ESRD End-Stage Renal Disease
ESUR European Society of Urogenital Radiology
FGF-23 Fibroblast growth factor-23
GBCA Gadolinium-based contrast agent
GFR Glomerular filtration rate
GN Glomerulonephritis
GRADE Grading of Recommendations Assessment,
Development and Evaluation
Hb Hemoglobin
HbA
1c
Hemoglobin A1c
HBV Hepatitis B virus
HDL-C High-density lipoprotein cholesterol
HOPE Heart Outcomes Prevention Evaluation
HOT Hypertension Optimal Treatment
HR Hazard ratio
HR-pQCT High-resolution peripheral quantitative computed
tomography
HUNT 2 Nord-Trøndelag Health Study (1995-1997)
ICD International Classification of Diseases
ICU Intensive-care unit
IDF International Diabetes Federation
IDMS Isotope-dilution mass spectrometry
IQR Interquartile range
IRMM Institute for Reference Materials and Measure-
ments
ItalKid Italian Pediatric Registry of Chronic Renal Failure
JCTLM Joint Committee for Traceability in Laboratory
Medicine
KDIGO Kidney Disease: Improving Global Outcomes
KDOQI Kidney Disease Outcomes Quality Initiative
LDL-C Low-density lipoprotein cholesterol
LIFE Losar tan Intervention For Endpoint Reduction
in Hypertension
LPD Low-protein diet
LVH Left ventricular hypertrophy
MAP Mean arterial pressure
MDRD Modification of Diet in Renal Disease
MESA Multi-Ethnic Study of Atherosclerosis
http://www.kidney-international.org
& 2013 KDIGO

MI Myocardial infarction
MRI Magnetic resonance imaging
NAPRTCS North American Pediatric Renal Trials and
Collaborative Studies
NCEP III Third Report of the National Cholesterol
Education Program
NECOSAD Netherlands Cooperative Study on the Adequacy
of Dialysis Study Group
NHANES National Health and Nutrition Examination Survey
NICE National Institute for Health and Clinical
Excellence
NIH National Institutes of Health
NKDEP National Kidney Disease Education Program
NKF National Kidney Foundation
NSAID Nonsteroidal anti-inflammatory drug
NSF Nephrogenic systemic fibrosis
NT-proBNP N-terminal pro-BNP
ONTARGET Ongoing Telmisartan Alone and in Combina-
tion with Ramipril Global Endpoint trial
OR Odds ratio
PAD Peripheral arterial disease
PCR Protein-to-creatinine ratio
PER Protein excretion rate
PICARD Program to Improve Care in Acute Renal Disease
PICODD Population, Intervention or Predictor, Compa-
rator, Outcome, study Design, and Duration of
follow-up
PREVEND Prevention of Renal and Vascular Endstage
Disease
PTH Parathyroid hormone
QOL Quality of life
RAAS Renin-angiotensin-aldosterone system
RBC Red blood cell
RCT Randomized controlled trial
RENAAL Reduction of Endpoints in NIDDM with the
Angiotensin II Antagonist Losartan
RR Relative risk
RRT Renal replacement therapy
SCORE Systematic COronary Risk Evaluation
SCr Serum creatinine
SCysC Serum cystatin C
SD Standard deviation
SEEK Study for the Evaluation of Early Kidney disease
SHARP Study of Heart and Renal Protection
SPECT Single photon emission computed tomography
SUA Serum uric acid
TREAT Trial to Reduce Cardiovascular Events with
Aranesp Therapy
UKPDS United Kingdom Prospective Diabetes Study
USA-PRC USA–People’s Republic of China Collaborative
Study of Cardiovascular and Cardiopulmonary
Epidemiology
USRDS US Renal Data System
VADT Veterans Affairs Diabetes Trial
Val-HeFT Valsartan Heart Failure Trial
VLPD Very low-protein diet
WBC White blood cell
WHO World Health Organization
http://www.kidney-international.org
& 2013 KDIGO
Kidney International Supplements (2013) 3, xiii xiii

Notice
Kidney International Supplements (2013) 3, 1; doi:10.1038/kisup.2012.73
SECTION I: USE OF THE CLINICAL PRACTICE GUIDELINE
This Clinical Practice Guideline document is based upon systematic literature searches last
conducted in June 2011, supplemented with additional evidence through November 2012. It is
designed to provide information and assist decision making. It is not intended to define a
standard of care, and should not be construed as one , nor should it be interpreted as prescribing
an exclusive course of management. Variations in practice will inevitably and appropriately occur
when clinicians take into account the needs of individual patients, available resources, and
limitations unique to an institution or ty pe of practice. Every health-care professional making
use of these recommendations is responsible for evaluating the appropriateness of applying them
in any particular clinical situation. The recommendations for research contained within this
document are general and do not imply a specific protocol.
SECTION II: DISCLOSURE
Kidney Disease: Improving Global Outcomes (KDIGO) makes every effort to avoid any actual or
reasonably perceived conflicts of interest that may arise as a result of an outside relationship or a
personal, professional, or business interest of a member of the Work Group. All members of the
Work Group are required to complete, sign, and submit a disclo sure and attestation form
showing all such relationships that might be perceived as or are actual conflicts of interest. This
document is updated annually and information is adjusted accordingly. All reported information
is published in its entirety at the end of this document in the Work Group members’ Biographic
and Dis closure Information section, and is kept on file at the National Kidney Foundation
(NKF), former Managing Agent for KDIGO.
http://www.kidney-international.org
& 2013 KDIGO
Copyright & 2012 by KDIGO. All rights reserved.
Single photocopies may be made for personal use as allowed by national copyright
laws. Special rates are available for educational institutions that wish to make photocopies
for non-profit educational use. No part of this publication may be reproduced, amended, or
transmitted in any form or by any means, electronic or mechanical, including photocopying,
recording, or any information storage and retrieval system, without explicit permission in
writing from KDIGO. Details on how to seek permission for reproduction or translation,
and further information about KDIGO’s permissions policies can be obtained by contacting
To the fullest extent of the law, neither KDIGO, Kidney Inte rnational Supplements, National
Kidney Foundation (KDIGO’s former Managing Agent) nor the authors, contributors, or
editors, assume any liability for any injury and/or damage to persons or property as a matter
of products liability, negligence or otherwise, or from any use or operation of any methods,
products, instructions, or ideas contained in the material herein.
Kidney International Supplements (2013) 3,1 1

Foreword
Kidney International Supplements (2013) 3, 2; doi:10.1038/kisup.2012.74
It is our hope that this document will serve several useful
purposes. Our primary goal is to improve patient care. We
hope to accomplish this, in the short term, by helping
clinicians know and better understand the evidence (or lack
of evidence) that determines current practice. By providing
comprehensive evidence-based recommendations, this guide-
line will also help define areas where evidence is lacking and
research is needed. Helping to define a research agenda is an
often neglected, but very important, function of clinical
practice guideline development.
We used the Grading of Recommendations Assessment,
Development and Evaluation (GRADE) system to ra te the
quality of evidence and the strength of recommendations.
In all, there were 12 (17.1%) recommendations in this
guideline for which the overall quality of evidence was
graded ‘A,’ whereas 36 (51.4%) were graded ‘B,’ 17 (24.3%)
were graded ‘C,’ and 5 (7.1%) were graded ‘D.’ Although
there are reasons other than quality of evidence to make
a grade 1 or 2 recommendation, in general, there is a
correlation between the quality of overall evidence and the
strength of the recommendation. Thus, there were 43
(62.3%) recommendations graded ‘1’ and 26 (37.7%) graded
‘2.’ There were 9 (13.0%) recommendations graded ‘1A,’ 23
(33.3%) were ‘1B,’ 10 (14.5%) were ‘1C,’ and 1 (1.4%) was
‘1D.’ There were 2 (2.9%) recommendations graded ‘2A,’
13 (18.8%) were ‘2B,’ 7 (10.1%) were ‘2C,’ and 4 (5.8%)
were ‘2D.’ There were 41 (37.3%) statements that were not
graded.
Some argue that recommendations should not be made
when evidence is weak. However, clinicians still need to make
decisions in their daily practice, and they often ask, ‘‘What do
the experts do in this setting?’’ We opted to give guidance,
rather than remain silent. These recommendations are often
rated with a low strength of recommendation and a low
quality of evidence, or were not graded. It is important for
the users of this guideline to be cognizant of this (see Notice).
In every case these recommendations are meant to be a place
for clinicians to start, not stop, their inquiries into specific
management questions pertinent to the patients they see in
daily practice.
We wish to thank the Work Group Co-Chairs, Drs. Adeera
Levin and Paul Stevens, along with all of the Work Group
members who volunteered countless hours of their time
developing this guideline. We also thank the Evidence Review
Team members and staff of the National Kidney Foundation
who made this project possible. Finally, we owe a special debt
of gratitude to the many KDIGO Board members and
individuals who volunteered time reviewing the guideline,
and making ver y helpful suggestions.
Bertram L Kasiske, MD David C Wheeler, MD, FRCP
KDIGO Co-Chair KDIGO Co-Chair
http://www.kidney-international.org
& 2013 KDIGO
2
Kidney International Supplements (2013) 3,2

Work Group Membership
Kidney International Supplements (2013) 3, 3; doi:10.1038/kisup.2012.75
http://www.kidney-international.org
& 2013 KDIGO
WORK GROUP CO-CHAIRS
Adeera Levin, MD, FRCPC
University of British Columbia
Vancouver, Canada
Paul E Stevens, MB, FRCP
East Kent Hospitals University
NHS Foundation Trust
Canterbury, United Kingdom
WORK GROUP
EVIDENCE REVIEW TEAM
Tufts Center for Kidney Disease Guideline Development and Implementation
Tufts Medical Center, Boston, MA, USA:
Katrin Uhlig, MD, MS, Project Director; Director, Guideline Development
Dana Miskulin, MD, MS, Staff Nephrologist
Amy Earley, BS, Project Coordinator
Shana Haynes, MS, DHSc, Research Assistant
Jenny Lamont, MS, Project Manager
In addition, support and supervision were provided by:
Ethan M Balk, MD, MPH; Program Director, Evidence Based Medicine
Rudy W Bilous, MD
Newcastle University and James Cook University Hospital
Middlesbrough, United Kingdom
Edmund J Lamb, PhD, FRCPath
East Kent Hospitals University
NHS Foundation Trust
Canterbury, United Kingdom
Josef Coresh, MD, PhD, MHS
Johns Hopkins University
Baltimore, USA
Andrew S Levey, MD
Tufts Medical Center
Boston, USA
Angel LM de Francisco, MD, PhD
Hospital Universitario Valdecilla
Santander, Spain
Miguel C Riella, MD, PhD, FACP
Evangelic University Hospital
Curitiba, Brazil
Paul E de Jong, MD, PhD
University Medical Center Groningen
Groningen, The Netherlands
Michael G Shlipak, MD, MPH
VA Medical Center, UCSF
San Francisco, USA
Kathryn E Griffith, BM, BS, MSc, MRCP, MRCGP
University Health Centre, York University
York, United Kingdom
Haiyan Wang, MD
Peking University First Hospital
Beijing, China
Brenda R Hemmelgarn, MD, PhD, FRCP(C)
University of Calgary
Alberta, Canada
Colin T White, MD, FRCPC
University of British Columbia
Vancouver, Canada
Kunitoshi Iseki, MD
University Hospital of the Ryukyus
Nishihara, Okinawa, Japan
Christopher G Winearls, MB, DPhil, FRCP
Oxford Radcliffe Hospitals NHS Trust
Oxford, United Kingdom
Kidney International Supplements (2013) 3,3 3

Abstract
Kidney International Supplements (2013) 3, 4; doi:10.1038/kisup.2012.76
The Kidney Disease: Improving Global Outcomes (KDIGO) 2012 Clinical Practice Guideline for
the Evaluation and Management of Chronic Kidney Disease (CKD) serves to update the 2002
KDOQI Clinical Practice Guidelines for Chronic Kidney Disease: Evaluation, Classification, and
Stratification following a decade of focused research and clinical practice in CKD. The document
aims to provide state-of-the-art guidance on the evaluation, management and treatment for all
patients with CKD. Specifically, the guideline retains the definition of CKD but presents an
enhanced classification framework for CKD; elaborates on the identification and prognosis of
CKD; discusses the management of progression and complications of CKD; and expands on the
continuum of CKD care: timing of specialist referral, ongoing management of people with
progressive CKD, timing of the initiation of dialysis, and finally the implementation of a
treatment program which includes comprehensive conservative management. The development
of the guideline followed an explicit process of evidence review and appraisal. Treatment
approaches are addressed in each chapter and guideline recommendations are based on
systematic reviews of relevant trials. Practical comments or statements which serve as
educational purposes are ungraded, but included as important information for the readership.
Appraisal of the quality of the evidence and the strength of recommendations followed the
GRADE approach. Ongoing areas of controversies, limitations of the evidence, and international
relevance are discussed and additional suggestions are provided for future research.
Keywords: Albuminuria; Chronic kidney disease; Classification; Clinical practice guideline;
Evidence-based recommendation; GFR; Glomerular filtration rate; KDIGO; Proteinuria;
Systematic review
CITATION
In citing this document, the following format should be used: Kidney Disease: Improving Global
Outcomes (KDIGO) CKD Work Group. KDIGO 2012 Clinical Practice Guideline for the
Evaluation and Management of Chronic Kidney Disease. Kidney inter., Suppl. 2013; 3: 1–150.
http://www.kidney-international.org
& 2013 KDIGO
4 Kidney International Supplements (2013) 3,4

Summary of Recommendation Statements
Kidney International Supplements (2013) 3, 5–14; doi:10.1038/kisup.2012.77
http://www.kidney-international.org
& 2013 KDIGO
Chapter 1: Definition and classification of CKD
1.1: DEFINITION OF CKD
1.1.1: CKD is defined as abnormalities of kidney structure or function, present for 4 3 months, with implications for
health. (Not Graded)
1.2: STAGING OF CKD
1.2.1: We recommend that CKD is classified based on cause, GFR category, and albuminuria category (CGA). (1B)
1.2.2: Assign cause of CKD based on presence or absence of systemic disease and the location w ithin the kidney of
observed or presumed pathologic-anatomic findings. (Not Graded)
1.2.3: Assign GFR categories as follows (Not Graded):
1.2.4: Assign albuminuria* categories as follows (Not Graded):
*
note that where albuminuria measurement is not available, urine reagent strip results can be substituted (Table 7)
1.3: PREDICTING PROGNOSIS OF CKD
1.3.1: In predicting risk for outcome of CKD, identify the following variables: 1) cause of CKD; 2) GFR category;
3) albuminuria category; 4) other risk factors and comorbid conditions. (Not Graded)
Criteria for CKD (either of the following present for 43 months)
Markers of kidney damage (one or more) Albuminuria (AER Z30 mg/24 hours; ACR Z30 mg/g [Z3 mg/mmol])
Urine sediment abnormalities
Electrolyte and other abnormalities due to tubular disorders
Abnormalities detected by histology
Structural abnormalities detected by imaging
History of kidney transplantation
Decreased GFR GFR o60 ml/min/1.73 m
2
(GFR categories G3a–G5)
Abbreviations: CKD, chronic kidney disease; GFR, glomerular filtration rate.
GFR categories in CKD
GFR category GFR (ml/min/1.73 m
2
) Terms
G1 Z90 Normal or high
G2 60–89 Mildly decreased*
G3a 45–59 Mildly to moderately decreased
G3b 30–44 Moderately to severely decreased
G4 15–29 Severely decreased
G5 o15 Kidney failure
Abbreviations: CKD, chronic kidney disease; GFR, glomerular filtration rate.
*Relative to young adult level
In the absence of evidence of kidney damage, neither GFR category G1 nor G2 fulfill the criteria for CKD.
Albuminuria categories in CKD
AER
ACR (approximate equivalent)
Category (mg/24 hours) (mg/mmol) (mg/g) Terms
A1 o30 o3 o30 Normal to mildly increased
A2 30-300 3-30 30-300 Moderately increased*
A3 4300 430 4300 Severely increased**
Abbreviations: AER, albumin excretion rate; ACR, albumin-to-creatinine ratio; CKD, chronic kidney disease.
*Relative to young adult level.
**Including nephrotic syndrome (albumi n excretion usually 42200 mg/24 hours [ACR 42220 mg/g; 4220 mg/mmol]).
Kidney International Supplements (2013) 3, 5–14
5
1.3.2: In people with CKD, use estimated risk of concurrent complications and future outcomes to guide decisions
for testing and treatment for CKD complications. (Not Graded)
1.3.3: In populations with CKD, group GFR and albuminuria categories with similar relative risk for CKD outcomes
into risk categories. (Not Graded)
1.4: EVALUATION OF CKD
1.4.1: Evaluation of chronicity
1.4.1.1: In people with GFR o60 ml/min/1.73 m
2
(GFR categories G3a-G5) or markers of kidney damage, review
past history and previous measurements to determine duration of kidney disease. (Not Graded)
K If duration is 43 months, CKD is confirmed. Follow recommendations for CKD.
K If duration is not 43 months or unclear, CKD is not confirmed. Patients may have CKD or acute kidney
diseases (including AKI) or both and tests should be repeated accordingly.
1.4.2: Evaluation of cause
1.4.2.1: Evaluate the clinical context, including personal and family history, social and environment al factors,
medications, physical examination, laboratory measures, imaging, and pathologic diagnosis to determine
the causes of kidney disease. (Not Graded)
1.4.3: Evaluation of GFR
1.4.3.1: We recommend using serum creatinine and a GFR estimating equation for initial assessment. (1A)
1.4.3.2: We suggest using additional tests (such as cystatin C or a clearance measu rement) for confirmatory testing
in specific circumstances when eGFR based on serum creatinine is less accurate. (2B)
1.4.3.3: We recommend that clinicians (1B):
K use a GFR estimating equation to derive GFR from serum creatinine (eGFR
creat
) rather than relying on
the serum creatinine concentration alone.
K understand clinical settings in which eGFR
creat
is less accurate.
1.4.3.4: We recommend that clinical laboratories should (1B):
K measure serum creatinine using a specific assay with calibration traceable to the international standard reference
materials and minimal bias compared to isotope-dilution mass spectrometry (IDMS) reference methodology .
K report eGFR
creat
in addition to the serum creatinine concentration in adult s and specify the equation
used whenever reporting eGFR
creat
.
Persistent albuminuria categories
Description and range
A1 A2 A3
Normal to
mildly
increased
Moderately
increased
Severely
increased
<30 mg/g
<3 mg/mmol
30-300 mg/g
3-30 mg/mmol
>300 mg/g
>30 mg/mmol
GFR categories (ml/min/ 1.73 m
2
)
Description and range
G1 Normal or high ≥90
G2 Mildly decreased 60-89
G3a
Mildly to moderately
decreased
45-59
G3b
Moderately to
severely decreased
30-44
G4 Severely decreased 15-29
G5 Kidney failure <15
Prognosis of CKD by GFR
and Albuminuria Categories:
KDIGO 2012
Green: low risk (if no other markers of kidney disease, no CKD); Yellow: moderately increased risk; Orange: high risk; Red, very high risk.
6 Kidney International Supplements (2013) 3,5–14
summary of recommendation statements

K report eGFR
creat
in adults using the 2009 CKD-EPI creatinine equation. An alternative creatinine-based
GFR estimating equa tion is acceptable if it has been shown to improve accuracy of GFR estimates
compared to the 2009 CKD-EPI creatinine equation.
When reporting serum creatinine:
K We recommend that serum creatinine concentration be reported and rounded to the nearest whole
number when expressed as standard international units (lmol/l) and rounded to the nearest 100
th
of a
whole number when expressed as conventional units (mg/dl).
When reporting eGFR
creat
:
K We recommend that eGFR
creat
should be reported and rounded to the nearest whole number and
relative to a body surface area of 1.73 m
2
in adults using the units ml/min/1.73 m
2
.
K We recommend eGFR
creat
levels less than 60 ml/min/1.73 m
2
should be reported as ‘‘decreased.’’
1.4.3.5: We suggest measuring cystatin C in adults w ith eGFR
creat
45–59 ml/min/1.73 m
2
who do not have markers
of kidney damage if confirmation of CKD is requir ed. ( 2C)
K If eGFR
cys
/eGFR
creat-cys
is also o60 ml/min/1.73 m
2
, the diagnosis of CKD is confirmed.
K If eGFR
cys
/eGFR
creat-cys
is Z60 ml/min/1.73 m
2
, the diagnosis of CKD is not confirmed.
1.4.3.6: If cystatin C is measured, we suggest that health professionals (2C):
K use a GFR estimating equation to derive GFR from serum cystatin C rather than relying on the serum
cystatin C concentration alone.
K understand clinical settings in which eGFR
cys
and eGFR
creat-cys
are less accurate.
1.4.3.7: We recommend that clinical laboratories that measure cystatin C should (1B):
K measure serum cystatin C using an assay w ith calibration traceable to the international standard
reference material.
K report eGFR from serum cystatin C in add ition to the serum cystatin C concentration in adults and
specify the equation used whenever reporting eGFR
cys
and eGFR
creat-cys
.
K report eGFR
cys
and eGFR
creat-cys
in adults using the 2012 CKD-EPI cystatin C and 2012 CKD-EPI
creatinine-cystatin C equa tions, respectively, or alternative cystatin C-based GFR estimating
equations if they have been shown to improve accuracy of GFR estimates compared to the 2012
CKD-EPI cystatin C and 2012 CKD-EPI creatinine-cystatin C equations.
When reporting serum cystatin C:
K We recommend reporting serum cystatin C concentration rounded to the nearest 100
th
of a whole
number when expressed as conventional units (mg/l).
When reporting eGFR
cys
and eGFR
creat-cys
:
K We recommend that eGFR
cys
and eGFR
creat-cys
be reported and rounded to the nearest whole number
and relative to a body surface area of 1.73 m
2
in adults using the units ml/min/ 1.73 m
2
.
K We recommend eGFR
cys
and eGFR
creat-cys
levels less than 60 ml/min/1.73 m
2
should be reported as
‘‘decreased.’’
1.4.3.8: We suggest measuring GFR using an exogenous filtration marker under circumstances where more
accurate ascertainment of GFR will impact on treatment decisions. (2B)
1.4.4: Evaluation of albuminuria
1.4.4.1: We suggest using the following measurements for initial testin g of proteinuria (in descending order of
preference, in all cases an early morning urine sample is preferred) (2B):
1) urine albumin-to-creatinine ratio (ACR);
2) urine protein-to-creatinine ratio (PCR);
3) reagent strip urinalysis for total protein with automated reading;
4) reagent strip urinalysis for total protein with manual reading.
1.4.4.2: We recommend that clinical laboratories report ACR and PCR in untimed urine samples in addition to
albumin concentration or proteinuria concentrations rather than the concentrations alone. (1B)
1.4.4.2.1: The term microalbum inuria should no longer be used by laboratories. (Not Graded)
Kidney International Supplements (2013) 3, 5–14 7
summary of recommendation statements

1.4.4.3: Clinicians need to understand settings that may affect interpretation of measurements of albuminuria
and order confirmatory tests as indicated (Not Graded):
K Confirm reagent strip positive albuminuria and proteinuria by quantitative laboratory measurement
and express as a ratio to creatinine wherever possible.
K Confirm ACRZ30 mg/g (Z3 mg/mmol) on a random untimed urine with a subsequent early
morning urine sample.
K If a more accurate estimate of albuminuria or total proteinuria is required, measure albumin
excretion rate or total protein excretion rate in a timed urine sample.
1.4.4.4: If significant non-albumin proteinuria is suspected, use assays for specific urine proteins (e.g., a
1
-micro-
globulin, monoclonal heavy or light chains, [known in some countries as ‘‘Benc e Jon es’’ proteins]). (Not Graded)
Chapter 2: Definition, identification, and prediction of
CKD progression
2.1: DEFINITION AND IDENTIFICATION OF CKD PROGRESSION
2.1.1: Assess GFR and album inuria at least annually in people with CKD. Assess GFR and albuminuria more often
for individuals at higher risk of prog ression, and/or where measurement will impact therapeutic decisions
(see figure below). (Not Graded)
2.1.2: Recognize that small fluctuations in GFR are common and are not necessarily indicative of progression.
(Not Graded)
2.1.3: Define CKD progression based on one of more of the following (Not Graded):
K Decline in GFR category (Z90 [G1], 60–89 [G2], 45–59 [G3a], 30–44 [G3b], 15–29 [G4], o15 [G5] ml/min/
1.73 m
2
). A certain drop in eGFR is defined as a drop in GFR catego ry accompanied by a 25% or greater
drop in eGFR from baseline.
K Rapid progression is defined as a sustained decline in eGFR of mo re than 5 ml/min/1.73 m
2
/yr.
K The confidence in assessing progression is increased with increasing number of serum creatinine
measurements and duration of follow-up.
2.1.4: In people with CKD progression, as defin ed in Recommendation 2.1.3, review current management, examine
for reversible causes of progression, and consider referral to a specialist. (Not Graded)
Guide to Frequency of Monitoring
(number of times per year) by
GFR and Albuminuria Category
Persistent albuminuria categories
Description and range
A1 A2 A3
GFR categories (ml/min/1.73 m
2
)
Description and range
G1
Normal or high ≥90 1 if CKD 1 2
G2
Mildly decreased 60–89 1 if CKD 1 2
G3a
Mildly to moderately
decreased
45–59 1 2 3
G3b
Moderately to
severely decreased
30–44 2 3 3
G4
Severely decreased 15–29 3 3 4+
G5
Kidney failure <15 4+ 4+ 4+
Normal to
mildly
increased
Moderately
increased
Severely
increased
<30 mg/g
<3 mg/mmol
30–300 mg/g
3–30 mg/mmol
>300 mg/g
>30mg/mmol
GFR and albuminuria grid to reflect the risk of progression by intensity of coloring (green, yellow, orange, red, deep red). The numbers in
the boxes are a guide to the frequency of monitoring (number of times per year).
8 Kidney International Supplements (2013) 3,5–14
summary of recommendation statements

2.2: PRED ICTORS OF PROGRESSION
2.2.1: Identify factors associated with CKD progression to inform prognosis. These include cause of CKD,
level of GFR, level of albuminuria, age, sex, race/ethnicity, elevated BP, hyperglycemia, dyslipidemia,
smoking, obesity, history of cardi ovascular disease, ongoing exposure to nephrotoxic agents, and others.
(Not Graded)
Chapter 3: Management of progression and
complications of CKD
3.1: PREVENTI ON OF CKD PROGRESSION
BP and RAAS interruption
3.1.1: Individualize BP targets and agents according to age, coexistent cardiovascular disease and other comorbidities,
risk of progression of CKD, presence or absence of retinopathy (in CKD patients with dia betes), and tolerance
of treatment as described in the KDIGO 2012 Blood Pressure Guideline. (Not Graded)
3.1.2: Inquire about postural dizziness and check for postural hypotension regularly when treating CKD patients with
BP-lowering drugs. (Not Graded)
3.1.3: Tailor BP treatment regimens in elderly patients with CKD by carefully considering age, comorbidities and
other therapies, with gradual escalation of treatment and close attention to adverse events related to BP
treatment, including electrolyte disorders, acute deterioration in kidney function, orthostatic hypotension and
drug side effects. (Not Graded)
3.1.4: We recommend that in both diabetic and non-diabetic adults with CKD and urine albumin excretion o30 mg/
24 hours (or equivalent*) whose office BP is consistently 4140 mm Hg systolic or 490 mm Hg diastolic be
treated with BP-lowering drugs to maintain a BP that is consistently r140 mm Hg systolic and r90 mm Hg
diastolic. (1B)
3.1.5: We suggest that in both diabetic and non-diabetic adults with CKD and with urine albumin excretion of
Z30 mg/24 hours (or equivalent*) whose office BP is consistently 4130 mm Hg systolic or 480 mm Hg
diastolic be treated with BP-lowering drugs to maintain a BP that is consistently r130 mm Hg systolic and
r80 mm Hg diastolic. (2D)
3.1.6: We suggest that an ARB or ACE-I be used in diabetic adults with CKD and urine albumin excretion 30–300 mg/
24 hours (or equivalent*). (2D )
3.1.7: We recommend that an ARB or ACE-I be used in both diabetic and non-diabetic adults with CKD and urine
albumin excretion 4300 mg/24 hours (or equivalent*). (1B)
3.1.8: There is insufficient ev idence to recommend combining an ACE-I with ARBs to prevent progression of CKD.
(Not Graded)
3.1.9: We recommend that in children with CKD, BP-lowering treatment is started when BP is consistently above the
90
th
percentile for age, sex, and height. (1C)
3.1.10:We suggest that in children with CKD (particularly those with proteinuria), BP is lowered to consistently
achieve systolic and diasto lic readings less than or equal to the 50
th
percentile for age, sex, and height, unless
achieving these targ ets is limited by signs or symptoms of hypotension. (2D)
3.1.11:We suggest that an ARB or ACE-I be used in children with CKD in whom treatment with BP-lowering drugs is
indicated, irrespective of the level of proteinuria. (2D)
*Approximate equivalents for albumin excretion rate per 24 hours—expressed as protein excretion rate per 24 hours, albumin-to-creatinine ratio, protein-to-
creatinine ratio, and protein reagent strip results— are given in Table 7, Chapter 1.
CKD and risk of AKI
3.1.12: We recommend that all people w ith CKD are considered to be at increased risk of AKI. (1A)
3.1.12.1: In people with CKD, the recommendations detailed in the KDIGO AKI Guideline should be
followed for management of those at risk of AKI during intercurrent illness, or when undergoing
investigation and procedures that are likely to increase the risk of AKI. (Not Graded)
Kidney International Supplements (2013) 3, 5–14 9
summary of recommendation statements

Protein intake
3.1.13: We suggest lowering protein intake to 0.8 g/kg/day in adults with diabetes (2C) or without diabetes ( 2B ) and
GFR o30 ml/min/ 1.73 m
2
(GFR categories G4-G5), with appropriate education.
3.1.14: We suggest avoiding high protein intake (41.3 g/kg/day) in adults with CKD at risk of progression. (2C)
Glycemic cont rol
3.1.15: We recommend a target hemoglobin A
1c
(HbA
1c
)ofB 7.0% (53 mmol/mol) to prevent or delay progression of
the microvascular complications of diabetes, including diabetic kidney disease. (1A)
3.1.16: We recommend not treating to an HbA
1c
target of o7.0% (o53 mmol/mol) in patients at risk of
hypoglycemia. (1B)
3.1.17: We suggest that target HbA
1c
be extended above 7.0% (53 mmol/mol) in individuals with comorbidities or
limited life expectancy and risk of hypoglycemia. (2C)
3.1.18: In people with CKD and diabetes, glycemic control should be part of a multifactorial intervention strategy
addressing blood pressure control and cardiovascular risk, prom oting the use of angiotensin -converting
enzyme inhibition or angiotensin receptor blockade, statins, and antiplatelet therapy where clinically
indicated. (Not Graded)
Salt intake
3.1.19: We recommend lowering salt intake to o 90 mmol (o2 g) per day of sodium (corresponding to 5 g of sodium
chloride) in ad ults, unless contraindicated (see rationale). (1C)
3.1.19.1: We recommend restriction of sodium intake for children with CKD who have hypertension (systolic and/
or diastolic blood pressure 495
th
percentile) or prehypertension (systolic and/or diastolic blood pressure
490
th
percentile and o95
th
percentile), following the age-based Recommended Daily Intake. (1C)
3.1.19.2: We recommend supplemental free water and sodium supplements for children with CKD and polyuria to
avoid chronic intravascular depletion and to promote optimal growth. (1C)
Hyperuricem ia
3.1.20: There is insufficient evidence to suppor t or refute the use of agents to lower serum uric acid concentrations in
people with CKD and either symptomatic or asymptomatic hyperuricemia in order to delay progression of CKD.
(Not Graded)
Lifestyle
3.1.21: We recommend that people with CKD be encouraged to undertake physical activity compatible w ith
cardiovascular health and tolerance (aiming for at least 30 minutes 5 times per week), achieve a healthy weight
(BMI 20 to 25, according to country specific demographics), and stop smoking. (1D)
Additional dietary advice
3.1.22: We recommend that individuals with CKD receive expert dietary advice and information in the context of an
education program, tailored to severity of CKD and the need to intervene on salt, phosphate, potassium, and
protein intake where indicated. (1B)
3.2: COMPLICATIONS ASSOCIATED WITH LOSS OF KIDNEY FUNCTION
Definition and identification of anemia in CKD
3.2.1: Diagnose anemia in adults and children 415 years with CKD when the Hb concentration is o13.0 g/dl
(o130 g/l) in males and o12.0 g/dl (o120 g/ l) in females. (Not Graded)
3.2.2: Diagnose anemia in children with CKD if Hb concentration is o11.0 g/dl (o110 g/l) in children 0.5–5 years,
o11.5 g/dl (115 g/l) in chil dren 5–12 years, and o12.0 g/dl (120 g/l) in children 12-15 years. (Not Graded)
Evaluation of an emia in people with CKD
3.2.3: To identify anemia in people with CKD measure Hb concentration (Not Graded):
K when clinically indicated in people with GFR Z60 ml/min/1.73 m
2
(GFR categories G1-G2);
K at least annually in people with GFR 30–59 ml/min/1.73 m
2
(GFR categories G3a-G3b);
K at least twice per year in people with GFRo30 ml/min/1.73 m
2
(GFR categories G4-G5).
10 Kidney International Supplements (2013) 3,5–14
summary of recommendation statements

3.3: CKD METABOLIC BONE DISEASE INCLUDING LABORATORY ABNORMALITIES
3.3.1: We recommend measuring serum levels of calcium, phosphate, PTH, and alkaline phosphatase activity at least
once in adults with GFR o45 ml/min/1.73 m
2
(GFR categories G3b-G5) in order to determine baseline values
and inform prediction equations if used. (1C)
3.3.2: We suggest not to perform bone mineral density testing routinely in those with eGFR o45 ml/min/1.73 m
2
(GFR categories G3b-G5), as information may be misleading or unhelpful. (2B)
3.3.3: In people with GFR o45 ml/min/1.73 m
2
(GFR categories G3b-G5), we suggest maintaining serum phosphate
concentrations in the normal range according to local laboratory reference values. (2C)
3.3.4: In people with GFR o45 ml/min/1.73 m
2
(GFR categories G3b-G5) the optimal PTH level is not known.
We suggest that people with levels of intact PTH above the upper normal limit of the assay are first evaluated
for hyperphosphatemia, hypocalcemia, and vitamin D deficiency. (2C)
Vitamin D supplementation and bisphosphonates in people with CKD
3.3.5: We suggest not to routinely prescribe vitamin D supplements or vitamin D analogs, in the absence of suspected or
documented deficiency, to suppress elevated PTH concentrations in people with CKD not on dialysis. (2B)
3.3.6: We suggest not to prescribe bisphosphonate treatment in people with GFR o30 ml/min/1.73 m
2
(GFR categories
G4-G5) without a strong clinical rationale. (2B)
3.4: ACIDOSIS
3.4.1: We suggest that in people with CKD and serum bicarbonate concentrations o22 mmol/l treatment with oral
bicarbonate supplementation be given to maintain serum bicarbonate within the normal range, unless
contraindicated. (2B)
Chapter 4: Other complications of CKD: CVD,
medication dosage, patient safety, infections,
hospitalizations, and caveats for investigating
complications of CKD
4.1:CKD AND CVD
4.1.1: We recommend that all people with CKD be considered at increased risk for cardiovascular disease. (1A)
4.1.2: We recommend that the level of care for ischemic heart disease offered to people with CKD should not be
prejudiced by their CKD. (1A)
4.1.3: We suggest that adults with CKD at risk for atherosclerotic events be offered treatment with antiplatelet agents
unless there is an increased bleeding risk that needs to be balanced against the possible cardiovascular benefits. (2B)
4.1.4: WesuggestthatthelevelofcareforheartfailureofferedtopeoplewithCKDshouldbethesameasisofferedto
those without CKD. (2A)
4.1.5: In people with CKD and heart failure, any escalation in therapy and/or clinical deterioration should prompt
monitoring of eGFR and serum potassium concentration. (Not Graded)
4.2: CAVEATS WHEN INTERPRETING TESTS FOR CVD IN PEOPLE WITH CKD
BNP/N-terminal-proBNP (NT-proBNP)
4.2.1: In people with GFR o60 ml/min/1.73 m
2
(GFR categories G3a-G5), we recommend that serum concentrations
of BNP/NT-proBNP be interpreted w ith caution and in relation to GFR with respect to diagnosis of heart
failure and assessment of volume status. (1B)
Troponins
4.2.2: In people with GFRo60 ml/min/1.73 m
2
(GFR categories G3a-G5), we recommend that serum concentrations
of troponin be interpreted with caution with respect to diagnosis of acute coronary syndrome. (1B)
Kidney International Supplements (2013) 3, 5–14 11
summary of recommendation statements

Non-invasive testing
4.2.3: We recommend that people with CKD presenting with chest pain should be investigated for underlying
cardiac disease and other disorders according to the same local practice for people without CKD (and
subsequent treatment should be initiated similarly). (1B)
4.2.4: We suggest that clinicians are familiar with the limitations of non-invasive cardiac tests (e.g., exercise
electrocardiography [ECG], nuclear imaging, echocardiography, etc.) in adults with CKD and interpret the
results accordingly. (2B)
4.3: CKD AND PERIPHERAL ARTERIAL DISEASE
4.3.1: We recommend that adults with CKD be regularly examined for signs of peripheral arterial disease and be
considered for usual approaches to therapy. (1B)
4.3.2: We suggest that adults with CKD and diabetes are offered regular podiatric assessment. (2A)
4.4: MEDICATION MANAGEMENT AND PATIENT SAFETY IN CKD
4.4.1: We recommend that prescribers should take GFR into account when dr ug dosing. (1A)
4.4.2: Where precision is required for dosing (due to narrow therapeutic or toxic range) and/or estimates may be unreliable
(e.g., due to low muscle mass), we recommend methods based upon cystatin C or direct measurement of GFR. (1C)
4.4.3: We rec ommend temporary disc ontinuation of potentially nephrotoxic and renally excreted drugs in people with a
GFR o60 ml/min/1.73 m
2
(GFR cate go rie s G3a-G 5) who hav e seriou s inter curr en t illn ess that inc re ase s the risk of
AKI. These agents include, but are not limited to: RAAS blockers (including ACE-I s, ARBs, aldosterone inhibitors,
direct renin inhibitors), diuretics, NSAIDs, metformin, lithium, and digoxin. (1C)
4.4.4: We recommend that adults with CKD seek medical or pharmacist advice before using ov er-the-c ounter medicines or
nutritional protein supplements. (1B)
4.4.5: We recommend not using herbal remedies in people with CKD . (1B)
4.4.6: We recommend that metformin be continued in people with GFR Z45 ml/min/1.73 m
2
(GFR categories G1-G3a);
its use should be reviewed in those with GFR 30–44 ml/min/1.73 m
2
(GFR category G3b); and it should be
discontinued in people with GFR o30 ml/min/1.73 m
2
(GFR categories G4-G5). (1C)
4.4.7: We recommend that all people taking potentially nephrotoxic agents such as lithium and calcineurin
inhibitors should have their GFR, electrolytes and drug levels regularly monitored. (1A)
4.4.8: People with CKD should not be denied therapies for other conditions such as cancer but there shoul d be
appropriate dose adjustment of cytotoxic drugs according to knowledge of GFR. (Not Graded)
4.5: IMAGING STUDIES
4.5.1: Balance the risk of acute impairment in kidney function due to contrast agent use against the diag nostic value
and therapeutic implications of the investigation. (Not Graded)
Radiocontrast
4.5.2: We recommend that all people with GFR o60 ml/min/1.73 m
2
(GFR categories G3a-G5) undergoing elective
investigation involving the intravascular administration of iodinated radiocontrast media should be managed
according to the KDIGO Clinical Practice Guideline for AKI including:
K Avoidance of high osmolar agents (1B );
K Use of lowest possible radiocontrast dose (Not Graded);
K Withdrawal of potentially nephrotoxic agents before and after the procedure (1C);
K Adequate hydration with saline before, during, and after the procedure (1A);
K Measurement of GFR 48–96 hours after the procedure (1C).
Gadolinium-based contrast media
4.5.3: We recommend not using gadolinium-containing contrast media in people with GFR o15 ml/min/1.73 m
2
(GFR category G5) unless there is no alternative appropriate test. (1B)
4.5.4: We suggest that people w ith a GFR o30 ml/min/1.73 m
2
(GFR categories G4-G5) who require gadolinium-
containing contrast media are preferentially offered a macrocyclic chelate preparation. (2B)
Bowel preparation
4.5.5: We recommend not to use oral phosphate-containing bowel preparations in people with a GFR o60 ml/min/
1.73 m
2
(GFR categories G3a-G5) or in thos e known to be at risk of phosphate nephropathy. ( 1A)
12 Kidney International Supplements (2013) 3,5–14
summary of recommendation statements

4.6: CKD AND RISKS FOR INFECTIONS, AKI, HOSPITALIZATIONS, AND MORTALITY
CKD and risk of infections
4.6.1: We recommend that all adult s with CKD are offered annual vaccination with influenza vaccine, unless
contraindicated. (1B)
4.6.2: We recommend that all adults with eGFR o30 ml/min/1.73 m
2
(GFR categories G4-G5) and those at high risk
of pneumococcal infection (e.g., nephrotic syndrome, diabetes, or those receiving immunosuppression) receive
vaccination with polyvalent pneumococcal vaccine un less contraindicated. (1B)
4.6.3: We recommend that all adults with CKD who have received pneumococcal vaccination are offered
revaccination within 5 years. (1B)
4.6.4: We recommend that all adults who are at high risk of progression of CKD and have GFR o30 ml/min/1.73 m
2
(GFR categories G4-G5) be immunized ag ainst hepatitis B and the response confirmed by appropriate
serological testing. (1B)
4.6.5: Consideration of live vaccine should include an appreciation of the patient’s immune status and should be in
line with recommendations from official or governmental bodies. (Not Graded)
4.6.6: Pediatric immunization schedules should be followed according to official international and regional
recommedations for children with CKD. (Not Graded)
CKD and risk of AKI
4.6.7: We recommend that all people with CKD are considered to be at increased risk of AKI. (1A)
4.6.7.1: In people with CKD, the recommendations detailed in the KDIGO AKI Guideline should be followed
for management of those at risk of AKI during intercurrent illness, or when undergoing investigation
and procedures that are likely to increase the risk of AKI. (Not Graded)
CKD and risk of hospitalization and mortality
4.6.8: CKD disease management programs should be developed in order to optimize the community management of
people with CKD and reduce the risk of hospital admission. (Not Graded)
4.6.9: Interventions to reduce hospitalization and mortality for people with CKD should pay close attention to the
management of associat ed comor bid conditions and cardi ovascular disease in particular. (Not Graded)
Chapter 5: Referral to specialists and models of care
5.1: REFERRAL TO SPECIALIST SERVICES
5.1.1: We recommend referral to specialist kidney care services for people with CKD in the following circumstances (1B):
K AKI or abrupt sustained fall in GFR;
K GFR o30 ml/min/1.73 m
2
(GFR categories G4-G5)*;
K a consistent finding of significant albuminuria (ACR Z300 mg/g [Z30 mg/mmol] or AER Z300 mg/
24 hours, approximately equivalent to PCR Z500 mg/g [Z50 mg/mmol] or PER Z500 mg/24 hours);
K progression of CKD (see Recommendation 2.1.3 for definition);
K urinary red cell casts, RBC 420 per high power field sustained and not readily explained;
K CKD and hypertension refractory to treatment with 4 or more antihypertensive agents;
K persistent abnormalities of serum potassium;
K recurrent or extensive nephrolithiasis;
K hereditary kidney disease.
5.1.2: We recommend timely referral for planning renal replacement therapy (RRT) in people with progressive CKD
in whom the risk of kidney failure within 1 year is 10–20% or higher
w
, as determined by validated risk
prediction tools. (1B)
*If this is a stable isolated finding, formal referral (i.e., formal consultation and ongoing care management) may not be necessary and advice
from specialist services may be all that is required to facilitate best care for the patients. This will be health-care system dependent.
w
The aim is to avoid late referral, defined here as referral to specialist services less than 1 year before start of RRT.
Kidney International Supplements (2013) 3, 5–14 13
summary of recommendation statements
5.2: CARE OF THE PA TIENT WITH PROGRE SSIVE CKD
5.2.1: We suggest that people with progressive CKD should be managed in a multidisciplinary care setting. (2B)
5.2.2: The multidisciplinary team should include or have access to dietary counseling, education and counseling
about different RRT modalities, transplant options, vascular access surgery, and ethical, psychological, and
social care. (Not Graded)
5.3: TIMING THE INITIATION OF RRT
5.3.1: We suggest that dialysis be initiated when one or more of the following are present: symptoms or signs
attributable to kidney failure (serositis, acid-base or electrolyte abnormalities, pruritus); inability to control
volume status or blood pressure; a progressive deterioration in nutritional status refractory to dietary
intervention; or cognitive impairment. This often but not invariably occurs in the GFR range between 5 and
10 ml/min/1.73 m
2
.(2B)
5.3.2: Living donor preemptive renal transplantation in adults should be considered when the GFR is o20 ml/min/
1.73 m
2
, and there is evidence of progressive and irreversible CKD over the preceding 6–12 months.
(Not Graded)
5.4: STRUCTURE AND PROCESS OF COMPREHENSIVE CONSERVATIV E MANAGEMENT
5.4.1: Conservative management should be an option in people who choose not to pursue RRT and this should be
supported by a comprehensive management program. (Not Graded)
5.4.2: All CKD programs and care providers should be able to deliver advance care planning for people with
a recognized need for end-of-life care, including those people undergoing conservative kidney care. (Not
Graded)
5.4.3: Coordinated end-of-life care should be available to peop le and families through either primary care or
specialist care as local circumstances dictate. (Not Graded)
5.4.4: The comprehensive conservative management program should include protocols for symptom and pain
management, psychological care, spiritual care, and culturally sensitive care for the dying patient and their
family (whether at home, in a hospice or a hospital setting), followed by the provision of culturally
appropriate bereavement support. (Not Graded)
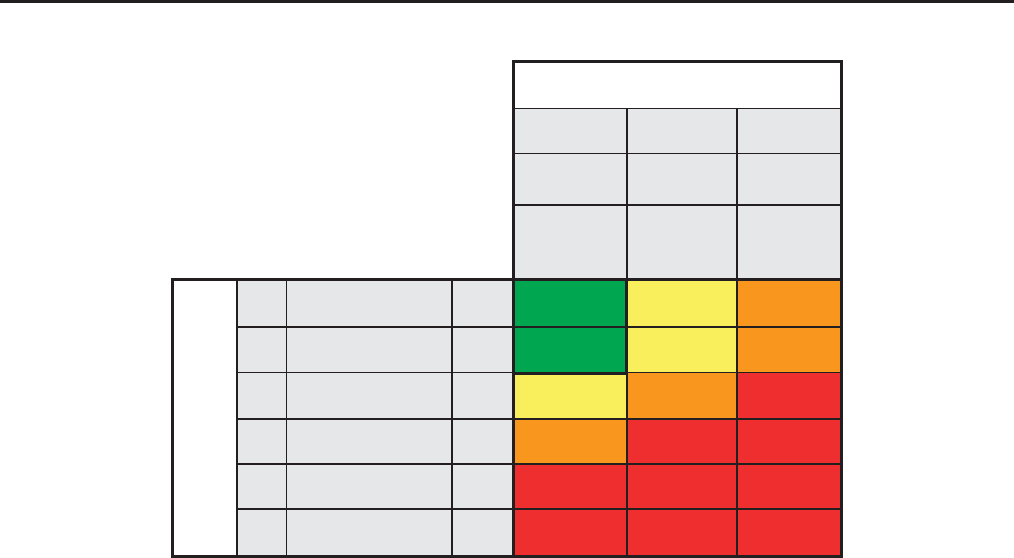
Persistent albuminuria categories
Description and range
A1 A2 A3
Normal to
mildly
increased
Moderately
increased
Severely
increased
GFR categories (ml/min/ 1.73 m
2
)
Description and range
G1 Normal or high ≥90 Monitor Refer*
G2 Mildly decreased 60–89 Monitor Refer*
G3a 45–59 Monitor Monitor Refer
G3b 30–44 Monitor Monitor Refer
G4 Severely decreased 15–29 Refer* Refer* Refer
G5 Kidney failure <15 Refer Refer Refer
Mildly to moderately
decreased
Moderately to
severely decreased
<30 mg/g
<3 mg/mmol
30–300 mg/g
3–30 mg/mmol
>300 mg/g
>30 mg/mmol
Referral decision making by GFR and albuminuria. *Referring clinicians may wish to discuss with their nephrology service depending on
local arrangements regarding monitoring or referring.
14 Kidney International Supplements (2013) 3,5–14
summary of recommendation statements

Introduction: The case for updating and context
Kidney International Supplements (2013) 3, 15–18; doi:10.1038/kisup.2012.63
Justification for updating the guideline for CKD Definition,
Evaluation, Classification, and Stratification from an
international perspective
In 2002, the US-based Kidney Disease Outcomes Quality
Initiative (KDOQI) group published a guide line on defini-
tion, classification and evaluatio n of chronic kidney disease
(CKD). The guideline proposed uniform definitions of CKD
together with a staging system
1
and described issues related
to measurement of kidney func tion that had not previously
been identified by the clinical community. This publication
revolutionized the concept and management of CKD,
generating substanti al research and controversy, stimulating
discussion, and influencing public policy and laboratory
practice. The research generated has led to new insig hts
which require contextualizing in the current era, providing
the evidence drivers for updating guidance for defining,
diagnosing, staging and managing CKD, and promoting
improved care of those with early CKD. Successive interna-
tional controversies conferences, under the direction of
Kidney Disease: Improving Global Outcomes (KDIGO), have
shaped the scope of this international update through:
1. Facilitating global implementation of the definition and
classification of CKD , identifying areas of uncertainty , and
developing a collaborative research agenda to impro ve the
evidence base and facilitate implementation (Nove mber 2004).
2. Evaluating the definition and classification of CKD from a
global public health perspective (October 2006).
3. Reviewing the definition and classification of CKD
based on data on patient prognosis derived from a
unique research collaboration on prognosis (October
2009).
Given the international interest in understanding and
improving the outcomes of people living with kidney disease
and the tremendous amount of data generated since 2002, a
need was identified to review, revise, and update the original
2002 KDOQI guideline.
There has been a wealth of published data highlighting
the risk of adverse consequences and outcomes in people
with albumin excretion rate (AER) 430 mg/24 hours and/or
glomerular filtration rate (GFR) o60 ml/min/1.73 m
2
(GFR
categories G3a-G5), irrespective of the etiology or duration of
reduced kidney function. Description of the relationship
between GFR, albuminuria and prognosis has significantly
improved the understanding of CKD in multiple popula-
tions.
2–5
Internationally, the widespread use of albumin-to-
creatinine ratio (ACR) and reagent strip urine testing to
detect elevated albuminuria together with reporting of
estimated GFR (eGFR) has led to easier identification of
people with CKD. However elevated albuminuria or reduced
GFR alone are not necessarily indicators of need for specialist
referral. Clinicians and medical systems are still adjusting to
the improved ‘‘identification’’ of CKD and guidance about
appropriate stratification of risk and modified action plans
for different subgroups of individuals regarding further
evaluation, referral, or treatment is needed.
The goal of this guideline is to clarify the definition and
classification system of CKD, and to develop appropriate
guidance as to the management and care of people with CKD.
In addition, we present a framework which should foster an
extended collaborative research agenda over the next decade
and inform guidelines in the future.
Kidney disease is an important problem worldwide
Kidney disease is defined as an abnormality of kidney
structure or function with implications for the health of an
individual, which can occur abruptly, and either resolve or
become chronic. CKD is a general term for heterogeneous
disorders affecting kidney structure and function with
variable clinical presentation, in part related to cause, severity
and the rate of progression. The concept of CKD evolved
after the recognition of the contribution of disordered kidney
structure and function on the health of individuals across a
wide range of severity.
1
The utility of the concept is that
recognition of CKD will have implications for the individual
and their care. Kidney failure is traditionally considered as
the most serious outcome of CKD. Symptoms are usually due
to complications of decreased kidney function and when
severe, they can be treated only by dialysis or transplantation.
Earlier stages of kidney disease are often asymptomatic, are
detected during the evaluation of comorbid conditions, and
may be reversible. Rapidly progressive diseases may lead to
kidney failure within months but most diseases evolve over
decades, and so me patients do not progress during many
years of follow-up.
Figure 1 shows a conceptual model for the development,
progression, and complications of CKD.
1,6
The model
includes antecedents asso ciated with increased risk for
development of CKD, stages of disease, and complications
including death. Risks for development of CKD may be
categorized either as susceptibility to kidney disease due to
sociodemographic and genetic factors or exposure to factors
that can initiate kidney disease. Abnormalities in kidney
structure (damage) usually precede abnormalities in func-
tion. Outcomes of CKD may be progression, as shown by the
horizontal arrows, and complications, as shown by the
diagonal arrows, or both.
http://www.kidney-international.org introduction
& 2013 KDIGO
Kidney International Supplements (2013) 3, 15–18 15
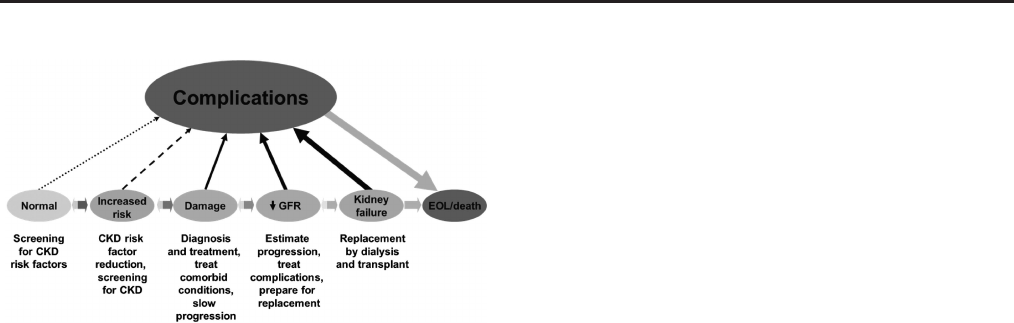
Although the need for treatment of chronic kidney failure
with dialysis and/or kidney transplantation arises in only
1% of people with CKD, it remains the most expensive of
chronic diseases and reduces lifespan significan tly. The costs
of dialysis and transplantation consume disproportionate
amounts within the health-care budgets in all jurisdictions
(5% of annual budgets consumed by less than 1% of the
population). Failure to recognize CKD results in neglect of its
consequences and complications, and late referral of people
with advanced CKD resulting in worse renal replacement
therapy (RRT) outcomes. In addition, there is a growing
body of evidence that indicates people with CKD are at
increased risk of acute kidney injury (AKI), which is also
associated with poor outcomes and may accelerate progres-
sion of CKD. Therefore, identification of people at earlier
time points in the trajectory of CKD, with appropriate
management and earlier referral of those who would benefit
from specialist kidney services, should lead to both economic
and clinical benefits.
In those countries where access to dialysis and transplan-
tation services may be limited or unavailable, the final
consequence of progressive CKD is death. In all locations,
irrespective of availability of dialysis and transplantation,
early identification of CKD therefore assumes great im-
portance, as delay or prevention of progression has the
potential to prolong health and save lives for much lower cost
than RRT. Although etiologies vary in frequency or absolute
numbers in different countries, the proportion of people with
important antecedents to CKD, such as diabetes, is growing
alarmingly worldwide in both developed and developing
countries.
Complications of CKD affect all organ systems. Kidney
failure leads to the commonly recognized symptoms of
uremia. Less severe CK D has been recognized as an
independent risk factor for cardiovascular disease (CVD)
and other common conditions affecting the elderly, such as
infection and impairments in physical function and cogni-
tion. In addition, CKD is associated with increased risk from
adverse effects of drugs, intravascular radiocontrast admin-
istration, surgery and other invasive procedures. Altogether,
these complications are associated with higher morbidity,
mortality and cost. If CKD is detected early, the associated
complications and the progression to kidney failure can be
delayed or even prevented through appropriate interventions.
Regular testing of high-risk groups (i.e., people with diabetes,
hypertension, CVD, structural renal tract disease, multi-
system diseases with potential kidney involvement such as
systemic lupus er ythematosus, family history of kidney
failure, hereditary kidney disease, the elderly, those receiving
potential nephrotoxic drugs or those opportunistically found
to have hematuria or proteinuria) can give an early
indication of kidney damage, thus permitting the introduc-
tion of available interventions at an early stage, and the
testing of novel interventions with potential added value.
Factors associated with progression of CKD and with
increased cardiovascular risk are overlapping to a large
extent. Thus targeting of those risk factors that are modifiable
may both reduce CVD in people with CKD and reduce
progression of CKD to end-stage renal disease (ESRD). There
is strong evidence that blockade of the renin-angiotensin-
aldosterone system (RAAS) is a blood pressure (BP) lowering
strategy which is more effective in reducing risk of kidney
and cardiovascular disease in the presence of albuminuria.
The development of guidance for health-care providers will
provide opportunities to improve the care of people w ith kidney
disease. We hope that this publication serves to stimulate
strategic research initiatives from basic, translational, clinical
and health outcome perspectives.
General summary for the reader: what you will and
will not find in this guideline
1. The guideline will offer best practice and evidence-based
advice on the evaluation and approach to management
of CKD.
a. The target population for the guideline is all people
identified with CKD who are not on RRT (i.e., not on
dialysis or have not received a kidney transplant).
b. The target population includes adults and children.
The guideline w ill cover the spectrum of individuals
with CKD, from children to the elderly who form
important subgroups, underscoring current issues at
the extremes of age with respect to the evidence base,
especially in relation to implementation and manage-
ment issues. Where the guideline does not apply to
Figure 1 | Conceptual model of CKD. Continuum of
development, progression, and complications of CKD and
strategies to improve outcomes. Horizontal arrows between
circles represent development, progression, and remission of CKD.
Left-pointing horizontal arrowheads signify that remission is less
frequent than progression. Diagonal arrows represent occurrence
of complications of CKD, including drug toxicity, endocrine and
metabolic complications, cardiovascular disease, and others such
as infection, cognitive impairment, and frailty. Complications
might also arise from adverse effects of interventions to prevent
or treat the disease. CKD, chronic kidney disease; EOL, end-of-life
care and/or conservative management; GFR, glomerular filtration
rate. Adapted from Levey AS, Stevens LA, Coresh J.
6
Conceptual
model of CKD: applications and implications.
Am J Kidney Dis. 2009; 53:S4-16 with permission from the
National Kidney Foundation; accessed http://download.journals.
elsevierhealth.com/pdfs/journals/0272-6386/PIIS02726386080
17186.pdf
16 Kidney International Supplements (2013) 3, 15–18
introduction

children, statements to that effect will be made. It is
beyond the scope of this guideline to address all issues
related to children with CKD, given the heteroge-
neous nature of this group of individuals who range
from newborn to post-adolescents, with specific
physiological differences w ithin each of those groups.
Specific evidence and rationale will be articulated as
appropriate in each section.
c. The target condition is CKD of any or unknown
etiology. Identifying the cause of the CKD is strongly
encouraged, both because treatment may need to be
adjusted according to etiology and because it
influences the prognosis and relative importance of
risk factors associated with CKD. A comprehensive
list of possible etiologies is not practical and guidance
on detailed work-up for specific causes of CKD is
beyond the scope of this document (readers will be
referred to other pertinent sources). We will describe
how knowledge of the etiolo gy of CKD in an
individual may be important in prognostication and
management.
d. The target audience of the guideline includes
nephrologists, primary care physicians, non-nephro-
logy specialists (e.g., cardiologists, diabetologists, etc),
clinical chemists and other practitioners caring for
adults and children with CKD. The guideline is also
expected to be suitable for use in public policy and
other health-care arenas.
e. As a global guideline it is written for use in different
health-care settings, but unavoidably its full imple-
mentation relies on health-care resources that
are not universally available. We recognize this overtly
in some of the discussion sections within the
guideline.
f. The target health-care settings include primary,
secondary, and ter tiary care.
2. The guideline will provide information, advice, and
education to support self-management for people with
CKD and aid caregivers with the diagnosis and manage-
ment of CKD. To avoid redundancy and potential for
becoming outdated, the reader is asked to refer to existing
KDIGO guidance on anemia, metabolic bone disease, BP,
AKI, hepatitis C, lipid management, glomerulonephritis
(GN) and other pertinent guidelines.
3. The guideline will provide a blueprint for an approach to
CKD care in an international context. While the guideline
will be sensitive to issues related to ethnicity and also
geographical considerations, it is expected that subsequent
regional adaptation will be required for specific health-
care settings or contexts.
4. Research recommendations in general are described to
inform a framework for ongoing research agendas in the
international community. We have attempted to identify
important study questions in need of answers. Through
identification of gaps in knowledge, the reader will be
better able to define methodologies, definitions of
populations, and outcome measures of relevance to study
designs in the future.
Topics that will not be covered
This document is not intended to provide enough detail to
replace training and education in nephrology, nor is it
intended to serve as a textbook of medicine or nephrology.
Thus, there are some specific topics that will not be
covered. Specifically we will not discuss:
1. Evaluation and management of people receiving RRT
(management of kidney failure by dialys is or kidney
transplantation).
2. Specific approaches to the diagnosis of people with AKI
and other acute kidney diseases. This topic has been
extensively reviewed in KDIGO Clinical Practice Guideline
for Acute Kidney Injury.
7
3. Diagnostic work-up or treatment of specific causes of
CKD, including GN.
8
4. Management of pregnancy in women with CKD or of
pregnant women who develop kidney disease.
5. Detailed management of endocrine and metabolic com-
plications of CKD. Thes e are reviewed extensively by
recent KDIGO guidelines on CKD-MBD,
9
management of
BP
10
and anemia
11
in CKD.
6. Detailed management of CVD and CVD risk factors in
CKD. This is reviewed in a recent KDIGO publication.
12
7. Drug dosing in CKD. This topic has be en addressed in a
recent KDIGO publication.
13
8. Details of resource implications and barriers to imple-
mentation are beyond the scope of this guideline.
By virtue of its being international, the variability in
these aspects by country, region, and even jurisdiction is
vast. We look to the individual commentaries from
around the world to inform those aspects more fully.
Brief overview on methodology
The Work Group included an international group of kidney
specialists, primary care physicians, diabetologists, epide-
miologists, clinical chemists, administrato rs, and a profes-
sional Evidence Review Team (ERT) who provided support
and guidance to the group. Details of the methods used by
the ERT are described in Methods for Guideline Development,
along with the systematic searches for areas identified by
Work Group members and performed by the ERT.
The recommendations and statem ents created herein
will serve to direct both care and research in the next
decade. Importantly, we expect the renewed classification
system and risk stratification concepts to direct research and
enrollment into trials which address test therapies to improve
patient outcomes.
Statement grading and wording. The methods for formu-
lating recommendations were based on modified Grading
of Recommendations Assessment, Development, and
Evaluation (GRADE) system, and have used the words
Kidney International Supplements (2013) 3, 15–18 17
introduction

‘recommend’ when the balance of evidence would support
the notion that the majority of patients would benefit from
the implementation of this recommendation. The words
‘suggest’ are used when the balance of evidence would
support the notion that some patients would benefit from the
implementation of the recommendation, but that individual
patient, physician and health-care system considerations
would be necessary to adopt the practice. There are
also ungraded statements many of which are often key
practice points or educational issues (Table 1). The Work
Group had struggled whether to organi ze them differently or
move them to the rationale section. Ultimately they remain
here in the guideline statement format so that they are not
overlooked by those wishing to unde rstand the condition
better.
A significant proportion of statements in this guideline are
ungraded because the grading system is most appropriate
for statements of intervention. The international system,
GRADE, allows for such statements which guide thoughts
and attitude, and not specific actions. In the descriptive
statements identifying, classifying, and defining the condition
of CKD, grading is not possible. Since few studies have
compared different methods of evaluation or care models,
those statements too are difficult to grade. Thus, grading of
specific statements is reserved for interventions or alternative
diagnostic test strategies for which there is a substantial body
of evidence.
Consideration of health benefits, side effects, and risks.
These have been considered when for mulating the recom-
mendations but given the paucity of data in many of the areas
reviewed, this has been less consistent than the Work Group
would have liked. We see this as an area of research and
future study that will inform future updates.
Review process. As with all KDIGO guidelines a two step
process was used. This included a review by the Board of
Directors, with feedback to the Work Group Chairs followed
by rev isions to the document. The public review, consisting
of inte rested stakeholders from international communities,
organizations and individ uals, was then undertaken. The
draft document was sent to a total of 2320 exter nal reviewers,
with 293 responses received and tabulated. The feedback was
carefully rev iewed and where appropriate, suggested changes
were incorporated into the final document. In the interest of
transparency, the Work Group prepared individual responses
to each reviewer comment and these will be posted on the
KDIGO website.
Planned update. At the current time there is no official
plan to update the guideline in its entirety. Given the breadth
and depth of the current undertaking and with knowledge of
new studies and applications of some of these recommenda-
tions, the Work Group recommends that individual sections
of this guideline be updated every 3-5 years as new evidence
becomes available. We believe that this will be more practical
for the readership.
Table 1 | KDIGO nomenclature and description for grading recommendations
Implications
Grade* Patients Clinicians Policy
Level 1
‘‘We
recommend’’
Most people in your situation would
want the recommended course of
action and only a small proportion
would not.
Most patients should receive the
recommended course of action.
The recommendation can be evaluated
as a candidate for developing a policy
or a performance measure.
Level 2
‘‘We suggest’’
The majority of people in your situation
would want the recommended course
of action, but many would not.
Different choices will be appropriate
for different patients. Each patient
needs help to arrive at a management
decision consistent with her or his
values and preferences.
The recommendation is likely to
require substantial debate and
involvement of stakeholders before
policy can be determined.
*The additional category ‘‘Not Graded’’ was used, typically, to provide guidance based on common sense or where the topic does not allow adequate application of evidence.
The most common examples include recommendations regarding monitoring intervals, counseling, and referral to other clinical specialists. The ungraded recommendations
are generally written as simple declarative statements, but are not meant to be interpreted as being stronger recommendations than Level 1 or 2 recommendations.
18 Kidney International Supplements (2013) 3, 15–18
introduction

Chapter 1: Definition and classification of CKD
Kidney International Supplements (2013) 3, 19–62; doi:10.1038/kisup.2012.64
1.1: DEFINITION OF CKD
1.1.1: CKD is defined as abnormalities of kidney struc-
ture or function, present for 43 months, with
implications for health (Table 2). (Not Graded)
RATIONALE
The definition of CKD remains intact, but we have clarified
the classification and risk stratification as indicated below.
The addition of ‘with implications for health’ is intended to
reflect the notion that a variety of abnormaliti es of kidney
structure or function may exist, but not all have implications
for health of individuals, and therefore need to be
contextualized.
Kidney damage refers to a broad range of abnormalities
observed during clinical assessment, which may be insensitive
and non-specific for the cause of disease but may precede
reduction in kidney function (Table 2). Excretory, endocrine
and metabolic functions decline together in most chronic
kidney diseases. GFR is generally accepted as the best overall
index of kidney function. We refer to a GFR o60 ml/min/
1.73 m
2
as decreased GFR (Table 2) and a GFR o15 ml/min/
1.73 m
2
as kidney failure. AKI may occur in patients with
CKD and hasten the progression to kidney failure.
14
Complications include drug toxicity, metabolic and
endocrine complications, increased risk for CVD, and a
variety of other recently recognized complications, including
infections, frailty, and cognitive impairment.
15–18
Complica-
tions may occur at any stage, often leading to death without
progression to kidney failure. Complications may also arise
from adverse effects of in terventions to prevent or treat the
disease and associated comorbidity.
Criteria for CKD
Defining terms: The following section aims to define specific
terms and concepts so as to ensure clarity among all users.
In addition, the rationale for including these terms is included.
Table 3 provides a justification for the criteria for CKD. The
criteria for definition of CKD are objective and can be
ascertained by means of simple laboratory tests without
identification of the cause of disease, thereby enabling
detection of CKD by non-nephrologist physicians and other
health professionals.
Duration 43 Months
Kidney diseases may be acute or chronic. We explicitly but
arbitrarily define duration of 43 months (490 days) as
delineating ‘‘chronic’’ kidney disease. The rationale for
defining chronicity is to differentiate CKD from acute kidney
diseases (such as acute GN), including AKI, which may
require different interventions, and have different etiologies
and outcomes.
7
We did not define acute kidney disease
(AKD) because there does not appear be an evidence base for
a precise definition.
The duration of kidney disease may be documented or
inferred based on the clinical context. For example, a patient
with decreased kidney function or kidney damage in the
midst of an acute illness, w ithout prior documentation of
kidney di sease, may be inferred to have AKI. Resolution over
days to weeks would confirm the diagnosis of AKI. A patient
with similar findings in the absence of an acute illness may be
inferred to have CKD, and if followed over time would be
confirmed to have CKD. In both cases, repeat ascertainment
of kidney function and kidney damage is recommended for
accurate diagnosis. The timing of the evaluation depends on
clinical judgment, with earlier evaluation for the patients
suspected of having AKI and later evaluation for the patient
suspected of having CKD. For further details on the
Evaluation of CKD, see Chapter 1.4.
Reversibility. Most kidney diseases do not have symptoms
or findings until later in their course and are detected only
when they are chronic. Most causes of CKD are irreversible
with a life-long course, and treatment aimed at slowing
progression to kidney failure. However, chronicity is not
synonymous with irreversibility. In some cases, CKD is
entirely reversible, either spontaneously or with treatment,
and in other cases, treatment can cause partial regression of
kidney damage and improvement in function (e.g., immuno-
suppressive therapies for GN). Even kidney failure may be
reversed with transplantation. Because of the long course of
most cases of CKD, patients often have one or more episodes
of AKI, superimposed upon CKD.
Decreased GFR
The kidney has many functions, including excretory,
endocrine and metabolic functions. The GFR is one compo-
nent of excretory function, but is widely accepted as the best
overall index of kidney function because it is generally
reduced after widespread structural damage and most other
kidney functions decline in parallel with GFR in CKD.
We chose a threshold of GFR o60 ml/min/1.73 m
2
(GFR
categories G3a-G5) for 43 months to indicate CKD. A GFR
o60 ml/min/1.73 m
2
is less than half of the normal value in
young adult men and women of approximately 125 ml/min/
1.73 m
2
. Figure 2 shows a compilation of GFR measurements
in apparently healthy men and women in the US and Europe
by age from more than 40 years ago.
20
The age-associated
GFR decline is observed in longitudinal as well as cross
http://www.kidney-international.org chapter 1
& 2013 KDIGO
Kidney International Supplements (2013) 3, 19–62 19

Table 2 | Criteria for CKD (either of the following present for 43 months)
Markers of kidney damage (one or more) Albuminuria (AERZ30 mg/24 hours; ACRZ30 mg/g [Z3 mg/mmol])
Urine sediment abnormalities
Electrolyte and other abnormalities due to tubular disorders
Abnormalities detected by histology
Structural abnormalities detected by imaging
History of kidney transplantation
Decreased GFR GFRo60 ml/min/1.73 m
2
(GFR categories G3a-G5)
Abbreviations: CKD, chronic kidney disease; GFR, glomerular filtration rate.
Table 3 | Criteria for definition of CKD
19
Criteria Comment
Duration 43 months, based on documentation or
inference
Duration is necessary to distinguish chronic from acute kidney diseases
K Clinical evaluation will often enable documentation or inference of duration
K Documentation of duration is usually not declared in epidemiologic studies
GFR o60 ml/min/1.73 m
2
(GFR categories G3a-G5) GFR is the best overall index of kidney function in health and disease
K The normal GFR in young adults is approximately 125 ml/min/1.73 m
2
. GFR
o15 ml/min/1.73 m
2
(GFR category G5) is defined as kidney failure
K Decreased GFR can be detected by current estimating equations for GFR based
on SCr or cystatin C but not by SCr or cystatin C alone
K Decreased eGFR can be confirmed by measured GFR, if required
Kidney damage as defined by structural abnormalities or
functional abnormalities other than decreased GFR
Albuminuria as a marker of kidney damage [increased glomerular permeability],
urine AER Z30 mg/24 hours, approximately equivalent to urine ACRZ30 mg/g
(Z3 mg/mmol)*
K The normal urine ACR in young adults is o10 mg/g (o1 mg/mmol)
K Urine ACR 30-300 mg/g (3–30 mg/mmol; category A2) generally corresponds to
‘‘microalbuminuria,’’ now referred to as ‘‘moderately increased’’
K Urine ACR 4300 mg/g (430 mg/mmol; category A3) generally corresponds to
‘‘macroalbuminuria,’’ now termed ‘‘severely increased’’
K Urine ACR42200 mg/g (220 mg/mmol) may be accompanied by signs and
symptoms of nephrotic syndrome (e.g., low serum albumin, edema, and high
serum cholesterol)
K Threshold value corresponds approximately to urine reagent strip values of trace
or +, depending on urine concentration.
K High urine ACR can be confirmed by urine albumin excretion in a timed urine
collection expressed as AER
Urinary sediment abnormalities as markers of kidney damage
K Isolated non-visible (microscopic) hematuria with abnormal RBC morphology
(anisocytosis) in GBM disorders
K RBC casts in proliferative glomerulonephritis
K WBC casts in pyelonephritis or interstitial nephritis
K Oval fat bodies or fatty casts in diseases with proteinuria
K Granular casts and renal tubular epithelial cells in many parenchymal diseases
(non-specific)
Renal tubular disorders
K Renal tubular acidosis
K Nephrogenic diabetes insipidus
K Renal potassium wasting
K Renal magnesium wasting
K Fanconi syndrome
K Non-albumin proteinuria
K Cystinuria
Pathologic abnormalities detected by histology or inferred (examples of causes)
K Glomerular diseases (diabetes, autoimmune diseases, systemic infections, drugs,
neoplasia)
K Vascular diseases (atherosclerosis, hypertension, ischemia, vasculitis, thrombotic
microangiopathy)
K Tubulointerstitial diseases (urinary tract infections, stones, obstruction, drug
toxicity)
K Cystic and congenital diseases
20
Kidney International Supplements (2013) 3, 19–62
chapter 1

sectional studies, but varies substantially among individuals
within the population.
21
More recent data in kidney donors
confirm these general trends.
22,23
Limited data are available
for non-whites in the US and Europe or in other countries,
although data suggest that the normal range for measured
GFR and the age-associated decline is similar.
24–26
A GFR o60 ml/min/1.73 m
2
can be detected by routine
laboratory testing. Current estimating equations for GFR
(eGFR) based on serum creatinine (SCr), but not SCr alone,
are sensitive for detecting measured GFRo60 ml/min/
1.73 m
2
.
27
A decreased eGFR using SCr can be confirmed
by GFR estimation using an alternative filtration marker
(cystatin C) or GFR measurement, as necessary.
A GFR o60 ml/min/1.73 m
2
is associated with a higher
risk of complications of CKD than in subjects with CKD and
conserved GFR. The causal mechanisms underlying these
associations are not fully understood. We consider three main
types of complications, which are of relevance to all patients
with CKD and reduced GFR, irrespective of country, age or
etiology:
Drug toxicity. Altered pharmacokinetics of drugs excreted
by the kidney and an increased risk of drug-interactions are
common and require adjustment in the dosage of many
drugs (see Chapter 4.4).
13
At lower GFR, altered pharmaco-
kinetics and pharmacodynamics of drugs not excreted by the
kidney may also be observed. Errors in drug dosing are
common in patients with CKD and may be associated with
toxicity to the kidney (resulting in AKI) or systemic toxicity,
resulting in threats to patient safety.
Metabolic and endocrine complications. As GFR declines a
variety of complications reflecting loss of endocrine or
exocrine function of the kidneys develop including anemia,
acidosis, malnutrition, bone and mineral disorders (des-
cribed in Chapters 3 and 4).
Risk of CVD and death. A meta-analysis by the CKD Prog-
nosis Consortium demonstrated associations of eGFR o60 ml/
min/1.73 m
2
with subsequent risk of all-cause and cardio-
vascular mortality , kidney failure, AKI, and CKD progr ession in
the general population and in populations with increased risk
for CVD .
3-5
Figure 3 shows the relationship for total and
cardiov ascular mortality in general population cohorts. The
risk for all outcomes was relatively constant between eGFR
of 75-105 ml/min/1.73 m
2
, with a suggestion of a U-shaped
curve for total mortality . The increased relative risk (RR) for
all outcomes was significant for eGFR of o60 ml/min/1.73 m
2
.
Kidney Damage
Damage to the kidney can be within the parenchyma,
large blood vessels or collecting systems, and is most often
inferred from markers rather than direct exami nation of
kidney tissue. The markers of kidney damage often provide
a clue to the likely site of damage within the kidney and in
association with other clinical findings, the cause of kid ney
disease.
Proteinuria. Proteinuria is a general term for the presence
of increased amounts of protein in the urine. Proteinuria may
reflect abnormal loss of plasma proteins due to a) increased
glomerular permeability to lar ge molecular weight proteins
(albuminuria or glomerular proteinuria), b) incomplete
tubular reabsorption of normally filtered low-molecular-
weight proteins (tubular proteinuria), or c) increased plasma
concentration of low-molecular-weight proteins (overpro-
duction proteinuria, such as immunoglobuli n light chains).
Proteinuria may also reflect abnorm al loss of proteins derived
from the kid ney (renal tubular cell constituents due to
tubular damage) and lower urinary tract. Albuminuria,
tubular proteinuria and renal tubular cell constituents
are pathognomonic of kidney damage. In addition, findings
Table 3 | Continued
Criteria Comment
Structural abnormalities as markers of kidney damage detected by imaging
(ultrasound, computed tomography and magnetic resonance with or without contrast,
isotope scans, angiography)
K Polycystic kidneys
K Dysplastic kidneys
K Hydronephrosis due to obstruction
K Cortical scarring due to infarcts, pyelonephritis or associated with vesicoureteral
reflux
K Renal masses or enlarged kidneys due to infiltrative diseases
K Renal artery stenosis
K Small and hyperechoic kidneys (common in more severe CKD due to many
parenchymal diseases)
History of kidney transplantation
K Kidney biopsies in most kidney transplant recipients have histopathologic
abnormalities even if GFR is 460 ml/min/1.73 m
2
(GFR categories G1-G2) and
ACR is o30 mg/g (o3 mg/mmol)
K Kidney transplant recipients have an increased risk for mortality and kidney
failure compared to populations without kidney disease
K Kidney transplant recipients routinely receive subspecialty care
Abbreviations: ACR, albumin-to-creatinine ratio; AER, albumin excretion rate; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; GBM, glomerular
basement membrane; GFR, glomerular filtration rate; RBC, red blood cell; SCr, serum creatinine; WBC, white blood cell.
*For conversion, see Table 7, Chapter 1
Kidney International Supplements (2013) 3, 19–62
21
chapter 1

from experimental and clinical studies have suggested an
important role for proteinuria in the pathogenesis of disease
progression of CKD.
28
Albuminuria. Albuminuria refers to abnormal loss of
albumin in the urine. Albumin is one type of plasma protein
found in the urine in normal subjects and in larger quantity
in patients with kidney disease.
For a number of reasons, clinical terminology is changing
to focus on albuminuria rather than proteinuria: a) albumin
is the principal compone nt of urinary protein in most kidney
diseases; recent recommendations for measurement of urine
proteins emphasize quantification of albuminuria rather than
total protein; b) recent epidemiologic data from studies
around the world demonstrate a strong graded relationship
of the quantity of urine albumin with both kidney and CVD
risk; and c) later recommendations in these guidelines classify
kidney disease by level of albuminuria. In this guideline, we
will refer to proteinuria when discussing general concepts
and will refer either to total protein, albumin or other specific
proteins when discussing measurements, patterns, and
interpretation of proteinuria.
Albuminuria is a common but not uniform find ing in
CKD. It is the earliest marker of glomerular diseases,
including diabetic glomerulosclerosis, where it generally
appears before the reduction in GFR. It is a marker of
hypertensive nephrosclerosis but may not appear until after
the reduction in GFR. It is often associated with underlying
hypertension, obesity, and vascular disease, where the
underlying renal pathology is not known.
Normative values for albuminuria and proteinuria are
generally expressed as the urinary loss rate. The urina ry loss
rate of albumin and protein has commonly been referred to
as AER and protein excretion rate (PER), respectively,
although in the strict physiological sense they are not
excreted. The terms AER and PER will be retained herein.
We chose a threshold for urinary AER of Z30 mg/
24 hours sustained for 43 months to indicate CKD.
This value is considered to be approximately eq uivalent
to an ACR in a random untimed urine sample of Z30 mg/g
or Z3 mg/mmol. The rationale for this threshold is as
follows:
K An AER of Z30 mg/24 hours (ACRZ30 mg/g [Z3 mg/
mmol]) is greater than 3 times the normal value in young
adult men and women of approximately 10 mg/24 hours
(ACR 10 mg/g or 1 mg/mmol).
K An AER of Z30 mg/24 hours (A CRZ30 mg/g [Z3mg/
mmol]) may sometimes be detectable as ‘trace ’ using a urine
reagent strip, depending on urine concentration, but this is
not a consistent finding until AER exceeds approximately
300 mg/24 hours (A CRZ300 mg/g [Z30 mg/mmol]). As
described later , trace or positive reagent strip values/readings
can be confirmed by A CR, and an elevated ACR can be
confirmed by urine AER in a timed urine collection, as
necessary .
K An AERZ30 mg/24 hours (ACRZ30 mg/g [Z3 mg/
mmol]) is associated with an increased risk for
complications of CKD. A meta-analysis by the CKD
Prognosis Consortium demonstrated associations of an
ACRZ30 mg/g (Z3 mg/mmol) or reagent strip 1 þ
protein with subsequent risk of all-cause and cardio-
vascular mortality, kidney failure, AKI, and CKD
progression in the general population and in populations
with increased risk for CVD
3-5
(Figure 4).
Figure 2 | Normal values for GFR by age. GFR is shown for men (Panel a) and women (Panel b) of various ages, with the GFR measured as
the urinary clearance of inulin. The horizontal line indicates a GFR value of 60 ml/min/1.73 m
2
, which is the threshold for the definition of
CKD. Solid lines represent the mean value of GFR per decade of age, and dashed lines represent the value 1 SD from the mean value of GFR
per decade of age. CKD, chronic kidney disease; GFR, glomerular filtration rate; SD, standard deviation. Adapted with permission from
Wesson L.
20
Physiology of the Human Kidney. Grune & Stratton: New York, 1969.
22 Kidney International Supplements (2013) 3, 19–62
chapter 1
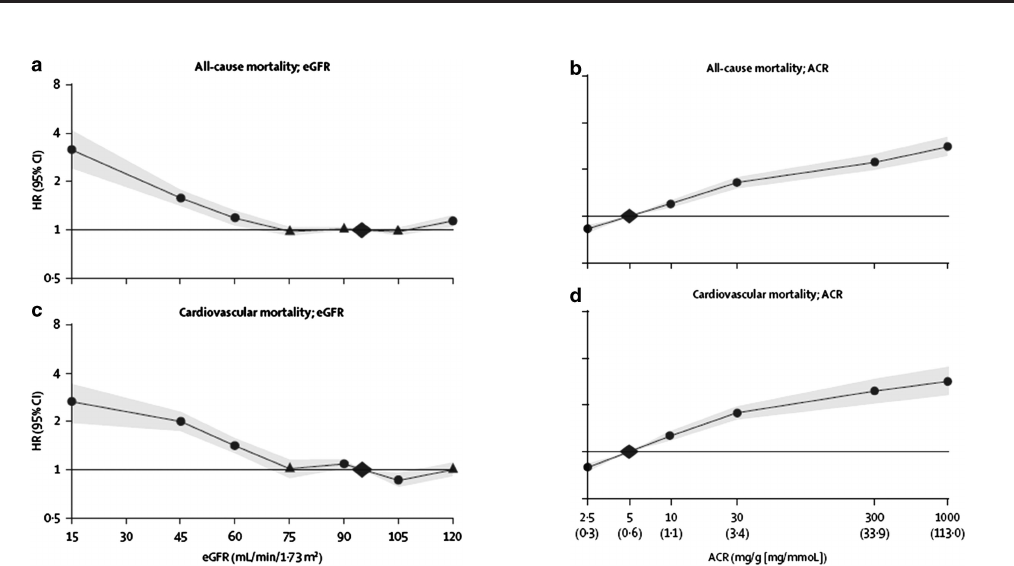
Urine sediment abnormalities. Formed elements, suc h as
cells, casts, crystals, and microorganisms may appear in the
urine sediment in a variety of disorders of the kidney and
urinary tract, but renal tubular cells, red blood cell (RBC)
casts, white blood cell (WBC) casts, coarse granular casts,
wide casts, and large numbers of dysmorphic RBCs are
pathognomonic of kidney damage.
Electrolyte and other abnormalities due to tubular disorders.
Abnormalities of electrolytes and other solutes may result from
disorders of renal tubular reabsorption and secretion. These
syndromes are uncommon but pathognomonic of kidney
disease. Often the diseases are genetic without underlying
pathologic abnormalities. Other diseases are acquired, due to
drugs or toxins, and are usually with prominent tubular
pathologic lesions.
Pathologic abnormalities directly observed in kidney tissue
obtained by biopsy.
Evidence of abnormalities of renal
parenchyma in kidney biopsies irrespective of eGFR or other
markers of kidney damage must be acknowledged as an
important parameter in defining kidney damage. The
pathologic classification of diseases of the renal parenchyma
reflects the localization of the disease to glomeruli, vessels,
tubules and interstitium, or cysts. Renal biopsies are
performed in the minority of CKD patients.
Imaging abnormalities. Imaging techniques allow the
diagnosis of diseases of the renal structure, vessels and/or
collecting systems. Thus, patients with significant structural
abnormalities are considered to have CKD if the abnor-
mality persists for greater than 3 months (note that this does
not include simple cysts and clinical context is required
for action).
History of kidney transplantation. Kidney transplant re-
cipients are defined as having CKD, irrespective of the level of
GFR or presence of markers of kidney damage. The rationale
Figure 3 | Relationship of eGFR with mortality. HRs and 95% CIs
for all-cause (a) and cardiovascular mortality (c) according to
spline eGFR. HRs and 95% CIs (shaded areas) are adjusted for ACR,
age, sex, ethnic origin, history of CVD, systolic BP, diabetes,
smoking, and total cholesterol. The reference (diamond) was eGFR
95 ml/min/1.73 m
2
and ACR 5 mg/g (0.6 mg/mmol), respectively.
Circles represent statistically significant and triangles represent
not significant. ACR, albumin-to-creatinine ratio; BP, blood
pressure; CI, confidence interval; CVD, cardiovascular disease;
eGFR, estimated glomerular filtration rate; HR, hazard ratio.
Reprinted from The Lancet, vol 375, Matshushita K, van de Velde
M, Astor BC, et al.
4
Association of estimated glomerular filtration
rate and albuminuria with all-cause and cardiovascular mortality
in general population cohorts: a collaborative meta-analysis, p.
2073-2081, 2010, with permission from Elsevier; accessed http://
download.thelancet.com/pdfs/journals/lancet/
PIIS0140673610606745.pdf
Figure 4 | Relationship of albuminuria with mortality. HRs and
95% CIs for all-cause (b) and cardiovascular mortality (d)
according to ACR. HRs and 95% CIs (shaded areas) are adjusted for
age, sex, ethnic origin, history of CVD, systolic BP, diabetes,
smoking, and total cholesterol and spline eGFR. The reference
(diamond) was ACR 5 mg/g (0.6 mg/mmol) and eGFR 95 ml/min/
1.73 m
2
, respectively. Circles represent statistically significant and
triangles represent not significant. ACR plotted in mg/g. To
convert ACR in mg/g to mg/mmol multiply by 0.113. Approximate
conversions to mg/mmol are shown in parentheses. ACR,
albumin-to-creatinine ratio; BP, blood pressure; CI, confidence
interval; CVD, cardiovascular disease; eGFR, estimated glomerular
filtration rate; HR, hazard ratio. Reprinted from The Lancet, vol
375, Matshushita K, van de Velde M, Astor BC, et al.
4
Association of
estimated glomerular filtration rate and albuminuria with all-cause
and cardiovascular mortality in general population cohorts: a
collaborative meta-analysis, p. 2073-2081, 2010, with permission
from Elsevier; accessed http://download.thelancet.com/pdfs/
journals/lancet/PIIS0140673610606745.pdf
Kidney International Supplements (2013) 3, 19–62 23
chapter 1

for this designation is that biopsies in kidney trans-
plant recipients reveal pathologic abnormalities even in
patients without decreased GFR or albuminuria. Kidney
transplant recipients have an increased risk of mortality and
kidney outcomes compared to the general population and
they require specialized medical management.
29
Implications for Health
CKD is associated with a wide range of complications leadi ng
to adverse health outcomes. For some complications, the
causal pathway between kidney disease and adverse outcomes
is well-known. For these complications, there are clinical
practice guidelines for testing and treatment for modifiable
factors to prevent adverse outcomes. Since 2002 a large
number of epidemiologic studies have linked decreased GFR
and albuminuria to the risk of adverse health outcomes not
previously identified as CKD complications. The exploration
of the mechanisms for the relationships of CKD with
these complications is a rapidly growing topic for basic
and clinical research. Be cause of the high prevalence,
adverse outcomes, and high cost of CKD, especi ally kidney
failure, some countries have developed public health
programs for early identification and treatment of CKD
and its complications. The effectiveness of these programs is
being evaluated.
Implications for Clinical Practice and Public Policy
CKD was first defined in the 2002 KDOQI Guidelines and
endorsed at subsequent KDIGO Controv ersies Conferences
with minor modifications.
30,31
The definition of CKD proposed
here is intended for use in clinical practice, research and public
health, and has not changed. Thus, the updated version does
notchangeanyoftheinitiativesthathavebeencommenced
with respect to public policy. We recognize the variation around
the world regar ding measurement of urine albumin versus total
protein in clinical practice, and we anticipate variation in
implementation of the guideline until more widespread
dissemination of the guideline has occurred. For additional
discussion about methods for ascertainment of urine albumin
versus total protein, see R ec ommendation 1.4.4 (Evaluation of
albuminuria). The implications of highlighting the importance
of albuminuria for general practitioners in evaluation and
prognostication may help with identification and care planning.
Nonetheless, a number of concerns about the definition remain,
which are clarified below .
30,32–36
Areas of Controversy, Confusion, or Non-consensus and
Clarification of Issues and Key Points
General concerns:
a) The use of single thresholds without consideration of
patient specific factors
The use of single thresholds to define decreased GFR and
increased AER, without consideration for cause of disease,
age, sex, race-ethnicity and clinical context is consistent with
the use of single thresholds for disease markers to define
other chronic non-communicable diseases, such as hyperten-
sion, diabetes, and hypercholesterolemia, that primarily affect
the elderly and are associated with an increased risk for
cardiovascular mortality. Biologic variability and error in
ascertainment of GFR and AER can lead to misclassification
and false negative and false positive diagnosis. Furthermore,
these single thresholds appear to differentiate groups of
individuals and outcomes, irrespective of specific patient
characteristics in a multitude of studies. However, they
correspond to thresholds for RRs for complications, rather
than predictions of absolute risk. Furthermore, as with any
diagnostic tests, findings must be interpreted with considera-
tions of likelihood of disease based on the clinical context but
this should not negate the application of a standard
definition for CKD.
Specific concerns:
b) Relationship of CKD criteria to aging
Epidemiologic studies show an increased prevalence of
decreased eGFR and increased ACR in older subjects. There
has been vigorous debate as to whether decreased GFR or
increased ACR in older people represent a disease or ‘‘normal
aging.’’ Numerous studies show pathologic abnormalities
associated with aging, including glomerular sclerosis, tubular
atrophy a nd vascular sclerosis. The cause for this association
is not clear but has been hypothesized to reflect disparate
processes, such as vascular disease or senescence.
37–39
Irrespective of cause, there appears to be increased risk
associated with decreased eGFR or increased ACR in older
people, and for this reason, we consider all individuals with
persistently decreased GFR or increased albuminuria to have
CKD. Comparison of the magnitude of risk to younger
individuals is complicated. As with other CVD risk factors,
absolute risk appears to be higher in older than in younger
individuals, but RR appears to be lower.
3-5
Note is also made
that healthy older individuals do not necessarily have
decreased GFR, so that while one may expect some decline,
levels below 60 ml/min/1.73 m
2
in individuals without
comorbidity is the exception.
20
c) Isolated decreased GFR without markers of kidney
damage
A variety of clinical circumstances are associated with GFR
o60 ml/min/1.73 m
2
for 43 months in the absence of
known structural alterations. Below are examples of these
conditions and the rationale for considering them as CKD:
K Heart failure, cirrhosis of the liver, and hypothyroidism.
Decreased GFR complicates the management of the
primary disease and patients with these disorders with
decreased GFR have a worse prognosis than those
without decreased GFR. In addition, renal biopsy in
these patients may reveal renal parenchymal lesions.
K Kidney donors. The usual level of GFR in kidney donors
after transplantation is approximately 70% of the pre-
donation level, in the range of 60-90 ml/min/1.73 m
2
in
most donors. However, a minority of donors have GFR
o60 ml/min/1.73 m
2
. The prognosis of these donors
24 Kidney International Supplements (2013) 3, 19–62
chapter 1

compared to those with higher GFR has not been
carefully studied. However, as with decreased GFR due
to recognized kidney diseases, donors with decreased
GFR require closer follow-up for adjustment of drug
doses.
K Malnutrition. The level of GFR is affected by habitual
protein intake.
40
Healthy adults with lower protein
intake may have lower mean GFR, but usually do not
have GFR o60 ml/min/1.73 m
2
. Older studies of
patients with protein-calorie malnutrition and more
recent studies of subjects with anorexia nervosa have
documented reduced measured GFR that can improve
following restoration of nutri tional status. However,
renal biopsies may reveal structural abnormalities in
these conditions and decreased GFR can complicate their
management.
d) Isolated albuminuria without decreased GFR
As described later, transient ACR Z30 mg/g (Z3 mg/
mmol) can occur in disorders other than CKD. Remission of
albuminuria within 3 months in association with recovery
from these disorders is not defined as CKD. Patients with
persistent albuminuria would be considered to have CKD.
Below are examples of these conditions and the rationale for
considering them as CKD:
K Obesity and metabolic syndrome. Albuminuria can be
associated w ith obesity and metabolic syndrome, and can
remit during weight loss. The mechanism of albuminuria
in these conditions is not known but renal biopsies may
reveal prominent vascular lesions. Patients with obesity
and metabolic syndrome are at increased risk for
development of diabetes and hypertension. The risk of
persistent albuminuria in this condition has not been
carefully studied.
K Orthostatic (postural) protei nuria.
41
Albuminuria may
rarely be observed in the upright but not recumbent
posture in patients with the syndrome of postural
proteinuria. This condition is not associate d with an
increased risk of long-term adverse outcomes but a
thorough evaluation is required to exclude other causes
of CKD. Exclusion is generally possible by study ing a first
pass early morning urine (EMU) after overnight
recumbency: total protein loss of 41000 mg/24 hours is
unlikely to be explained by orthostatic proteinuria.
e) Remission of decreased GFR or markers of kidney
damage
If decreased GFR and markers of kidney damage resolve
while on treatment, the patient would be considered to have
treated CKD, consistent with nomenclature for treated
hypertension, treated diabetes, or treated hypercholesterole-
mia if blood pressure, blood glucose and blood cholesterol
are within normal range while on medications. If resolution
of decreased GFR and markers of kidney damage is sustained
after withdrawal of treatment, the patient would be
considered to have a history of CKD.
f) Kidney disease in the absence of decreased GFR and
markers of kidney damage
A GFR Z60 ml/min/1.73 m
2
may reflect a decline from a
higher value, and an AER of o30 mg/24 hours (ACR
o30 mg/g or o3 mg/mmol) may reflect a rise from a lower
value. Both findings may be asso ciated with a pathologic
process, even in the absence of other markers of kidney
damage. Although such pati ents do not fulfill the criteri a for
CKD, a clinician’s high index of suspicion may warrant
additional diagnostic testing or close follow-up to detect the
onset of CKD.
Pediatric Considerations
In general the definition of CKD in adults applies to children
(birth-18 years) with the followi ng exceptions or allowances:
K the criteria for duration 43 months does not apply to
newborns or infants r3 months of age.
K the criteria of a GFR o60 ml/m in/1.73 m
2
does not apply
to children o2 years of age in whom an age appropriate
value should be applied.
K a urinary total protein or albumin excretion rate above
the normal value for age may be substituted for
albuminuria Z30 mg/24 hours.
K all electrolyte abnormalities are to be defined in light of
age normative values.
Developmental renal abnormalities account for as many as
30-50% of the children with CKD or ESRD.
42
As such many
infants while born with normal SCr for age will in fact meet
the definition of CKD based on structural abnormalities
despite the appearance of a normal GFR and may be classified
as such within the first few days of life.
Normal GFR in newborns is less than 60 ml/min/1.73 m
2
,
and it is not until approximately 2 years of age that one
expects to see body surface area (BSA) adjusted GFR values
comparable to those seen in the adult.
43,44
The expected
increases in GFR that occur in the first months of life are due
to increases in mean arterial pressure (MAP), decrease in
renal vascular resistance, and redistribution of intrarenal
blood flows to the superficial cortical nephrons in the
newborn and increases in glomerular size and capillary
permeability in the infant.
45–48
As such direct application
of the GFR threshold values in the current CKD definition
would not be appropriate in children less than 2 years
of age as their normative maximal values would be below
those of the adult or older child; hence most neonates
and infants would be classified a priori at a decreased GFR
based not on a reduction in GFR from a higher value, but
rather failure of maturity of the kidney.
Numerous references exist for fetal,
49
neonatal term,
44,48
pre-term,
46,50,51
infant, child and adolescent GFR values
43,44
and the reader is strongly encouraged to use such references
when comparison to a normative range is required for
approximating the reduction in renal clearance of the
individual child. It should be noted that across these ages
the method of GFR measurement has often varied with the
Kidney International Supplements (2013) 3, 19–62 25
chapter 1

majority of such measurements in the neonate (term or
preterm) or infant being derived from urinary collections and
creatinine clearance (CrCl) measurements, whereas the older
children and adolescents are often investigated with exo-
genous markers including inulin, radionuclides, and other
markers such as iohexol or iothalamate.
The most comprehensive list of GFR based on the gold
standard of inulin clearance and stratified by age for
both term and preterm babies and children up to the
age of young adults can be found in Schwartz and Furth’s
review on GFR measurements and estimation in pediatric
CKD.
52
Similarly, age relevant normative values should be
utilized when interpreting urinary protein (albumin)
excretions as well as other important urinary and
serum laboratory values. Such values may be found in a
number of pediatric nephrology texts. For neo nates
and infants this includes Waters
53
and for post-neonate to
young adults, more comprehensive values can be found in
Langlois.
54
1.2 STAGING OF CKD
1.2.1: We recommend that CKD is classified based on
cause, GFR category, and albuminuria category
(CGA). (1B)
RATIONALE
This statement is worded in this way because a classification
encompassing cause and severity, as expressed by the level of
GFR and the level of albuminuria, links to risks of adverse
outcomes including mortality and kidney outcomes. These
factors will therefore guide management of CKD and this
recommended classification is consistent with other classifi-
cation systems of disease which are based on the general
domains of cause, duration and severity which provide a
guide to prognosis. We included only kidney measures as
factors in the classification of kidney disease, although we
acknowledge that factors other than kidney measures, such as
level of BP, also affect prognosis in CKD.
This recommended staging with inclusion of two additional
domains represents a revision of the previous CKD guidelines,
which included staging only by level of GFR. Cause of disease is
included because of its fundamental importance in pr edicting
the outc ome of CKD and choice of cause-specific tr eatments.
W ith inclusion of cause of kidney disease in the classification,
we considered that it was no longer necessary to retain the use
of the letter ‘ ‘T’’ to refer to kidney transplant recipients.
Albuminuria is included as an additional expr ession of severity
of disease not only because it is a marker of the severity of
injury but also because albuminuria itself strongly associates
with progr ession of kidney disease. Numerous studies have
identified the adverse prognostic implication of albuminuria
irrespectiv e of level of kidney function.
We propose that this classification of CKD by
Cause, GFR
and
Albuminuria, respectively be referred to as CGA
staging. It can be used to inform the need for specialist
referral, general medical management, and indications for
investigation and therapeutic interventions. It will also be a
tool for the study of the epidemiology, natural history, and
prognosis of CKD.
Pediatric Considerations
The principles inherent in this guideline are fully applicable
to children.
While large scale trials in children relating cause, GFR and
albuminuria or proteinuria are rare, the principles of a
multimodal classification in these three spheres should apply
to children.
To date the only large scale trial utilizing a validated
exogenously measured GFR (iohexol) and urinar y protein
excretion in a well-described cohort of children with renal
disease is the Chronic Kidney Disease in Children (CKiD)
trial.
55
They have enrolled over 600 children aged 1-16 years
and have described GFR and urinary proteinuria related
outcomes in the areas of neurodevelopment, cognition,
behavior, cardiovascular health and risk, and somatic growth.
They have also collected samples fo r ongoing and future
genetic study. While these data are sparse in relation to
overall adult numbers, this represents one of the largest
pediatric nephrology trials. The use of true measured GFR,
the quality and completeness of the data, and the long term
longitudinal follow-up will form the basis for the best
evidence-based outcomes in children w ith CKD for the
foreseeable future. A recent review article by Copelovitch
et al.
56
summarizes the major findings of the trial up to the
present time.
1.2.2: Assign cause of CKD based on presence or absence of
systemic disease and the location within the kid ney
of observed or presumed pathologic-anatomic
findings. (Not Graded)
RATIONALE
This statement has been included so as to ensure that
clinicians are alerted to the fact that CKD is not a diagnosis in
and of itself, and that the assignmen t of cause is important
for prognostication and treatment.
The cause of CKD has been traditionally assigned
based on presence or absence of underlying systemic
diseases and location of known or presumed pathologic-
anatomic abnormalities. The distinction between systemic
diseases affecting the kidney and primary kid ney diseases
is based on the origin and locu s of the disease process.
In primary kidney disease the process arises and is confined
to the kidney whereas in systemic diseases the kidney
is only one victim of a specific process, for example
diabetes mellitus. Certain genetic diseases cross this bound-
ary by affecting different tissues, e.g., adult polycystic
kidney disease. The location of pathologic-anatomic findings
is based on the magnitude of proteinuria, findings from the
urine sediment examination, imaging, and renal pathol ogy.
26 Kidney International Supplements (2013) 3, 19–62
chapter 1

Table 4 represents an example of a classification of causes
of kidney diseases based on these two domains.
There is wide geographic variation in the cause of kidney
disease. In developed countries, hypertension and diabetes
are the most frequent causes of CKD, especially in the elderly.
In populations with a high prevalence of diabetes and
hypertension, it can be difficult to distinguish CKD due to
hypertension and diabe tes from CKD due to other disorders.
In other countries, other causes of CKD may be as frequent as
hypertension and diabetes (e.g., glomerular di sease in East
Asia) or coexist with them. Spe cialized diagnostic testing,
such as kidney biopsy or invasive imaging studies are
performed only when it is essential to confirm some
diagnoses and the benefits justify the risks and cost. It is
anticipated that cause of disease will not be known with
certainty for many patients with CKD but can be either
inferred or not known.
Pediatric Considerations
The principles inherent in this guideline are fully applicable
to children.
1.2.3: Assign GFR categories as follows [Table 5] (Not
Graded):
RATIONALE
The purpose of this statement is to ensure clarity in
communication. The terms associated with each of the
GFR categories are descriptors which need to be taken in
the context of the individual and are all references to
normal young adults. Note that mildly decreased kidney
function (G2) in the absence of other markers, does not
constitute CKD.
The associations of lower categories of GFR and risks of
metabolic and endocrine complications formed the basis of the
previous stratification into 5 stages. This current classification
further acknowledges the importance of dividing Stage 3 based
on data supporting different outcomes and risk profiles into
categories G3a and G3b (Figure 5). A number of other
concurrent complications are associated with decreased cate-
gories of GFR including infection, impaired cognitive and
physical function, and threats to patient safety.
57
Figures 6 and 7 detail the RRs of decreased eGFR
and increasing ACR with future complications, including
mortality and kidney outcomes.
30
Even for the group with
the lowest value of albuminuria, the increased RR for all
outcomes is significant for eGFRs below 60 ml/min/1.73 m
2
in the continuous analysis and in the range of 45–59 ml/min/
1.73 m
2
for the categorical analysis.
Pediatric Considerations
In children o2 years of age with CKD, the GFR categories as
per the adult in Table 5 do not apply; these children should
be categorized as having normal, moderately reduced, or
severely reduced age-adjusted GFR.
No currently agreed upon set of international normative
values or categories exist for GFR in children under the age of
1-2 years. However, the international pediatric nephrology
community has embraced the adult CKD staging system as
per the 2002 KDOQI guidelines in children over the age of
2 years, as suggested by Hogg et al.
43
Table 4 | Classification* of CKD based on presence or absence of systemic disease and location within the kidney of pathologic-
anatomic findings
Examples of systemic diseases
affecting the kidney
Examples of primary kidney diseases (absence of
systemic diseases affecting the kidney)
Glomerular diseases Diabetes, systemic autoimmune diseases, systemic
infections, drugs, neoplasia (including amyloidosis)
Diffuse, focal or crescentic proliferative GN; focal and
segmental glomerulosclerosis, membranous nephropathy,
minimal change disease
Tubulointerstitial
diseases
Systemic infections, autoimmune, sarcoidosis,
drugs, urate, environmental toxins
(lead, aristolochic acid), neoplasia (myeloma)
Urinary-tract infections, stones, obstruction
Vascular diseases Atherosclerosis, hypertension, ischemia, cholesterol
emboli, systemic vasculitis, thrombotic
microangiopathy, systemic sclerosis
ANCA-associated renal limited vasculitis, fibromuscular
dysplasia
Cystic and congenital
diseases
Polycystic kidney disease, Alport syndrome,
Fabry disease
Renal dysplasia, medullary cystic disease, podocytopathies
Abbreviations: ANCA, antineutrophil cytoplasmic antibody; CKD, chronic kidney disease, GN, glomerulonephritis
Genetic diseases are not considered separately because some diseases in each category are now recognized as having genetic determinants.
*Note that there are many different ways in which to classify CKD. This method of separating systemic diseases and primary kidney diseases is only one, proposed by the
Work Group, to aid in the conceptual approach.
Table 5 | GFR categories in CKD
GFR category GFR (ml/min/1.73 m
2
) Terms
G1 Z90 Normal or high
G2 60–89 Mildly decreased*
G3a 45–59 Mildly to moderately decreased
G3b 30–44 Moderately to severely decreased
G4 15–29 Severely decreased
G5 o15 Kidney failure
Abbreviations: CKD, chronic kidney disease; GFR, glomerular filtration rate.
*Relative to young adult level
In the absence of evidence of kidney damage, neither GFR category G1 nor G2 fulfill
the criteria for CKD.
Kidney International Supplements (2013) 3, 19–62
27
chapter 1

As indicated in Pediatric Considerations for Guideline 1.1,
the normative GFR values for children less than 2 years vary
quite widely by both age and method of measurement. More
importantly these values are expected to increase in a non-
linear fashion over the first 2 years of life with significant
changes seen in the first few months post-birth and no
current evidence of presence of comorbid conditions at any
given level of measured or estimated GFR in this population.
As such, specific categorization of G1-5 as suggested in this
Recommendation would seem not be of value, and might be
misleading if applied to a child less than 2 years of age.
With this in mind, it is suggested that based on the chosen
method of GFR measurement or comparison for the
individual (i.e., CrCl, radioactive or cold exogenous serum
markers, or estimating formula), that one should attempt to
classify the child under the age of 2 years as having normal,
moderate or severe reductions in GFR based on the
normative range and standard deviations (SDs) for the
method. No evidence exists for this recommendation but
recognition that values of GFR more than 1 SD below the
mean would seem likely to raise concern of the clinician and
foster t he need for closer monitoring. For drug dosing
adjustments it is suggested that those children with GFRs
below the mean by 41 but o2 SD be classified as having a
moderate reduction in GFR whereas those more than 2 SD
below the mean for the method be classified as having a
severe reduction in GFR.
1.2.4: Assign albuminuria* categories as follows [Table 6]
(Not Graded):
*note that where albuminuria measurement is
not available, urine reagent strip resul ts can be
substituted (Table 7)
RATIONALE
The purpose of this statement is to ensure communication
and to reflect that albuminuria category is an important
predictor of outcomes. The association of high levels of
proteinuria with signs and sy mptoms of nephrotic syndrome
is well known. The detection and evaluation of lesser
quantities of proteinuria have gained additional significance
as multiple studies have demonstrated its diagnostic,
pathogenic, and prognostic importance. There is a contin-
uous risk associated with albuminuria but the use of a simple
categorical approach was selected to simplify the concept for
Figure 5 | Age-standardized rates of death from any cause
(panel a), cardiovascular events (panel b), and hospitalization
(panel c), according to the eGFR among 1,120,295 ambulatory
adults. eGFR, estimated glomerular filtration rate. From N Engl J
Med, Go AS, Chertow GM, Fan D, et al.
58
Chronic kidney disease and
the risks of death, cardiovascular events, and hospitalization, 351:
1296-1305. Copyright & (2004) Massachusetts Medical Society.
Reprinted with permission from Massachusetts Medical Society;
accessed http://www.nejm.org/doi/pdf/10.1056/NEJMoa041031
Table 6 | Albuminuria categories in CKD
AER
ACR (approximate
equivalent)
Category
(mg/24 hours) (mg/mmol) (mg/g)
Terms
A1 o30 o3 o30 Normal to mildly
increased
A2 30-300 3-30 30-300 Moderately increased*
A3 4300 430 4300 Severely increased**
Abbreviations: AER, albumin excretion rate; ACR, albumin-to-creatinine ratio;
CKD, chronic kidney disease.
*Relative to young adult level.
**Including nephrotic syndrome (albumin excretion usually 42200 mg/24 hours
[ACR 42220 mg/g; 4220 mg/mmol]).
28 Kidney International Supplements (2013) 3, 19–62
chapter 1

clinical practice. Several groups had suggested sub-
dividing one or more GFR categories based on albuminuria
category.
For the detection of diabetic nephropathy some guidelines
recommend the use of different ACR thresholds for males
and females (425 mg/g [42.5 mg/mmol] and 435 mg/g
[43.5 mg/mmol], respectively) to take into account varia-
tions in creatinine excretion. A single threshold is used in
North America (30 mg/g or 3.4 mg/mmol). Earlier KDIGO
guidance was reluctant to adopt gender-specific thresholds
due to greater complexity, uncertainty about assay precision,
and effects of race, ethnicity, diet and measures of body size
on creatinine and this stance is maintained here. For
simplicity, and to reflect the fact that it is an approximation,
3.4 mg/mmol as the current guideline threshold has been
rounded to 3.0 mg/mmol.
There is a graded increase in risk for higher albuminuria
categories, at all GFR categories, without any clear threshold
value. Even for subjects with GFR 4 60 ml/min/1.73 m
2
,
the increased RR is statistically significant for urine
ACR Z30 mg/g (Z3 mg/mmol) for mortality and kidney
outcomes (Figures 6 and 7). The predictive ability of
albuminuria at all categories of GFR supports the suggestion
to add albuminuria categories to all GFR categories. Since the
relationship with albuminuria is continuous, the selection of
the number of categories and the cutoff values appears
arbitrary. The Work Group has recommended the classifica-
tion of albuminuria into only 3 categories, based on practical
considerations, but recognized that further subdivisions
within the category of o30 mg/24 hours (ACRo30 mg/g
or o3 mg/mmol) may be useful for risk stratification,
and that subdivisions within the category of 4300 mg/
24 hours (ACR4300 mg/g or 4 30 mg/mmol) may be
useful for diagnosis and management. Specifically there
is a recognition that nephrotic range proteinuria
(AER42200 mg/24 hours [ACR42200 mg/g;4220 mg/
mmol]; PER43000 mg/24 hours [43000 mg/g;4300 mg/
mmol]) confers uni que additional risks and is usually
associated with specific conditions (such as GN). As these
are relatively rare in general practices, the simplicity of the
AER categorization was preferred. Table 7 shows the
approximate relationships of categories of AER to other
measures of albuminuria and proteinuria.
Implications for Clinical Practice and Public Policy
Data from around the world suggest that CKD prevalence is
between 10-16% bu t information concerning population
prevalence by category of GFR and ACR is scant. Figure 8
shows the proportion of adults in the US by categories
of GFR and albuminuria.
19
While CKD is common, few
individuals have severely reduced GFR or kidney failure or
severely increased albuminuria.
0.5
1
2
4
8
16
Adjusted HR
15 30 45 60 75 90 105 120
eGFR, ml/min per 1.73 m
2
All-cause mortality
0.5
1
2
4
8
16
Adjusted HR
15 30 45 60 75 90 105 120
eGFR, ml/min per 1.73 m
2
Cardiovascular mortality
Summary of
relative risks
from
continuous
meta-analysis
0.5
1
4
16
64
256
1024
8192
Adjusted HR
15 30 45 60 75 90 105 120
eGFR, ml/min per 1.73 m
2
End stage renal disease
0.5
1
4
16
64
Adjusted HR
15 30 45 60 75 90 105 120
eGFR, ml/min per 1.73 m
2
Acute kidney injury
0.5
1
4
16
64
256
Adjusted OR
15 30 45 60 75 90 105
120
eGFR, ml/min per 1.73 m
2
Progressive CKD
Figure 6 | Summary of continuous meta-analysis (adjusted RRs) for general population cohorts with ACR. Mortality is reported for
general population cohorts assessing albuminuria as urine ACR. Kidney outcomes are reported for general population cohorts assessing
albuminuria as either urine ACR or reagent strip. eGFR is expressed as a continuous variable. The three lines represent urine ACR of o30,
30-299 and Z300 mg/g (o3, 3-29, and Z30 mg/mmol, respectively) or reagent strip negative and trace, 1 þ positive, Z2 þ positive. All
results are adjusted for covariates and compared to reference point of eGFR of 95 ml/min/1.73 m
2
and ACR of o30 mg/g (o3 mg/mmol) or
reagent strip negative (diamond). Each point represents the pooled RR from a meta-analysis. Solid circles indicate statistical significance
compared to the reference point (P o0.05); triangles indicate non-significance. Red arrows indicate eGFR of 60 ml/min/1.73 m
2
, threshold
value of eGFR for the current definition of CKD. ACR, albumin-to-creatinine ratio; CKD, chronic kidney disease; eGFR, estimated glomerular
filtration rate; HR, hazard ratio; OR, odds ratio, RR, relative risk. Reprinted with permission from Macmillan Publishers Ltd: Kidney
International. Levey AS, de Jong PE, Coresh J, et al. The definition, classification, and prognosis of chronic kidney disease: a KDIGO
controversies conference report. Kidney Int 2011; 80: 17-28
30
; accessed http://www.nature.com/ki/journal/v80/n1/full/ki2010483a.html
Kidney International Supplements (2013) 3, 19–62 29
chapter 1

The classification of kidney disease by cause, category of
GFR and category of albuminuria does not conform to the
International Classification of Diseases (ICD) maintained by
the World Health Organization (WHO). Currently the WHO
is developing an update (ICD 11). It will be important to
communicate and coordinate efforts with the kidney disease
subgroup for ICD 11. However, the proposed current
classification does address the need in clinical practice to
acknowledge the multiple dimensions and variables by which
individual patients are assessed. Table 8 gives examples of the
use of CGA nomenclature.
Definition of GFR categories have been delibera tely based
upon the concept of ‘‘true’’ GFR, whereas clinical practice
and research has predominantly used creatinine-based
estimates of GFR. The belief of the Work Group is that the
non-GFR determinants of creatinine and the imprecision of
creatinine-based GFR estimates have resulted in the absence
of strong dose-dependent association of eGFR with clinical
outcomes in the GFR range of 460 ml/min/1.73 m
2
. The
Work Group felt confident that GFR levels of Z90 ml/min/
1.73 m
2
portend better prognosis than GFR levels 60-89 ml/
min/1.73 m
2
, if they could be estimated accurately. Therefore,
the GFR categories include separate G1 (Z90 ml/min/
1.73 m
2
) and G2 (60-89 ml/min/1.73 m
2
) designations des pite
limited data from creatinine-based estimates that prognosis
differs between these two categories. It is also an acknowl-
edgement that the degree of precision of some of our
measurements may not be able to differentiate between these
2 categories reliably. As described later, studies that have used
cystatin C have found gradients in prognosis at eGFR levels
above 60 ml/min/1.73 m
2
, which supports the belief of the
committee that separating these 2 GFR categories is
appropriate for CKD classification.
Albuminuria categories are ‘‘wide’’ with respect to risk,
with significant gradients within each category. The decision
to propose only 3 categories is based on the perceived need
for simplification in clinical practice. In specialized clinical
nephrology centers, A3 (4300 mg/g or 430 mg/mmol) is
often more precisely assessed and divided into additional
categories. For example, nephrotic range proteinuria is
defined as PER43500 mg/24 hours or PCR (protein-
to-creatinine ratio) 43500 mg/g [4350 mg/mmol] which
is approximately equivalent to AER42200 mg/24 hours
or ACR42200 mg/g [220 mg/mmol]. It is clearly recognized
ACR
ACR ACR
ACR
300
All-cause mortality
ACR
ACR ACR
ACR
Cardiovascular mortality
1.1 1.5 2.2 5.0
Ref 1.4 1.5 3.1
1.0
1.3 1.7 2.3
eGFR
> 105
0.9 1.3 2.3 2.1
eGFR
90–105
Ref 1.5 1.7 3.7
eGFR
1.0 1.3 1.6 3.7
1.0 1.4 1.8 2.7
1.3 1.7 2.2 3.6
eGFR
60–75
1.1 1.4 2.0 4.1
eGFR
45–60
1.5 2.2 2.8 4.3
eGFR
5.3 3.6 4.7 6.6
30–45
eGFR
15–30
14 7.9 4.8 8.1
Kidney failure (ESRD) Acute kidney injury (AKI)
Progressive CKD
ACR
<10
ACR
10–29
ACR
30–299
ACR
300
eGFR
> 105
Ref Ref 7.8 18
eGFR
Ref Ref 11 20
ACR
<10
ACR
10–29
ACR
30–299
ACR
300
eGFR
> 105
Ref Ref 0.4 3.0
eGFR
ACR
<10
ACR
10–29
ACR
30–299
ACR
300
eGFR
> 105
Ref Ref 2.7 8.4
eGFR
eGFR
75–90
Ref Ref
3.8 48
eGFR
60–75
Ref Ref
7.4 67
eGFR
90–105
eGFR
75–90
Ref Ref 1.9 5.0
eGFR
60–75
Ref Ref
Ref Ref
3.2 8.1
FR
90–105
.
eGFR
75–90
Ref Ref
2.5 4.1
eGFR
60–75
Ref Ref 3.3 6.4
45–60
eGFR
30–45
56 74 294 763
eGFR
15–30
433 1044 1056 2286
45–60
.
eGFR
30–45
3.0 19 15 22
eGFR
15–30
4.0 12 21 7.7
45–60
.
6.
4
<10 10–29 30–299
<10
10–29 30–299
300
75–90
eGFR
> 105
eGFR
90–105
eGFR
eGFR
60–75
eGFR
45–60
eGFR
30–45
eGFR
15–30
75–90
1.9 2.3 3.3 4.9
2.2
2.7 5.23.4
90–105
0.9
3.3
Ref
Ref
24
5.8
5.2 22 40 147
eG
31 4.0
9.4 57
2.2
49 5.9
eGFR
eGFR
30–45
7.3 10 12 20
eGFR
15–30
17 17 21 29
Summary of
relative risks
from
categorical
meta-analysis
(dipstick included)
(–, ±, +, ++)
Figure 7 | Summary of categorical meta-analysis (adjusted RRs) for general population cohorts with ACR. Mortality is reported for
general population cohorts assessing albuminuria as urine ACR. Kidney outcomes are reported for general population cohorts assessing
albuminuria as either urine ACR or reagent strip. eGFR and albuminuria are expressed as categorical variables. All results are adjusted for
covariates and compared to the reference cell (Ref). Each cell represents a pooled RR from a meta-analysis; bold numbers indicate statistical
significance at P o0.05. Incidence rates per 1000 person-years for the reference cells are 7.0 for all-cause mortality, 4.5 for CVD mortality,
0.04 for kidney failure, 0.98 for AKI, and 2.02 for CKD progression. Colors reflect the ranking of adjusted RR. The point estimates for each cell
were ranked from 1 to 28 (the lowest RR having rank number 1, and the highest number 28). The categories with a rank number 1-8 are
green, rank numbers 9-14 are yellow, the rank numbers 15-21 are orange and the rank numbers 22-28 are colored red. (For the outcome of
CKD progression, two cells with RR o1.0 are also green, leaving fewer cells as yellow, orange and red). ACR, albumin-to-creatinine ratio; AKI,
acute kidney injury; CKD, chronic kidney disease; CVD, cardiovascular disease; eGFR, estimated glomerular filtration rate; ESRD, end-stage
renal disease; RR, relative risk. Reprinted with permission from Macmillan Publishers Ltd: Kidney International. Levey AS, de Jong PE, Coresh J,
et al.
30
The definition, classification, and prognosis of chronic kidney disease: a KDIGO controversies conference report. Kidney Int 2011; 80:
17-28; accessed http://www.nature.com/ki/journal/v80/n1/full/ki2010483a.html
30 Kidney International Supplements (2013) 3, 19–62
chapter 1
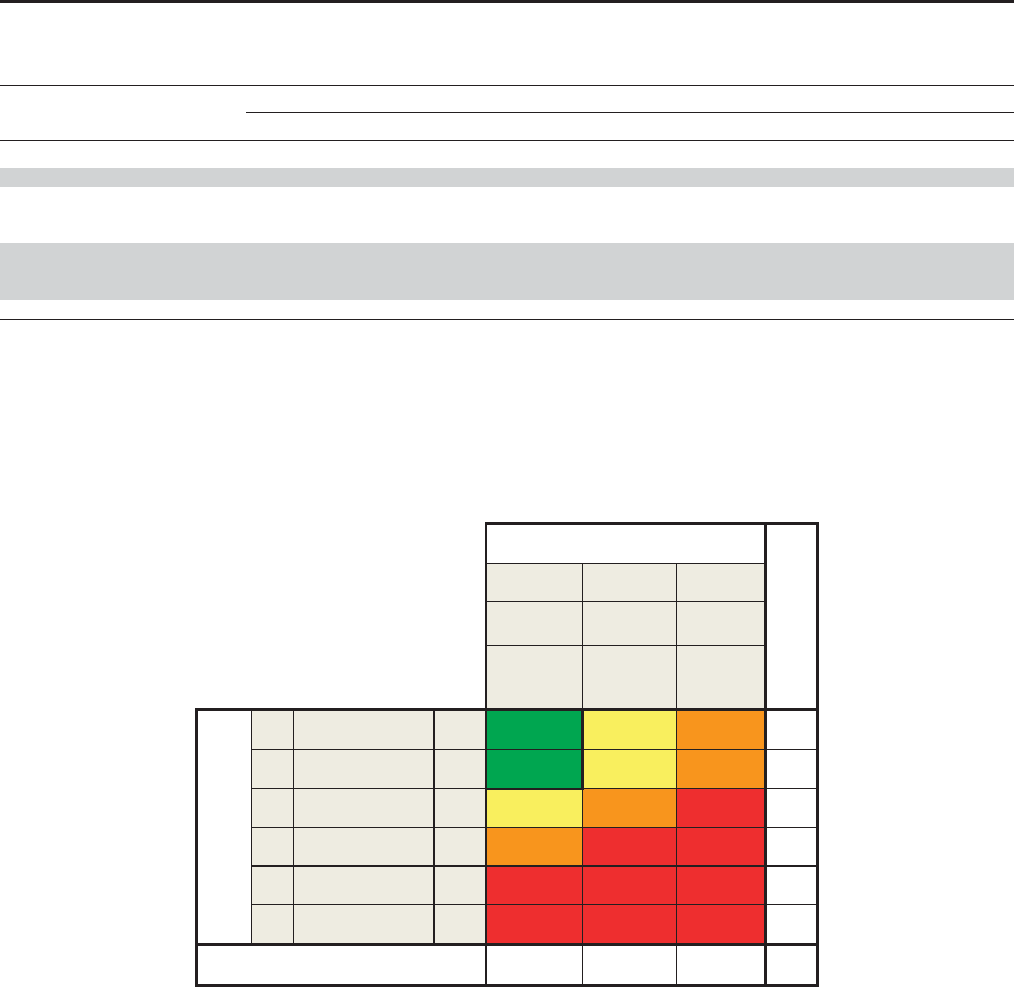
that these very high levels of proteinuria carry a different
risk than lower values with in the same category. Further
differentiation after quantification and evaluation would
inform treatment decisions for an individual patient. These
categories ser ve as an initial assessment and prognostication
tool; further classification is appropriate for specific circum-
stances and is not limited by the initial classification into only
3 categories.
Note that the term ‘microalbuminuria’ is not used and is
discouraged in this classification system. This will require a
formal education program and review of existing guidelines
in other disciplines so that consistency of terminology and
Table 7 | Relationship among categories for albuminuria and proteinuria
Categories
Measure Normal to mildly increased (A1) Moderately increased (A2) Severely increased (A3)
AER (mg/24 hours) o30 30–300 4300
PER (mg/24 hours) o150 150–500 4500
ACR
(mg/mmol) o33–30430
(mg/g) o30 30–300 4300
PCR
(mg/mmol) o15 15–50 450
(mg/g) o150 150–500 4500
Protein reagent strip Negative to trace Trace to + + or greater
Abbreviations: ACR, albumin-to-creatinine ratio; AER, albumin excretion rate; PCR, protein-to-creatinine ratio; PER, protein excretion rate.
Albuminuria and proteinuria can be measured using excretion rates in timed urine collections, ratio of concentrations to creatinine concentration in spot urine samples, and
using reagent strips in spot urine samples. Relationships among measurement methods within a category are not exact. For example, the relationships between AER and ACR
and between PER and PCR are based on the assumption that average creatinine excretion rate is approximately 1.0 g/d or 10 mmol/d. The conversions are ro unded for
pragmatic reasons. (For an exact conversion from mg/g of creatinine to mg/mmol of creatinine, multiply by 0.113.) Creatinine excretion varies with age, sex, race and diet;
therefore the relationship among these categories is approximate only. ACR o10 mg/g (o1 mg/mmol) is consid ered normal; ACR 10–30 mg/g (1-3 mg/mmol) is considered
‘‘high normal.’’ ACR 42200 mg/g (4220 mg/mmol) is considered ‘‘nephrotic range.’’ The relationship between urine reagent strip results and other measures depends on
urine concentration.
A2
A3
G1
Normal or high
≥90
55.6
1.9
0.4
57.9
G2
Mildly decreased
32.9
2.2
0.3
35.4
G3a
3.6
0.8
0.2
4.6
1.0 0.4 0.2 1.6
G4
Severely decreased
Moderately to
severely decreased
Mildly to moderately
decreased
60-89
45-59
30-44
G3b
0.2
0.1
0.1
0.4
G5
Kidney failure
15-29
<15
0.0
0.0
0.1
0.1
93.2
5.4
1.3
100.0
Percentage of US Population by
eGFR and Albuminuria
Category: KDIGO 2012 and
NHANES 1999-2006
Persistent albuminuria categories
Description and range
A1
Normal to
mildly
increased
Moderately
increased
Severely
increased
>300 mg/g
>30mg/mmol
30-300 mg/g
3-30 mg/mmol
<30 mg/g
<3 mg/mmol
GFR categories (ml/min/ 1.73m
2
)
Description and range
Figure 8 | Prevalence of CKD in the USA by GFR and albuminuria. Cells show the proportion of adult population in the USA. Data from
the NHANES 1999-2006, N ¼ 18,026. GFR is estimated with the CKD-EPI equation and standardized serum creatinine.
19
Albuminuria is
determined by one measurement of ACR and persistence is estimated as described elsewhere.
59
Values in cells do not total to values in
margins because of rounding. Category of very high albuminuria includes nephrotic range. Green, low risk (if no other markers of kidney
disease, no CKD); Yellow, moderately increased risk; Orange, high risk; Red, very high risk. ACR, albumin-to-creatinine ratio; CKD, chronic
kidney disease; CKD-EPI, CKD Epidemiology Collaboration; GFR, glomerular filtration rate; NHANES, National Health and Nutrition
Examination Survey. Modified with permission from Macmillan Publishers Ltd: Kidney International. Levey AS, de Jong PE, Coresh J, et al.
30
The definition, classification, and prognosis of chronic kidney disease: a KDIGO controversies conference report. Kidney Int 2011; 80: 17-28;
accessed http://www.nature.com/ki/journal/v80/n1/full/ki2010483a.html
Kidney International Supplements (2013) 3, 19–62 31
chapter 1

understanding of the changes are universal (see Recommen-
dation 1.4.4.2.1).
Pediatric Considerations
This statement would need to be altered for application in
pediatric practice in the following way. In children with CKD
any expression of abnormal urinary protein excretion,
irrespective of the marker:
K must account for variation in that measurement as seen
across age, sex, puberty and or body size (height, weight,
body mass index [BMI]).
K should account for the possi bility of tubular versus
glomerular proteinuria dominance dependent on the
underlying disease.
K may utilize proteinuria in place of albuminuria.
There is no set standard encompassing all children with
respect to the normal range of urinary protein (or albumin)
excretion. Values vary across age, sex, race, pubertal status,
the presence of obesity (high BMI) and may be modified by
exercise, fever, and posture.
60–63
In general, neonates and young infants/ children are both
expected and allowed to have higher urinary losses of both
glomerular and tubular proteinuria due to lack of maturation
in the proximal tubular reabsorption of proteins. The rough
equivalences for ACR and PCR quoted in the pediatric
literature are similar, but not identical to those quoted in the
adult literature. Normal ranges vary but at least one reference
suggests as much as 6-8 mg/m
2
/hr or 4240 mg/m
2
/day of
proteinuria as being acceptable at o6 months of age;
64
normal ranges for urinary albumin losses are not known at
this age.
The normal range of protein excretion for children 6-24
months of age in a 24-hour urine collection is quoted as
being o4 mg/m
2
/hr (o150 mg/m
2
/day), whereas the first
morning spot urine protein sample is said to be normal at
levels of o500 mg/g creatinine (o50 mg/mmol). In children
older than 24 months these values are o4 mg/m
2
/hr
(o150 mg/m
2
/day) for the 24-hour collection and PCR
o200 mg/g creatinine (o20 mg/mmol) in the first morning
urine sample, or a first morning urine ACR o30 mg/g
(o3 mg/mmol).
43,65
At all ages, total urinary protein excretion 440 mg/m
2
/hr
(43 grams/1.73 m
2
/day) is considered to represent ‘nephrotic
range’ loss of protein, with intermediate values, i.e.,
4-40 mg/m
2
/hr or its equivalent representing abnormal but
‘non-nephrotic’ losses.
43,65
Children older than 24 months of age are expected to
achieve normal (‘adult’) urinary protein values with the
caveat of an exagg erated postural loss of glomerular proteins
(albumin) as can commonly be seen in the 2-5% of the
adolescent population (i.e., orthostatic proteinuria).
62
Based on National Health and Nutrition Examination
Survey III (NHANES III) data from just under 6000 healthy
6-19 year old children using either immunonephelometry or
radioimmunoassay, the definition of urinary albumin excre-
tion was determined to be 30-300 mg/24 h collection;
20-200 mg/min in an overnight collection and 30-300 mg/g
creatinine (3-30 mg/ mmol) in a first morning uri ne sample.
66
Of note, to date the majority of studies that have
examined the effects of urinary protein losses or therapeutic
interventions have concentrated on so-called total protein
excretion or random or first morning PCRs. The utility
of measuring the albumin only fraction, and in particular
Table 8 | CGA staging of CKD: examples of nomenclature and comments
Cause
GFR
category
Albuminuria
category Criterion for CKD Comment
Diabetic kidney disease G5 A3 Decreased GFR, Albuminuria Most common patient in the low clearance clinic
Idiopathic focal sclerosis G2 A3 Albuminuria Common cause of nephrotic syndrome in childhood
Kidney transplant
recipient
G2 A1 History of kidney transplantation Best outcome after kidney transplantation
Polycystic kidney disease G2 A1 Imaging abnormality Most common disease caused by a mutation in a single
gene
Vesicoureteral ref lex G1 A1 Imaging abnormality Common condition in children
Distal renal tubular
acidosis
G1 A1 Electrolyte abnormalities Rare genetic disorder
Hypertensive kidney
disease
G4 A2 Decreased GFR and albuminuria Usually due to long-standing poorly controlled
hypertension, likely to include patients with genetic
predisposition- more common in blacks- who should
be referred to nephrologist because of severely
decreased GFR
CKD presumed due to
diabetes and hypertension
G4 A1 Decreased GFR Should be referred to nephrologist because of severely
decreased GFR
CKD presumed due to
diabetes and hypertension
G2 A3 Albuminuria Should be referred to nephrologist because of
albuminuria
CKD presumed due to
diabetes and hypertension
G3a A1 Decreased GFR Very common, may not require referral to nephrologist
CKD cause unknown G3a A1 Decreased GFR May be the same patient as above
Abbreviations: CGA, Cause, GFR category and albuminuria category; CKD, chronic kidney disease; GFR, glomerular filtration rate.
Note: Patients above the thick horizontal line are likely to be encountered in nephrology practice. Patients below the thick horizontal line are likely to be encountered in
primary care practice and in nephrology practice.
32 Kidney International Supplements (2013) 3, 19–62
chapter 1

quantitating this at the lower level of detection, i.e.,
o30 mg/g (o3 mg/mmol) creatinine, is only now being
investigated in more detail in large pediatric studies. As such
it should be recognized that in children the quantification of
total protein, as compared to the albumin only fraction, may
be the preferred method for assigning risk as it relates to
the presence of urinary protein loss.
In summary, for children older than 2 years of age the
assignment of ‘proteinuria’ categories can be used as per the
adult guidelines with the understanding that modification to
the upper limit of expected values may be necessary in
consideration of the factors outlined above. Although there is
a preference for reporting albumin values, currently many
clinicians still categorize these children based on total protein
and in the child o2 years of age or the adolescent with
demonstrable orthostatic proteinuria, the current albumin-
uria categories are unlikely to apply.
1.3: PREDICTING PROGNOSIS OF CKD
1.3.1: In predicting risk for outcome of CKD, identify
the following variables: 1) cause of CKD; 2) GFR
category; 3) albuminuria category; 4) other risk
factors and comorbid conditions. (Not Graded)
1.3.2: In people with CKD, use estimated risk of
concurrent complications and future outcomes
to guide decisions for testing and treatment for
CKD complications (Figure 9). (Not Graded)
1.3.3: In populations with CKD, group GFR and
albuminuria categories with similar relative risk
for CKD outcomes into risk categories (Figure 9).
(Not Graded)
RATIONALE
These statements are worded in this way because for all CKD
complications, prognosis will vary depending on: 1) cause; 2)
GFR; 3) degree of albuminuria; and 4) other comorbid
conditions. The relative strength of each of these factors will
vary for each complication or outcome of interest. Risk for
kidney disease end points, such as kidney failure and AKI, is
predominately driven by an individual patient’s clinical
diagnosis, GFR, and the degree of albuminuria or other
markers of kidney damage and injury. For CVD, risk will be
determined by history of CVD and traditional and non-
traditional CVD risk factors. For other conditions, the risk
will be determined by ris k factors specific for those
conditions. For all conditions, the cause of CKD, GFR
category, and albuminuria category will still have important
influence as ‘‘risk multipliers,’’ but will have smaller overall
influence on disease prediction than risk factors specific for
the condition. All these conditions have an impact on life
expectancy and quality of life (QOL) and contribute
substantially to predicting the prognosis of CKD. CKD is
associated with numerous complications directly or indirectly
related to the cause of CKD, decreased GFR, or albumi nuria
(Table 9).
The risk associations of GFR and albuminuria categories
appear to be largely independent of one another. Therefore,
neither the category of GFR nor the category of albuminuria
alone can fully capture prognosis for a patient with CKD. The
magnitude and gradients of risk across categories of GFR and
albuminuria will likely differ for each specific adverse event.
This heterogeneity across the GFR and ACR grids in RRs for
different outcomes makes it impractical to have a simple
hierarchical staging of prognosis across all cells. Thus, the
staging using CGA should be descriptive, but encompassing
the ordered categories of GFR and ACR (Fig ure 9).
The CGA staging system proposed in this guideline
provides a framework for future recommendations on CKD
clinical management. At present, much of the evidence on
clinical decision making in CKD is based solely on GFR. This
recommendation serves to highlight the multidimensional
Table 9 | Prognosis of CKD: Relationship of outcomes and strength of relationship to Cause (C), GFR (G), Albuminuria (A) and
other measures*
67,68
Kidney measures
Outcomes Cause GFR Albuminuria Other measures
Kidney outcomes
GFR decline +++ + +++ High BP, male sex, black race, younger age
Albuminuria rise +++ + +++ High BP, diabetes
AKI + +++ + Older age
Chronic kidney failure (GFR o15 ml/min/1.73 m
2
; category G5) +++ +++ + Younger age
Complications (current and future)
Drug toxicity + +++ + Drug exposure, liver disease
Endocrine and metabolic + +++ + Various
CVD and mortality ++ +++ +++ Older age, history of CVD, CVD risk factors
Others (infection, cognitive impairment, frailty, etc) ++ ++ ++ Older age, comorbid conditions
Abbreviations: AKI, acute kidney injury; BP, blood pressure; CVD, cardiovascular disease; GFR, glomerular filtration rate.
Plus signs indicate the strength of the risk relationship between the CKD characteristic and the outcome: +, somewhat associated; ++, moderately associated; +++, strongly
associated.
*Note that the + designations refer to strength of relationship not strength of evidence to support, and are based on consensus overview by the Work Group members.
Adapted with permission from Uhlig K, Levey AS.
68
Developing guidelines for chronic kidney disease: we should include all of the outcomes. Ann Intern Med 2012; 156(8):
599-601.
Kidney International Supplements (2013) 3, 19–62
33
chapter 1

aspect of CKD so as to ensure appropriate consideration of
the complexity of the condition.
Evidence Base
The evidence base from which these statements are
derived includes large observational cohort studies from
diverse populations. For some outcomes, including
mortality, CVD, and kidney disease progression, meta-
analyses have summarized the risk associations. For out-
comes that occur predominately in older adults (e.g.,
dementia, fracture), the evidence is largely limited to cohorts
of older people.
Extensive work by the CKD Prognosis Consortium has
defined the RRs across GFR and albuminuria categories for
several important outcomes, including all-cause mortality,
CVD, and kidney failure (Figures 6 and 7). Risk increases
incrementally in both directions - down the GFR categories
and across the albuminuria categories. Levels of risk can be
identified and grouped into categories, but they may di ffer
somewhat for each outcome. Additional research is needed
to map these GFR and albuminuria categories and cause
of kidney disease to other important outcomes of CKD
(Table 9).
International Relevance
The above statements appear to be robust when applied in
North America, Europe and Asia.
30
Thus, it appears for all
methods used to determine GFR and to detect albuminuria,
the use of the 3 parameters (cause, category of GFR and
category of albuminuria) influences prognosis irrespective of
ethnicity or country of origin.
Implications for Clinical Practice and Public Policy
Providers must incorporate cause of kidney disease, GFR
category and albuminuri a category in order to better develop
an accurate assessment of an individual’s prognosis related to
CKD. Many providers who are not nephrologists will need
guidance in the local methods for requesting and interpreting
a urine albumin assessment and an eGFR. Use of risk scores
which are being developed and refined is advised.
Public policy and estimates of total burden of illness in a
community need to take into account the incidence and
prevalence of specific conditions (such as diabetes and
congestive heart failure). In addition, knowledge of distribu-
tion of levels of eGFR and ACR may be valuable for resource
planning. Community or health-system based interventions
to reduce the incidence of kidney failure in populations
should be targeted and prioritized based on these 3 criteria.
The primary impact on clinical practice will relate to
kidney-specific complications of CKD and referral patterns to
help prevent and manage them. Decisions related to screen-
ing and monitoring CKD disorders will be informed and
guided by the CGA system. At present, this evidence for
issues such as management of anemia, CKD bone and
mineral disorders, and acid-base disorders has not been
organized and presented in this way.
Decisions on screening and referral strategies have major
impact on the costs and quality of health-care. The value of
Prognosis of CKD by GFR
and Albuminuria Categories:
KDIGO 2012
Persistent albuminuria categories
Description and range
A1 A2 A3
GFR categories (ml/min/ 1.73m
2
)
Description and range
G1 Normal or high ≥90
G2 Mildly decreased 60-89
G3a 45-59
G3b 30-44
G4 Severely decreased 15-29
G5 Kidney failure <15
Mildly to moderately
decreased
Moderately to
severely decreased
Severely
increased
Moderately
increased
Normal to
mildly
increased
>300 mg/g
>30 mg/mmol
30-300 mg/g
3-30 mg/mmol
<30 mg/g
<3 mg/mmol
Figure 9 | Prognosis of CKD by GFR and albuminuria category. Green, low risk (if no other markers of kidney disease, no CKD); Yellow,
moderately increased risk; Orange, high risk; Red, very high risk. CKD, chronic kidney disease; GFR, glomerular filtration rate; KDIGO, Kidney
Disease: Improving Global Outcomes. Modified with permission from Macmillan Publishers Ltd: Kidney International. Levey AS, de Jong PE,
Coresh J, et al.
30
The definition, classification, and prognosis of chronic kidney disease: a KDIGO controversies conference report. Kidney Int
2011; 80: 17-28; accessed http://www.nature.com/ki/journal/v80/n1/full/ki2010483a.html
34 Kidney International Supplements (2013) 3, 19–62
chapter 1

this revised system of classification is that it will allow the
evaluation of different referral patterns and the impact of
treatment strategies in those with diverse CGA assignment. In
this way, we will develop additional evidence which will
inform practice patterns. These will necessarily be developed
locally and reflect the values and economic realities of each
health-care system.
Areas of Controversy, Confusion, or Non-consensus and
Clarification of Issues and Key points
Current clinical practice has not overtly incorporated these 3
variables into all decision making activities. The utility of the
system will need to be vetted by those referring and those to
whom patients are referred. The overt description of the 3
dimensions of diagnosis and staging of kidney disease which
include the cause, the category of GFR and the category of
albuminuria, should help to inform referral and treatment
patterns of large groups of individuals. Risk calculators for
specific events are under development.
K The CGA classification system will be useful for
quantifying risk for specific outcomes of CKD but its
utility has not been fully assessed in clinical practice and
research studies.
K Additional evidence is requir ed before decisions on screen-
ing, monitoring, and referral patterns can be fully informed.
Pediatric Considerations
For Recommendation 1.3.1 the rationale and principles
behind this statement would apply to pediatrics though the
data are not available.
Unlike in adults, the knowledge of risk of progression or
outcomes of CKD is less robust in children, with the majority
of such information gleaned from either registry datasets or
longitudinal trials. In a 2008 report of a select group of patients
enrolled by various North American pediatric nephrology
centers in the North American Pediatric Renal Trials and
Collaborative Studies (N APRT CS) registry, 46% of nearly 7100
cases had reached a final ‘end point’ with 86% progressing to
ESRD over their time in the registry.
69
Data from the
prospective registry and population-based Italian Pediatric
Registry of Chronic Renal Failure (ItalKid) study demonstrated
a risk of progression to ESRD of B68% by age 20 years.
70
Cause of CKD. Specific information related to ra te of
progression for all pediatric causes of CKD is not easily
available. However data from the prospective longitudinal
CKiD trial has demonstrated a more rapid decline in renal
function in children whose underlying cause of CKD is
classified as glomerular with an annualized rate of change
in iohexol GFR of 10.5% as compared to those with
a non-glomerul ar cause in whom the annualized rate of
change is only 3.9%.
71
In terms of absolute rates of
change in measured iohexol GFR this translated, in a
separate analysis from the same datase t, into a median
change of GFR of 4.3 ml/min/1.73 m
2
versus 1.5 ml/min/
1.73 m
2
in the g lomerular versus non-glomerular groups,
respectively.
72
This paper also provides the only current
individual disease-specific estimate of annual decline in a
pediatric population. Table 10 illustrates that the median
values for annualized change in GFR for various diagnosis
categories.
Similarly, a randomized controlled trial (RCT) from
Europe
73
examining the effects of diet on rate of progression
demonstrated a statistically significant difference in CrCl
between their glomerular and non-glomerular cohorts at 2
years of follow-up; with the mean decline [SD] in the
glomerular group being 10.7 [11.3] versus 8.4 [13.5] ml/
min/1.73 m
2
in the non-glomerular patients (P ¼ 0.048).
GFR category. It is also well recognized that there is an
inverse relationship between the rates of progression of
kidney disease to the level of kidney function present at that
presentation with more rapid declin e seen in patients with
lower initial levels of GFR. Staples et al.
74
in their retro-
spective rev iew of the NAPRTCS CKD database involving
nearly 4200 children registered with GFR categories G2-G4
(GFR 15-89 ml/min/1.73m
2
) demonstrated significantly
higher rates of progression, defined by progression to GFR
category G5 (GFR o15 ml/min/1.73m
2
) or initiation of
dialysis or transplant, for children in GFR categories G3a-G4
(GFR 15–59 ml/min/1.73m
2
) as compared to those with
CKD and GFR category G2 (GFR 60–89 ml/min/1.73m
2
)at
time of enrollment: hazard ratio (HR) of GFR categories 3a
and 3b (GFR 30–59 ml/min/1.73m
2
) (GFR category 2 (GFR
60–89 ml/min/1.73m
2
) ¼ 1.00 as referent): 2.00; 95%
confidence interval (CI) 1.64-2.42; Po0.0001 and HR of
GFR category 4 (GFR 15–29 ml/min/1.73m
2
): 6.68 ; 95% CI
5.46-8.18; Po0.0001.
Albuminuria (proteinuria). Several studies have also de-
monstrated the effect of proteinuria on rate of progression of
CKD in children. Using registry data, and in non-glomerular
conditions the ItalKids trial
75
demonstrated a significantly
slower decline in CrCl in patients with baseline PCRs of
o200 mg/g (20 mg/mmol) and 200–900 mg/g (20–90 mg/
mmol) when compared to those patients with a PCR of
4900 mg/g (490 mg/mmol); slope þ 0.16
±
3.64 and
Table 10 | Annual percentage change in GFR across diagnosis
categories
Disease
Annualized percentage change
[number of patients]
Focal and segmental
glomerulosclerosis
-13.3% [N=34]
Hemolytic uremic syndrome -1.3% [N=27]
Other glomerular -15.5% [N=51]
Obstructive uropathy -4.6% [N=109]
Aplastic/hypoplastic/dysplastic
kidneys
-3.3% [N=96]
Reflux nephropathy -3.8% [N=82]
Autosomal recessive polycystic
kidney disease
-4.4% [N=18]
Other non-glomerular -2.5% [N=119]
Abbreviation: GFR, glomerular filtration rate.
Data from Furth et al.
72
Kidney International Supplements (2013) 3, 19–62 35
chapter 1

0.54
±
3.67 versus 3.61
±
5.47 (P o0.0001). This
translated to higher rates of kidney survival over 5 years in
the lower proteinuria groups, 96.7% and 94.1% versus
44.9%, (P o0.01). Multivariate analysis confirmed that the
baseline PCR correlated with a more rapid decline in CrCl for
any given level of baseline function.
In a prospective mu lticenter randomized trial of protein
intake on rates of progression in children aged 2-18 years of
age, Wingen et al. employed the Schwartz equation to
estimate CrCl and demonstrated that baseline proteinuria in
multivariate analysis was the most important independent
predictor of change in CrCl. The authors reported a partial
R
2
of 0.259 at 2 years follow-up and similar results were
found after the study was exte nded for a third year.
73
Life-
table analysis in this study also suggested a cutoff value of
50 mg/kg/day of proteinuria as a strong predictor of time to a
decline in CrCl410 ml/min/1.73 m
2
and found a risk ratio of
4.01 (95% CI 2.23–7.25; Po0.001).
Finally Wong et al.
76
used cross sectional data from the
prospective longitudinal CKiD trial to demonstrate that even
after controlling for age, race, BMI, cause of CKD and use of
RAAS antagonists they could expect an average decline in
measured GFR of 10% for every increase in urinary PCR of
14% (95% CI 10-18%).
Other risk factors and comorbid conditions. Many other
risk factors and comorbid conditions have also been
associated with greater risk of progression of CKD in adults
but only a few of these have been convincingly proven in
children due to lack of pediatric prospective trials.
Hypertension is by far the best studied of these risk factors
in children, with clear evidence from multiple sources to
document the value of aggressive BP control on slowing the
rate of progression of CKD. Wingen et al.
73
demonstrated the
importance of systolic BP in rate of progression in both
univariate and multivariate models. In this study Cox
proportional hazards analysis demonstrated a systolic BP
4120 mm Hg was an independent risk for decline in CrCl by
410 ml/min/1.73 m
2
; risk ratio was 3.1 (95% CI 1.74-5.53;
Po0.001).
The most important prospective pediatric BP trial to date,
the Effect of Strict Blood Pressure Control and ACE-
Inhibition on Progression of Chronic Renal Failure in
Pediatric Patients (ESCAPE) study, used ambulatory BP
monitoring (ABPM) and a fixed dose of ramipril plus
additional antihypertensive agents that do not tar get the
RAAS to assess (as primary outcomes) the time to decline of
50% in GFR or development of ESRD. Their results
demonstrated a 35% reduction in the risk of achiev ing the
primary end point in the more intensely treated BP: HR 0.65;
95% CI 0.44-0.94; P ¼ 0.02. Further sub-analysis as reported
in the KDIGO BP Guideline
10
demonstrated that kidney
survival was 66.1% at 5 year follow-up in patients with
systolic BPo90
th
percentile for age whereas it was 41% in the
patients who did not achieve thi s level of reduction
(P ¼ 0.0002); similar numbers were seen if diastolic BP was
the metric considered.
The issue of puberty and its effect on rate of progression
has recently been addressed by the ItalKids investigators.
77
While the methodology of their analysis is less than ideal as
they did not determine actual Tanner stages in the majority of
their cohort and used estimated rather than measured GFR,
they do appear to demonstrate a decrease in kidney survival
probability beginning around 10.9 years in girls and 11.6
years in boys with CKD. Of note, the rate of decline in kidney
survival, using these age points as ‘inflection’ or break points,
is dramatically increased in both sexes based on their
evidence provided in graphical form, although more precise
analyses are not possible from the data provided.
As in adults, other factors for consideration and value in
monitoring in children with respect to risk of progression
include obesity, metabolic acidosis, anemia, calcium-phos-
phate metabolism, chronic inflammation, diabetes, hyperur-
icemia, dyslipidemia, and smoking.
The most comprehensive rev iew of many of thes e factors
in children comes from a retrospective study of the
NAPRTCS CKD database. Staples et al.
74
demonstrated that
in a multivariate analysis of nearly 4200 children registered
with CKD and GFR categories G2-G4 (GFR 15–89 ml/min/
1.73m
2
), the following factors were significantly associated
with the risk of CKD progression (defined by progression to
GFR category G5 (GFRo15 ml/min/1.73m
2
) or initiation of
dialysis or transplant): age; primar y disease; GFR category;
registration year; hypertension; corrected calcium, phos-
phorus, albumin, and hematocrit; and as proxies, the use of
medications for anemia and short stature. The ability of this
paper to prove causation or value in treating any of these
conditions in hopes of delaying CKD progression is limited
by its retrospective nature, and the fact that data were
accrued from a voluntary registry.
There is optimism that prospective data from current large
pediatric trials such as CKiD
55
and the European Cardiovas-
cular Comorbidity in Children with CKD (4C) trial
78
will
lead to a better understanding of how risk factors may be
influencing the rate of progression of CKD in children.
For Recommendation 1.3.2 the rationale and principles
behind this statement would apply to pediatrics, though the
data are not available. Insufficient evidence currently exists
with respect to the predictive value of prevalent risk factors to
guide future decisions for testing or treatment for CKD
complications in an individual child.
It is hoped that well powered, prospective trials with
adequate follow-up, such as the CKiD
55
and European 4C
78
trials, will gather sufficient numbers of patients, comorbid-
ities, and outcomes to allow for predictive models to be built
in pediatric CKD that incorporate traditional and non-
traditional cardiac risk factors including dyslipidemia and
hypertension, proteinuria (albuminuria), specific disease-
related issues (e.g., diabetes, tubulopathy), prematurity, and
birth weight.
For Recommendation 1.3.3 the rationale and principles
behind this statement would apply to pediatrics, though the
data are not available. Current evidence and a paucity of
36 Kidney International Supplements (2013) 3, 19–62
chapter 1

numbers do not allow for the statistically relevant categor-
ization of RR for CKD outcomes based solely on GFR and
albuminuria or proteinuria. Again both the CKiD
55
and
European 4C
78
trials may be able to address these short-
comings.
1.4: EVALUATION OF CKD
1.4.1: Evaluation of chronicity
1.4.1.1: In people with GFR o60 ml/min/1.73 m
2
(GFR
categories G3a-G5) or markers of kidney
damage, review past history and previous
measurements to determine duration of kidney
disease. (Not Graded)
K If duration is 43 months, CKD is con-
firmed. Follow recommendations for CKD.
K If duration is not 43 months or unclear,
CKD is
not confirmed. Patients may have
CKD or acute kidney diseases (including
AKI) or both and tests should be
repeated accordingly.
RATIONALE
When evidence of CKD is first ascertained, proof of
chronicity can be obtained or confirmed by:
(i) review of past measurements of GFR;
(ii) review of past measurements of albuminuria or
proteinuria and urine examinations;
(iii) imaging findings such as reduced kidney size and
reduction in cortical thickness;
(iv) pathological findings such as fibrosis and atrophy;
(v) medical history especially duration of disorders known
to cause CKD;
(vi) repeat measurements within and beyond the 3 month
point.
Chronicity should not be assumed as AKI can present with
similar abnormalities.
Pediatric Considerations
See Pediatric Considerations for next section.
1.4.2: Evaluation of cause
1.4.2.1: Evaluate the clinical context, including personal
and family history, social and environmental
factors, medications, physical examination,
laboratory measures, imaging, and pathologic
diagnosis to determine the causes of kidney
disease. (Not Graded)
RATIONALE
Once the presence of CKD is proven it is essential to establish
a cause for this which will inform specific management and
modify risk projections. The diagnosis will be reached by
standard clinical method (i.e., history examination) and
special investigation, based on knowledge of the common
causes of CKD and their manifestations. Not all evaluations
are required in all patients, and will be directed by clinical
context, and resource availability. For most patients the
following evaluations are indicated:
K Reagent strip urinalysis to detect hematuria or pyuria. If
positive, use urine microscopy to detect RBC casts or
WBC casts.
K Ultrasound to assess kidney structure (i.e., kidney shape,
size, symmetry and evidence of obstruction) as clinically
indicated.
K Serum and urine electrolytes to assess renal tubular
disorders, as clinically indicated.
Many individuals found to have CKD will not have a
primary kidney disease but kidney damage caused by diabetes
mellitus, vascular disease, and hypertension. The issue for the
clinician will be to decide whether the presence of these is a
sufficient explanation and if not, to investigate further. The
prevalence of other conditions will vary depending on region,
age, and other factors.
It is beyond the scope of this guideline to describe how
specific diagnoses are reached but non-nephrologists in the
first instance should review the family history, medications,
symptoms and signs for manifestations of systemic diseases.
Urinalysis should be performed, along with imaging of the
kidneys if obstruction of the urinary tract or polycystic
kidney disease is considered.
Pediatric Considerations
For Recommendations 1.4.1.1 and 1.4.2.1, the statements
would need to be altered for application in pediatric practice
in the following way.
In any child with GFRo60 ml/min/1.73 m
2
(or more
than 1 SD below expected fo r their age and sex) or with
markers of kidney damage, a complete review of their past
history and previous measurement or estimate of renal
function and full consideration of the clinical context,
including prenatal history, drug exposures of fetus or mother,
genetic conditions, coincident organ abnormalities, physical
examination, fetal and post-natal laboratory measures
including amniotic fluid, pre- and post-natal imaging
and pathologic diagnosis including those of the fetus and
placenta should be used to determine the cause(s) of kidney
disease.
As noted in Pediatric Considerations for Recommendation
1.1.1, developmental renal abnormalities account for as many
as 30-50% of the children with CKD.
42
A careful review of all
fetal or maternal exposures, genetic risks factors, and any
relevant information on the intrauterine environment durin g
gestation are all relevant to the determi nation of the presence
of CKD either prior to or present immedi ately at the time of
delivery. An infant may be born with CKD, leading to
Kidney International Supplements (2013) 3, 19–62 37
chapter 1

immediate classification within the CGA framework – up to
and including that of dialysis dependency.
1.4.3 Evaluation of GFR
This section describes the various methods by which GFR
can be estimated. We describe laboratory techniques that satisfy
the requirements for robust result reporting and we compare
the accuracy of available equations for the purpose of reporting
eGFR using a single equation where applicable. We emphasize
equations based on standardized measurements of SCr, but also
consider newly developed equations based on standardized
measurements of serum cystatin C (SCysC) because they are
being introduced into clinical practice. We encourage practi-
tioners to have a clear understanding of the value and
limitations of both filtration markers, the importance of
standardization of assays for both, and to understand that
when an accurate assessment of kidney function is required,
direct measurement should be undertaken.
1.4.3.1: We recommend using serum creatinine and
a GFR estimating equation for initial assessment.
(1A)
1.4.3.2: We suggest using additional tests (such as cystatin
C or a clearance measurement) for confirmatory
testing in specific circumstances when eGFR based
on serum creatinine is less accurate. (2B)
RATIONALE
These statements specifically address the need to ensure that
estimating equations are put into routine clinical practice,
and that clinicians understand the utility of further evalua-
tion with additional methods if required.
GFR is measured by the clearance of an exogenous or
endogenous filtration marker.
27
All clearance methods are
complex so in clinical practice, GFR is estimated from the
serum concentration of the endogenous filtration marker
creatinine. Cystatin C is an alternative endogenous filtration
marker; other filtration markers are also under evaluation.
The principles of GFR estimation are discussed in the
rationale for recommendations regarding the use of creati-
nine as a filtration marker but the concepts apply to GFR
estimation from all endogenous filtration markers. Specific
comments about GFR estimation using cystatin C are
presented separately.
For most clinical circumstances, estimating GFR from SCr
is appropriate for diagnosis, staging, and tracking the
progression of CKD. However, like all diagnostic tests,
interpretation is influenced by varying test characteristics in
selected clinical circumstances and the prior probabilit y of
disease. In particular, an isolated decreased eGFR in
otherw ise healthy individuals is more likely to be false
positive than in individuals with risk factors for kidney
disease or markers of kidney damage. Confirmation of
decreased eGFR by measurement of an alternative endogen-
ous filtration marker (cystatin C) or a clearance measurement
is warranted in specific circumstances when GFR estimates
based on SCr are thought to be inaccurate and when
decisions depend on more accurate knowledge of GFR, such
as confirming a diagnosis of CKD, determining eligibility for
kidney donation, or adjusting dosage of toxic drugs that are
excreted by the kidneys.
79
The choice of confirmatory test
depends on the clinical circumstance and the availability of
methods where the patient is treated.
Pediatric Considerations
For Recommendation 1.4.3.1, the statements would need
to be altered for application in pediatric practice in the
following way. The use of SCr and a recently derived pediatric
specific GFR estimating equation, which incorporates a
height term,
80
is preferred over the use of SCr alone in the
initial assessment of pediatric renal function.
For Recommendation 1.4.3.2, this guideline is fully
applicable in pediatrics.
1.4.3.3: We recommend that clinicians (1B):
K use a GFR estimating equation to derive
GFR from serum creatinine (eGFR
creat
)
rather than relying on the serum creatinine
concentration alone.
K understand clinical settings in which
eGFR
creat
is less accurate.
RATIONALE
Estimating GFR from the SCr concentration al one requires
implicit judgments that are difficult in routine clinical care,
including reciprocal transformation, consideration of the
non-GFR determinants, and conversion to the GFR scale.
Using GFR estimating equations provides a more direct
assessment of GFR than SCr alone. The SCr concentration is
influenced by GFR and other physiological processes,
collectively termed ‘‘non-GFR determinants,’’ including
creatinine generation by muscle and dietary intake, tubular
creatinine secretion by organic anion transporters, and
extrarenal creatinine elimination by the gastrointestinal tract
(Figure 10).
GFR estimating equations are developed using regression to
relate the measured GFR to steady state SCr concentration and
a combination of demographic and clinical variables as
surrogates of the non-GFR determinants of SCr. By definition,
GFR estimates using SCr concentration are more accurate in
estimating measured GFR than the SCr concentration alone in
the study population in which they were developed. Sources of
error in GFR estimation from SCr concentration include non-
steady state conditions, non-GFR determinants of SCr,
measurement error at higher GFR, and interferences with
the creatinine assays (Table 11). GFR estimates are less precise
at higher GFR levels than at lower levels.
The clinician should remain aware of caveats for any
estimating equation which may influence the accuracy in a
given individual patient.
38 Kidney International Supplements (2013) 3, 19–62
chapter 1

Because of the physiologic and statistical considerations in
developing GFR estimating equations, GFR estimates are less
precise at higher GFR levels than at lower levels. In principle,
equations based on multiple endogenous filtration markers
can overcome some of the imprecision of GFR estimates at
higher levels, due to cancellation of errors from non-
correlated non-GFR determinants.
Pediatric Considerations
This guideline is fully applicable in pediatrics.
1.4.3.4: We recommend that clinical laboratories should (1B):
K measure serum creatinine using a specific
assay with calibration traceable to the inter-
national standard referenc e materials and
minimal bias compared to isotope-dilution
mass spectrometry (IDMS) reference metho-
dology.
K report eGFR
creat
in addition to the serum
creatinine concentration in adults and
specify the equation used whenever reporting
eGFR
creat
.
K report eGFR
creat
in adults using the 2009
CKD-EPI creatinine equation. An alternative
creatinine-based GFR estimating equation is
acceptable if it has been shown to improve
accuracy of GFR estimates compared to the
2009 CKD-EPI creatinine equation.
When reporting serum creatinine:
K We recommend that serum creatinine con-
centration be reported and rounded to the
nearest whole number when expressed as
standard international units (lmol/l) and
rounded to the nearest 100
th
of a whole
number when expressed as conventional units
(mg/dl).
Figure 10 | Determinants of the serum level of endogenous
filtration markers. The plasma level (P) of an endogenous
filtration marker is determined by its generation (G) from cells and
diet, extrarenal elimination (E) by gut and liver, and urinary
excretion (UV) by the kidney. Urinary excretion is the sum of
filtered load (GFR X P), tubular secretion (TS), and reabsorption
(TR). In the steady state, urinary excretion equals generation and
extrarenal elimination. By substitution and rearrangement, GFR
can be expressed as the ratio of the non-GFR determinants (G, TS,
TR, and E) to the plasma level. GFR, glomerular filtration rate.
Reprinted with permission of American Society of Nephrology,
Measured GFR as a confirmatory test for estimated GFR, Stevens
LA, Levey AS.
79
J Am Soc Nephrol 20: 2305-2313, 2009; permission
conveyed through Copyright Clearance Center, Inc.; accessed
http://jasn.asnjournals.org/content/20/11/2305.full.pdf
Table 11 | Sources of error in GFR estimating using creatinine
Source of error Example
Non-steady state
K AKI
Non-GFR determinants of SCr that differ from study populations
in which equations were developed
Factors affecting creatinine generation
K Race/ethnicity other than US and European black and white
K Extremes of muscle mass
K Extremes of body size
K Diet and nutritional status
K high protein diet
K creatine supplements
K Muscle wasting diseases
K Ingestion of cooked meat
Factors affecting tubular secretion of creatinine
K Decrease by drug-induced inhibition
K trimethoprim
K cimetidine
K fenofibrate
Factors affecting extra-renal elimination of creatinine
K Dialysis
K Decrease by inhibition of gut creatininase by antibiotics
K Increased by large volume losses of extracellular fluid
Higher GFR Higher biological variability in non-GFR determinants relative to GFR
K Higher measurement error in SCr and GFR
Interference with creatinine assay K Spectral interferences (e.g., bilirubin, some drugs)
K Chemical interferences (e.g., glucose, ketones, bilirubin, some drugs)
Abbreviations: AKI, acute kidney injury; GFR, glomerular filtration rate; SCr, serum creatinine.
Kidney International Supplements (2013) 3, 19–62
39
chapter 1

When reporting eGFR
creat
:
K We recommend that eGFR
creat
should be
reported and rounded to the nearest whole
number and relative to a body surface area of
1.73 m
2
in adults using the units ml/min/
1.73 m
2
.
K We recommend eGFR
creat
levels less than
60 ml/min/1. 73 m
2
should be reported as
‘ ‘decreased.’ ’
RATIONALE
The statement is worded this way to acknowledge that
calibration of assays is essential to interpretation of kidney
function measures. This recommendation is directed to
laboratories with the intent to clarify the details of such
calibration and the use of specific equations so as to facilitate
international standardization.
81
There are numerous assay methods for creatinine for use
in clinical laboratories. Variation in assigned values for SCr
concentration among methods is greater at low concentra-
tions, corresponding to high levels of GFR. Variation in
assays at low SCr concentrations contributes to imprecision
of GFR estimates at high GFR levels.
Currently available assays fall into two broad categories, the
alkaline picrate (Jaffe) assay and enzymatic assays. In general,
enzymatic assays are less biased compared to a standardized
refer ence material and less susceptible to interferenc es. All assays
are available on a number of platforms.
We recommend that laboratories use assays that are
traceable to pure creatinine standards via a valid calibration
hierarchy and that are specific and minimally-biased
compared with isotope-dilution mass spectrometry (IDMS)
reference method results. Results should be traceable to
reference materials and methods listed on the Joint
Committee for Traceability in Laboratory Medicine (JCTLM)
database. Ideally laboratories should move to enzymatic
assays for creatinine measurement: as a minimum, the use of
traditional kin etic or end point Jaffe assays should cease and
be replaced with IDMS aligned Jaffe methods.
Clinical laboratory information systems gen erally have
access to patient age and sex and thus can report eGFR based
on SCr age and sex, thus providing the clinician with the test
result in units which are recommended for interpretation.
Estimated GFR is now reported together with SCr when
creatinine is ordered in more than 75% of clinical
laboratories in the US.
82
In the UK, 93% of NHS laboratories
report eGFR with SCr,
83
as is the case in Australia, Canada,
and many European countries.
Selection of a single equation for use, where applicable,
would facilitate communication among providers, patients,
researchers and public health officials. Criteri a for selection
should be based on accuracy compared to measured GFR and
usefulness in clinical care and public health.
The interpretation of measured and eGFR is based on
comparison to normative values, which are adjusted for BSA
because of the physiologic matching of GFR to kidney size,
which is in turn related to BSA. The value of 1.73 m
2
reflects
the average value of BSA of 25-year old men and women in
the USA in 1927.
84
While it is known that modern
populations may have different normal values for BSA, the
1.73 m
2
value will be maintained for normalization purposes.
Drug dosing should be based on GFR which is not
adjusted for BSA. The effect of drug dosing based on GFR
adjusted for BSA compared to GFR unadjusted for BSA has
not been studied rigorously and more precise recommenda-
tions are not available.
Flagging decreased values for eGFR can alert clinicians to
the possibility of AKD or CKD, and may indicate the need for
additional investigations or treatments, including adjustment
of doses of drugs that are excreted by the kidney. However,
values for GFR between 60 and 89 ml/min/1.73 m
2
are mildly
decreased compared to the usual values in young healthy
people. Thus it is important that clinicians appreciate that
eGFR values that are not flagged because they are 460 ml/
min/1.73 m
2
are not necessarily normal.
Evidence Base
Numerous equations have been developed to estimate GFR
or CrCl in adults. In general, GFR estimating equations using
creatinine include age, sex, race, and body size as surrogates
for creatinine generation by muscle. For our review of GFR
estimating equations, we only considered equations that were
developing using assays that were traceable to reference
methods and study populations in which SCr concentration
was measured using traceable assays (Supplemental
Table 1).
85
Based on published data, only the Modification of Diet in
Renal Disease (MDRD) Study equation, Chronic Kidney
Disease Epidemiology Collaboration (CKD-EPI) equation
and modifications of these equations were developed using
creatinine assays traceable to the international reference
material for creatinine (Table 12).
86,87
The Cockcroft and
Gault formula and others were developed before standar-
dization of creatinine assays but cannot be re-expressed
for use with standardized creatinine assays (Supplemental
Table 2).
The MDRD Study equation was developed in 1999 and is
currently recommended for eGFR reporting in adults by the
National Kidney Disease Education Program (NKDEP) and
by the Department of Health in the UK. It uses standardized
SCr, age, sex, and race (black versus white and other) to
estimate GFR adjusted for BSA (ml/min/1.73 m
2
).
86,94
Because of imprecision at higher GFR, NKDEP recommends
that eGFR Z60 ml/min/1.73 m
2
computed using the MDRD
Study equation not be reported as a numeric value. For a
similar reason, the UK Department of Health recommends
not reporting eGFR 490 ml/min/1.73 m
2
using the MDRD
Study equation as a numeric value.
The CKD-EPI equation was developed in 2009 and uses
the same four variables as the MDRD Study equation.
87
The
CKD-EPI equation had l ess bias than the MDRD Study
equation, especially at GFRZ60 ml/min/1.73 m
2
, a small
40 Kidney International Supplements (2013) 3, 19–62
chapter 1

Table 12 | Equations based on serum creatinine assays in adults that are traceable to the standard reference material
Study Equation name Equation
Development and internal
validation population, N GFR measurement method SCr assay method
North America, Europe, and Australia
Levey et al.
86
MDRD equation 175 SCr
1.154
age
0.203
0.742 (if female)
1.212 (if black)
1628 patients enrolled in the
MDRD Study (mean age, 50.6 y)
125
I-Iothalamate (urine); GFR
measured in ml/min per 1.73 m
2
;
mean GFR, 39.8 ml/min per 1.73 m
2
(SD, 21.2)
Samples from MDRD Study were assayed
from 1988 to 1994 with the Beckman
Synchron CX3 kinetic Jaffe assay (Global
Medical Instrumentation, Inc., Ramsey,
Minnesota). Samples were reassayed in
2004 with the same instrument. The
Beckman assay (Global Medical
Instrumentation, Inc., Ramsey, Minnesota)
was calibrated to the Roche enzymatic
assay (Roche Diagnostics, Basel,
Switzerland), which is traceable to an IDMS
assay at NIST.
MDRD equation without ethnicity
factor*
175 SCr
1.154
age
0.203
0.742 (if female)
Levey et al.
87
CKD-EPI equation 141 min(SCr/k,1)
a
max(SCr/k,
1)
1.209
0.993
age
1.018
(if female) 1.159 (if black),
where k is 0.7 for females and 0.9 for
males, a is 0.329 for females and 0.411
for males, min indicates the minimum of
SCr/k or 1, and max indicates the
maximum of SCr/k or 1.
8254 participants from 6 research
studies and 4 clinical populations
(mean age, 47 y)
125
I-Iothalamate (urine); GFR
measured in ml/min per 1.73 m
2
;
mean GFR, 68 ml/min per 1.73 m
2
(SD, 40)
SCr values were recalibrated to
standardized SCr measurements at the
Cleveland Clinic by using a Roche enzymatic
assay (Roche–Hitachi P-Module instrument
with Roche Creatininase Plus assay,
Hoffman-La Roche, Basel, Switzerland).
Outside of North America, Europe, and Australia
Horio et al.
88
MDRD equation with Japanese
coefficient
0.808 175 SCr
1.154
age
0.203
0.742 (if female)
413 Japanese patients in 80
medical centers (mean age, 51.4 y)
Inulin (urine) GFR measured in ml/
min per 1.73 m
2
(ND for
development or internal validation
set)
SCr levels were measured by using a Hitachi
enzymatic creatinine assay and the values
obtained were compared with those of the
Cleveland Clinic.
Imai et al.
89
Japanese-modified MDRD
equation
0.741 175 SCr
1.154
age
0.203
0.742 (if female)
248 inpatients with CKD for
estimating and determining the
coefficient of the MDRD Study
equation for Japanese patients
(mean age, 50.1 y)
Inulin (urine) GFR measured in ml/
min per 1.73 m
2
(ND for
development or internal validation
set)
SCr levels were assayed by both enzymatic
and noncompensated kinetic Jaffe methods
for the 116 patients in the inulin clinical
study and by an enzymatic method for the
132 inpatients at Tokyo Women’s Medical
University between 2003 and 2004 and the
168 patients with CKD at the University of
Tsukuba Hospital from 1988 to 1994. The
SCr levels of the samples from the 101
patients from Tokyo Women’s Medical
University between 2001 and 2002 were
measured by a noncompensated kinetic
Jaffe method. SCr levels used in the inulin
clinical trial were simultaneously measured
by both the noncompensated kinetic Jaffe
method and enzymatic method at the
International Organization for
Standardization–approved central
laboratory. SCr values from the
noncompensated Jaffe method were 0.207
higher than those from the enzymatic
method throughout the measured range,
based on calibrated standards which were
indirectly calibrated with IDMS.
Kidney International Supplements (2013) 3, 19–62 41
chapter 1

Table 12 | Continued
Study Equation name Equation
Development and internal
validation population, N GFR measurement method SCr assay method
Praditpornsilpa et al.
90
MDRD equation with Thai
coefficient
175 SCr
1.154
age
0.203
0.742
(if female) 1.129 (if Thai)
250 cases of Thai patients with CKD
patients who were in stable
condition (mean age, 59.5 y)
99m
Tc-DTPA (plasma); GFR
measured in ml/min per 1.73 m
2
(ND for development or internal
validation set)
Fasting SCr levels measured by using a
Roche enzymatic assay (Roche Diagnostics,
Indianapolis, IN) and values adjusted by
using IDMS reference SCr (SRM 967) from
NIST. SCr levels also measured by a Roche
kinetic Jaffe assay Roche (Roche
Diagnostics, Indianapolis, IN) without
adjustments to IDMS reference. SCr levels
obtained from enzymatic and Jaffe assays
were used in each estimated GFR equation
accordingly.
Horio et al.
88
CKD-EPI equation with Japanese
coefficient
0.813 141 min(SCr/k,1)
a
max(SCr/k,1)
1.209
0.993
age
1.018 (if female) 1.159 (if black),
where k is 0.7 for females and 0.9 for
males, a is 0.329 for females and 0.411
for males, min indicates the minimum
of SCr/k or 1, and max indicates the
maximum of SCr/k or 1.
413 Japanese patients in 80
medical centers (mean age, 51.4 y)
Inulin (urine) GFR measured in
ml/min per 1.73 m
2
(ND for
development or internal validation
set)
SCr levels measured by Hitachi enzymatic
assay and values obtained compared with
those of the Cleveland Clinic.
Matsuo et al.
91
JSN-CKDI equation (equation 2) 171 SCr
1.004
age
0.287
0.782 (if female)
413 Japanese patients in 80
medical centers; data collected
from 1 December 2006 to 20 April
2007 (mean age, 51.4 y)
Inulin (urine)
Mean GFR, 59.1 ml/min per
1.73 m
2
(SD, 35.4)
SCr levels measured by Hitachi enzymatic
assay (Hitachi, Tokyo, Japan) and values
obtained compared with those of the
Cleveland Clinic.
3-variable Japanese equation
(equation 4)
194 SCr
1.094
age
0.287
0.739 (if female)
Levey et al.
92
Original MDRD equation, calibrated
to the Cleveland Clinic creatinine
measurements (study equation 2)
w
186 SCr
1.154
age
0.203
0.742 (if female)
1628 patients whose GFR was
measured as part of the MDRD
Study (mean age, 50.6 y)
125
I-Iothalamate (urine)
GFR measured in ml/min per
1.73 m
2
; mean GFR, 39.8 ml/min
per 1.73 m
2
(SD, 21.2)
Serum and urine creatinine levels measured
by using kinetic Jaffe assay.
Ma et al.
93
Original MDRD equation, calibrated
to Cleveland Clinic creatinine
measurements, with Chinese
coefficient (study equation 4)
w
186 SCr
1.154
age
0.203
0.742 (if
female) 1.227 (if Chinese), with SCr calibrated
to the Cleveland Clinic
454 patients from 9 renal institutes
of university hospitals located in 9
geographic regions of China (mean
age, 49.9 y)
99m
Tc-DTPA (plasma) GFR
measured in ml/min per 1.73 m
2
(ND for development or internal
validation set)
SCr levels measured by using Hitachi kinetic
Jaffe assay and values calibrated to the
Cleveland Clinic Laboratory.
Chinese equation (study
equation 6)
z
206 SCr
1.234
age
0.227
0.803 (if female),
with SCr calibrated to the Cleveland Clinic
Praditpornsilpa et al.
90
Thai estimated GFR equation 375.5 SCr
0.848
age
0.364
0.712 (if female);
r
2
= 0.869
250 Thai patients with CKD who
were in stable condition (mean
age, 59.5 y)
99m
Tc-DTPA (plasma) GFR
measured in ml/min per 1.73 m
2
(ND for development or internal
validation set)
Fasting SCr levels measured by using a
Roche enzymatic assay (Roche Diagnostics,
Indianapolis, IN) and values adjusted by
using IDMS reference SCr (SRM 967) from
NIST. SCr also measured by a Roche kinetic
Jaffe assay (Roche Diagnostics, Indianapolis,
IN) without adjustments to IDMS reference.
SCr levels obtained from enzymatic and
Jaffe assays used in each estimated GFR
equation accordingly.
Abbreviations: CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; GFR, glomerular filtration rate; IDMS, isotope-dilution mass spectrometry; JSN-CKDI, Japanese Society of Nephrology-
Chronic Kidney Disease Initiatives; MDRD, Modification of Diet in Renal Disease; ND, not documented; NIST, National Institute of Standards and Technology; SCr, serum creatinine; SRM, standard reference material; Tc-DTPA,
technetium-diethylenetriamine pentaacetic acid.
*African American coefficient of the MDRD Study equation.
wDerived by using the original MDRD equation (with the 186 coefficient). For use with an SCr traceable to the SRM, the 186 coefficient should be replaced with the 175 coefficient from the reexpressed MDRD equation.
zDerived by using the original MDRD equation (with the 186 coefficient). For use with an SCr traceable to the SRM, the SCr should be replaced by SCr 0.95, which represents the calibration factor relating the SCr assay in the
Cleveland Clinic laboratory to the standardized SCr assay.
Reprinted with permission from Earley A, Miskulin D, Lamb EJ, et al.
85
Estimating equations for glomerular filtration rate in the era of creatinine standardization: a systematic review. Ann Intern Med 2012; 156(11): 785-795.
42 Kidney International Supplements (2013) 3, 19–62
chapter 1

improvement in precision, and greater accuracy (Figure 11).
Most but not all studies from North America, Europe and
Australia show that the CKD-EPI equation is more accurate
than the MDRD Study equation, especially at higher GFR
(Table 13),
85
which enables reporting of numeric values
across the range of GFR. At this time, large commercial
clinical laboratories in the US have changed from using the
MDRD Study equation to the CKD-EPI equation for eGFR
reporting.
Lesser bias of the CKD-EPI equation compared to the
MDRD Study equation reflects higher eGFR throughout
most of the range for age and creatinine, especially in
younger individuals, women and whites. Higher eGFR results
in lower prevalence estimates for CKD in these groups
(Figure 12), with more accurate risk relationships of lower
eGFR and adverse outcomes (Figure 13).
107
To account for possible differences in muscle mass
and diet according to race, ethnicity and geographic region,
the MDRD Study and CKD-EPI equations have been
modified for use in other racial and ethnic groups and
in other countries. In some, but not all studies, these
modifications are associated with increased accuracy
(Table 14), and should be used in preference to unmodified
equations. Where tested, the CKD-EPI equation and its
modifications were generally more accurate than the MDRD
Study and its modifications. In the abs ence of specific
modifications for race, ethnicity, or regional difference, it is
reasonable to use the CKD-EPI equation for GFR estimation.
Reliance upon SCr alone is not an appropriate alternative
since the uncertainty about the effect of non- GFR deter-
minants affects interpretation of SCr as much as it affects
interpretation of eGFR. More widespread testing of GFR
estimating equations is necessary to resolve uncertainties
about the need for racial, ethnic, and geographic modi-
fications.
108
Pediatric Considerations
This recommendation would need to be altered for applica-
tion in pediatric practice in the following way.
K Creatinine measurements in all infants and children
should be derived from methods that minimize con-
founders and are calibrated against an international
standard.
K eGFR
creat
may only be reported when the height of the
child is known by the laboratory.
K If reporting eGFR
creat
laboratories should utilize the
most current and accurate pediatric derived equations
based on the demographic and laboratory markers
available.
In infants or small children the level of creatinine when
measured is often below that of the normal ‘bott om range’ of
the adult assay. As such laboratories measuring creatinine in
infants or small children must ensure their lower calibration
samples include the lowest end of the expected range of
values for the group of interest.
As the majority and the most accurate of the published
pediatric eGFR
creat
formulas require height, standard labora-
tory reporting of eGFR
creat
is neither practical nor recom-
mended in children. In a pediatric CKD population, and
using the plasma disappearance of iohexol as the gold
standard measure of GFR, Schwartz et al. derived a number
of novel GFR prediction equations.
80
Their analysis demon-
strated the importan ce of the height/SCr variable in the
population as it provided the best correlation with the
iohexol GFR (R
2
¼ 65%). The simplest of such formula, using
only height and SCr and a constant of either 41.3 or 0.413
depending on whether height was expressed as meters or
centimeters respectively, provided 79% of estimated GFRs
within 30% of the iohexol values and 37% of estimated GFRs
within 10% of the iohexol values.
Figure 11 | Performance of the CKD-EPI and MDRD Study equations in estimating measured GFR in the external validation data set.
Both panels show the difference between measured and estimated versus estimated GFR. A smoothed regression line is shown with
the 95% CI (computed by using the lowest smoothing function in R), using quantile regression, excluding the lowest and highest
2.5% of estimated GFR. To convert GFR from ml/min per 1.73 m
2
to ml/s per m
2
, multiply by 0.0167. CKI-EPD, Chronic Kidney Disease
Epidemiology Collaboration; CI, confidence interval; GFR, glomerular filtration rate; MDRD, Modification of Diet in Renal Disease. Reprinted
with permission from Levey AS, Stevens LA, Schmid CH, et al.
87
A new equation to estimate glomerular filtration rate. Ann Intern Med 2009;
150(9): 604-612.
Kidney International Supplements (2013) 3, 19–62 43
chapter 1

Table 13 | Performance comparison of creatinine-based GFR estimating equations in North America, Europe, and Australia
Measured GFR GFR estimation Results
Study Country Population Patients, N Reference standard
Value (SD), ml/min per
1.73 m
2
SCr calibration and assay Equation
Bias (95% CI),
ml/min per
1.73 m
2
*
Precision
(95% CI)
w
P
30
(95% CI),
%
z
Murata et al.
95
United States Adults who underwent an outpatient
iothalamate clearance at the Mayo Clinic in
Rochester, Minnesota (women, 45%; African
American, 2%; mean age, 56 y; donor or
potential donor, 13%; KTR, 26%)
5238
125
I-Iothalamate (urine) 55.9 (29.7) Roche Jaffe assay (Roche P- or D-
Modular or Roche Cobas C501 with
Roche Creatininase Plus assay; Roche
Diagnostics, Indianapolis, IN) with
demonstrated IDMS alignment
MDRD
CKD-EPI
4.1
0.7
ND 77.6
78.4
Levey et al.
87
United States External validation set comprising 16 studies
(women, 45%; white or other, 87%; black, 10%;
Hispanic, 2%; Asian, 2%; mean age, 50 y;
diabetes, 28%; donor, 16%; KTRs, 29%)
3896
125
I-Iothalamate
(urine) and others
68 (36) Roche enzymatic assay (Roche–Hitachi
P-Module instrument with Roche
Creatininase Plus assay, Hoffman-La
Roche, Basel, Switzerland) recalibrated
to standardized SCr at the Cleveland
Clinic
MDRD
CKD-EPI
5.5 (5.0 to
5.9)
2.5 (2.1 to
2.9)
0.274 (0.265
to 0.283)
y
0.250 (0.241
to 0.259)
y
80.6 (79.5 to
82.0)
84.1 (83.0 to
85.3)
Lane et al.
96
United States Patients before and after nephrectomy for
causes other than donation at the Cleveland
Clinic (women, 93%; white, 91%; black, 7%;
median age, 58 y)
425
125
I-Iothalamate (urine) 50 (IQR, 29 to 69) Measured at the Cleveland Clinic;
assay standardized against NIST
MDRD
CKD-EPI
1.0
1.7
15.0
||
13.8
||
75
80
Michels
et al.
97
The Netherlands Potential kidney donors and adults who
underwent GFR measurement for clinical
reasons at the Academic Medical Center in
Amsterdam (women, 56%; black, 12%; mean
age, 44 y)
271
125
I-Iothalamate (urine) 78.2 ml/min (33.4) Hitachi enzymatic assay (Hitachi H911;
Boehringer Mannheim, Mannheim,
Germany) validated against IDMS
MDRD
CKD-EPI
14.6 ml/min
12.3 ml/min
19.9
z
12.1
z
81.2
84.5
Tent et al.
98
The Netherlands Living kidney donors who donated from
1996–2007 (women, 57%; white, 100%; mean
age, 50 y)
253 before
donation
125
I-Iothalamate (urine) 115 ml/min (20) Roche enzymatic assay or Jaffe assay
on MEGA analyzer (Merck KGaA,
Darmstadt, Germany); both methods
calibrated to reference standard at
the Cleveland Clinic
MDRD
CKD-EPI
22 ml/min
(20 to 25)
14 ml/min
(11 to 16)
20 (14 to 26)
||
18 (14 to 22)
||
73 (68 to 79)
89 (85 to 93)
253 after
donation
73 ml/min (13) MDRD
CKD-EPI
15 ml/min
(14 to 16)
11 ml/min
(9 to 11)
12 (9 to 15)
||
12 (10 to 16)
||
71 (65 to 76)
89 (85 to 93)
Kukla et al.
99
United States KTRs receiving immunosuppression (women,
40%; white, 86%; mean age, 49 y; receiving
trimethoprim, 100%)
107 on
steroid-free
immunosuppression
early
posttransplantation
125
I-Iothalamate (urine) 55.5 (17.0) Measured by using a Jaffe CXR
Synchron method and, later, an IDMS-
traceable assay; Jaffe assay-based SCr
converted to IDMS-traceable values
MDRD
CKD-EPI
8.23
13.30
17.9
y
21.1
y
71.7
58.5
81 on steroid-free
immunosuppression at
1y
56.8 (17.7) MDRD
CKD-EPI
2.40
6.91
15.8
y
17.3
y
75.0
66.7
White et al.
100
Canada Stable KTRs (women, 36%; white, 92%;
receiving trimethoprim, 19%)
207
99m
Tc-DTPA (plasma) 58 ( 22) For the reexpressed MDRD Study and
CKD-EPI equations, SCr adjusted to
IDMS standard
MDRD
CKD-EPI
7.4
5.2
14.4
||
15.7
||
79 (73 to 84)
84 (78 to 88)
Po
¨
ge et al.
101
Germany Patients with stable renal function after
transplantation (women, 40%; white, 99%;
mean age, 49 y)
170
99m
Tc-DTPA (plasma) 39.6 (IQR, 11.8 to 82.9) Jaffe assay on Dimension RxLTM
analyzer (Dade Behring, Marburg,
Germany); assay adjusted for
calibration with the IDMS method
MDRD
CKD-EPI
4.49
8.07
10.0
z
10.9
z
71.8
64.1
Jones and
Imam
102
and
Jones
103
Australia Australian patients referred for routine GFR
measurements (women, 43%; mean age, 61 y)
169
99m
Tc-DTPA (plasma) 75 (IQR, 5 to 150) Roche Jaffe assay (Roche, Australia)
with demonstrated IDMS alignment
MDRD
CKD-EPI
3**
1.5**
ND 81
86
44 Kidney International Supplements (2013) 3, 19–62
chapter 1

Table 13 Continued
Measured GFR GFR estimation Results
Study Country Population Patients, N Reference standard
Value (SD), ml/min per
1.73 m
2
SCr calibration and assay Equation
Bias (95% CI),
ml/min per
1.73 m
2
*
Precision
(95% CI)
w
P
30
(95% CI),
%
z
Cirillo et al.
104
Italy White adults with and without kidney disease
(with kidney disease, 49%; women, 41%; mean
age, 47 y; diabetic nephropathy, 26 cases;
glomerulonephritis, 40 cases; PKD, 15 cases)
356 Inulin (plasma) 71.5 (36.3) Kinetic Jaffe assay (Bayer Express Plus;
Siemens, Munich, Germany)
standardized to NIST
MDRD
CKD-EPI
5.2
0.9
14.9
z
13.2
z
87.4
88.2
Eriksen
et al.
105
Norway Patients participating in the 6th Tromsø
population survey who had no previous
myocardial infarction, angina, stroke, diabetes,
or renal disease (women, 51%; mean age, 57 y)
1621 Iohexol (plasma) 91.7 (14.4) Hitachi enzymatic method (CREA Plus;
Roche Diagnostics, Mannheim,
Germany) standardized against IDMS
MDRD
CKD-EPI
1.3 (0.4 to 2.1)
2.9 (2.2 to 3.5)
18.2 (17.2 to
19.5)
||
15.4 (14.5 to
16.3)
||
93 (91 to 94)
95 (94 to 96)
Redal-Baigorri
et al.
106
Denmark Patients with cancer who were referred for
determination of GFR before chemotherapy
(women, 57%; mean age, 62 y)
185
51
Cr-EDTA (plasma) 85.1 (20.3) Jaffe (Abbott Architect C systems
8000, reagent 7D64; Abbott Park,
Illinois) method standardized to IDMS
MDRD
CKD-EPI
0.81 (IQR
1.56 to 3.19)
1.16 IQR
(0.76 to
3.09)
16.49
z
13.37
z
88.6
89.7
Abbreviations: CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; CI, confidence interval; Cr-EDTA, chromium-ethylenediamine tetraacetic acid; GFR, glomerular filtration rate; IDMS, isotope-dilution mass spectrometry;
IQR, interquartile range; KTR, kidney transplant recipient; MDRD, Modification of Diet in Renal Disease; ND, not documented; NIST, National Institute of Standards and Technology; P
30
, percentage of estimated GFR values within
30% of measured GFR; PKD, polycystic kidney disease; SCr, serum creatinine; Tc-DTPA, technetium-diethylenetriamine pentaacetic acid.
*Computed as estimated GFR minus measured GFR. Positive numbers indicate overestimation and negative numbers indicate underestimation of measured GFR. Smaller absolute values indicate lesser bias.
wLower values indicate greater precision.
zHigher values indicate greater accuracy. Among the 3 studies
99,101,104
that reported alternative measures of accuracy, results were consistent with P
30
in all. In addition to P
30
, three references
99,101,104
reported P
10
; Cirillo et al.
104
also reported P
20
.
y
Evaluated as the root mean squared error for the regression of estimated GFR on measured GFR.
||
Evaluated as the IQR for the differences between estimated and measured GFR.
z
Evaluated as the SD of the differences between estimated and measured GFR.
**Converted to raw scale by multiplying percentage of bias by measured GFR.
Reprinted with permission from Earley A, Miskulin D, Lamb EJ, et al.
85
Estimating equations for glomerular filtration rate in the era of creatinine standardization: a systematic review. Ann Intern Med 2012; 156(11): 785-795.
Kidney International Supplements (2013) 3, 19–62 45
chapter 1

Any eGFR
creat
formula used in children will preferably be
validated at the appropriate age and level of renal function,
and the labora tory methods used locally will be calibrated or
comparable to those used in the process of developing the
formula being applied. Currently the most robust pediatric
eGFR formulas, derived using iohexol disappearance and
creatinine measurements which were measured centrally and
calibrated and traceable to international standards come
from the CKiDs study.
80
The two most common creatinine-based formulas
recommended for use in clinical practice include:
Updated ‘‘Bedside’’ Schwartz equation:
eGFR (ml/min/1.73 m
2
) ¼ 41.3 (height/SCr), where height
is in meters and SCr is in mg/dl.
‘‘1B’’ Equation (incl ude blood urea nitrogen [BUN] not
cystatin C):
eGFR ( ml/min/1.73 m
2
) ¼ 40.7 (height/SCr)
0.64
(30/
BUN)
0.202
, where height is in meters, SCr and BUN are in
mg/dl.
The additional recommendation for laboratory reporting
of SCr is fully applicable in pediatrics.
When the individual clinician has information regarding
current and accurate height and applies the appropriate
pediatric formula, the recommendation to report an
individual child’s eGFR
creat
value of less than 60 ml/min/
1.73 m
2
as ‘‘decreased,’’ would be applicable in children over
the age of 2 years.
1.4.3.5: We suggest measuring cystatin C in adults with
eGFR
creat
45-59 ml/min/1.73m
2
who do not have
markers of kidney damage if confirmation of CKD
is required. (2C)
K If eGFR
cys
/eGFR
creat-cys
is also o60 ml/min/
1.73 m
2
, the diagnosis of CKD is confirmed.
K If eGFR
cys
/eGFR
creat-cys
is Z60 ml/min/1.73 m
2
,
the diagnosis of CKD is not confirmed.
RATIONALE
A major foundation of this guideline is that CKD classifica-
tion and staging should be influenced primarily by clinical
prognosis. As will be reviewed in the sections below,
abundant evidence has shown that GFR estimates based on
cystatin C are more powerful predictors of clinical outcomes
than creatinine-based eGFR. These findings have been
strongest for mortality and CVD events, and the prognostic
advantage of cystatin C is most apparent among individuals
with GFR 445 ml/min/1.73 m
2
. In addition, new finding s
show that using cystatin C in addition to SCr can lead to
improved accuracy of GFR estimation, including CKD
classification. In the opinion of the Work Group, these
considerations warrant new recommendations for GFR
estimation using cystatin C.
Evidence Base
Evidence supports the use of cystatin C-based eGFR within
the population of persons diagnosed with CKD based on an
eGFR
creat
45-59 ml/min/1.73 m
2
(G3a) but without albumin-
uria (A1) or other manifestations of kidney damage. This
group represents 3.6% of the US population and 41% of
people in the US estimated to have CKD based on eGFR
creat
and urine ACR alone (Figure 8), and there has been
substantial controversy over whether or not these persons
have CKD. Data described below indicate that use of cystatin
C to estimate GFR in this population leads to more accurate
estimation of GFR and prediction of risk for future adverse
events.
In several studies, eGFR
cys
has been measured in
populations with and without eGFR
creat
o60 ml/min/
1.73 m
2
, and participants were separated into those with
and without eGFR
cys
o60 ml/min/1.73 m
2
(Figure 14). Those
with both eGFR
creat
and eGFR
cys
o60 ml/min/1.73 m
2
, about
two-thirds of those with eGFR
creat
o60 ml/min/1.73 m
2
, had
Figure 12 | Comparison of distribution of GFR and CKD
prevalence by age (NHANES 1999-2004). GFR was categorized
on the basis of the classification system established by the
NKF-KDOQI. Top. Distribution of estimated GFR, by 4-ml/min per
1.73 m
2
categories. Values are plotted at the midpoint. Bottom.
Prevalence of CKD, by age. CKD, chronic kidney disease;
GFR, glomerular filtration rate; NKF-KDOQI, National Kidney
Foundation-Kidney Disease Outcomes Quality Initiative; NHANES,
National Health and Nutrition Examination Survey. Reprinted with
permission from Levey AS, Stevens LA, Schmid CH, et al.
87
A new
equation to estimate glomerular filtration rate. Ann Intern Med
2009; 150(9): 604-612.
46 Kidney International Supplements (2013) 3, 19–62
chapter 1

markedly elevated risks for death, CVD, and ESRD end points
compared with persons with eGFR
creat
460 ml/min/1.73 m
2
.
The Work Group therefor e c onsiders this group
to have ‘‘confirmed CKD. ’’ In contrast, about one-third of
those with eGFR
creat
o60 ml/min/1.73 m
2
had eGFR
cys
460 ml/
min/1.73 m
2
and this group were similar in risk for adverse
outcomes as persons with eGFR
creat
460 ml/min/1.73 m
2
.
New data from CKD-EPI also showed improved accuracy
in GFR estimation using both creatinine and cystatin C
(eGFR
creat-cys
) compared to either marker alone. In the
subgroup with eGFR
creat
45-59 ml/min/1.73 m
2
, the com-
bined equation correctly reclassified 16.8% of those with
eGFR 45-59 ml/min/1.73 m
2
to measured GFR Z60 ml/min/
1.73 m
2
.
113
The consensus of the Work Group was therefore that the
large group of persons with eGFR
creat
45-59 ml/min/1.73 m
2
without markers of kidney damage, but with eGFR
cys
/
eGFR
creat-cys
Z60 ml/min/1.73 m
2
couldbeconsiderednotto
hav e CKD . The removal of the diagnosis and label of CKD may
be reassuring to patients and may help clinicians to focus their
efforts on higher risk CKD patients.
The guideline statement suggesting the use of eGFR
cys
/
eGFR
creat-cys
requires several important qualifiers. First,
clinicians may not want or need to confirm the diagnosis
of CKD in patients with eGFR
creat
45-59 ml/min/1.73 m
2
without markers of kidney damage, either because the
likelihood of CKD is high because of the presence of risk
factors for CKD or presence of complications of CKD. Second,
cystatin C is not universally available, so it may not be practical
for a clinician to request a cystatin C blood test. Third, in
certain clinical settings, the cost of measuring cystatin C (US
$1–5) may be prohibitive. For all these reasons, the guideline
statement 1.4.3.5 is stated as a suggestion.
In addition to the population described above, eGFR
cys
may be useful as a confirmatory test in situations where
either the eGFR
creat
may be inaccurate or biased, or when the
clinical scenario warrants a secondary test (Recommendation
1.4.3.2). In these clinical situations, a clearance measurement
using an exogenous filtration marker may be optimal when it
is available. The measurement of eGFR
cys
/eGFR
creat-cys
would
be a relatively low-cost, feasible alternative when GFR
measurement is not practical. The Work Group believed that
measured urinary CrCl was an inferior confirmatory test
relative to either GFR measurement or GFR estimation using
both creatinine and cystatin C.
If cystatin C testing is desired, it is very important that
clinicians understand principles of GFR estimation using cystatin
C. As with creatinine, GFR should be estimated from cystatin C
and an appropriate equation should be chosen for the specific
clinical population (Recommendation 1.4.3.6), and an assay be
chosen for measurement that is traceable to the international
standard reference material (Recommendation 1.4.3.7).
Pediatric Considerations
The utility of this specific statement to pediatrics is unclear as
the vast majority of children with significant reductions in
GFR, e.g., below 60 ml/min/1.73 m
2
, have either structural
abnormalities or findings of renal damage as evidenced by
urinary or serum abnormalities. It is very unlikely that
isolated reduction in GFR would occur as in older adults. As
such, the confirmation of CKD will be made on criteria
beyond that of GFR alone.
1.4.3.6: If cystatin C is measured, we suggest that health
professionals (2C):
K use a GFR estimating equation to derive GFR
from serum cystatin C rather than relying on the
serum cystatin C concentration alone.
K understand clinical settings in which eGFR
cys
and eGFR
creat-cys
are less accurate.
Overall
Sex
Female
Male
Diabetes
No
Yes
Hypertension
No
Yes
Age, y
<65
≥65
Race/ethnicity
White
Black
Asian
Favors
MDRD
Favors
CKD-EPI
No. of
Cohorts
23
23
23
23
23
22
23
15
5
10
20
21
–0.1 0 0.1 0.2
NRI (95% CI)
All-cause mortality
Favors
MDRD
Favors
CKD-EPI
No. of
Cohorts
18
18
17
18
18
17
18
13
4
5
15
18
–0.1 0 0.1 0.2
NRI (95% CI)
Cardiovascular mortality
Favors
MDRD
Favors
CKD-EPI
No. of
Cohorts
5
5
3
2
3
5
5
3
2
2
5
5
–0.1 0 0.1 0.2
NRI (95% CI)
End-stage renal disease
Figure 13 | Meta-analysis of NRI for all-cause mortality, CVD mortality, and ESRD. NRI summarizes the risk of clinical outcomes among
participants who are reclassified from one estimated GFR category using the MDRD Study equation to another estimated GFR category
using the CKD-EPI equation compared with those who are not reclassified. NRI greater than zero favors the CKD-EPI equation. NRI less than
zero favors the MDRD Study equation. The sizes of the data markers are proportional to the inverse of the variance of the NRIs. CKD-EPI,
Chronic Kidney Disease Epidemiology Collaboration; CVD, cardiovascular disease; ESRD, end-stage renal disease; GFR, glomerular filtration
rate; MDRD, Modification of Diet in Renal Disease; NRI, net reclassification improvements. Reprinted with permission from Matsushita K,
Mahmoodi BK, Woodward M, et al.
107
Comparison of risk prediction using the CKD-EPI equation and the MDRD Study equation for
estimated glomerular filtration rate. JAMA 2012; 307(18): 1941-1951. Copyright & (2012) American Medical Association. All rights reserved.
Kidney International Supplements (2013) 3, 19–62 47
chapter 1

Table 14. Performance comparison of creatinine-based GFR estimating equations outside of North America, Europe, and Australia
Measured GFR GFR estimation Results
Study Country Population Patients, N Reference standard
Value (SD), ml/min
per 1.73 m
2
SCr calibration and assay Equation
Bias (95% CI),
ml/min per
1.73 m
2
*
Precision
(95% CI)
w
P
30
(95% CI),
%
z
Stevens et al.
109
China, Japan, and
South Africa
Populations from 3 studies in
China, Japan, and South Africa
(women, 48%; Asian, 90%; black,
10%; mean age, 49 y; diabetes,
6%; KTRs, 0%)
99 black patients
(South Africa)
248 Asian patients
(Japan)
125
I-Iothalamate (urine)
and other filtration
markers
61 (32)
55 (35)
Calibrated to standardized SCr
measurements by using Roche
(Roche-Hitachi P-Module
instrument with Roche
Creatininase Plus assay;
Hoffmann–La Roche, Basel,
Switzerland) enzymatic assay at
the Cleveland Clinic
CKD-EPI equation with 2-level
race or ethnicity coefficient
CKD-EPI equation with 4-level
race or ethnicity coefficient
CKD-EPI equation with 2-level
race or ethnicity coefficient
12.4 (7.6 to
18.3)
12.5 (7.6 to
18.4)
17.8 (14.7 to
20.1)
0.326 (0.292
to 0.361)
||
0.327 (0.292
to 0.362)
||
0.469 (0.424
to 0.515)
||
55.6 (46.5 to
64.6)
55.6 (46.5 to
64.6)
29.4 (23.8 to
35.1)
CKD-EPI equation with 4-level
race or ethnicity coefficient
21.4 (18.2 to
23.3)
0.507 (0.463
to 0.553)
||
36.3 (30.6 to
42.3)
675 Asian patients
(China)
53 (31) CKD-EPI equation with 2-level
race or ethnicity coefficient
2.7 (3.7 to
1.9)
0.325 (0.302
to 0.348)
||
73.2 (69.9 to
76.6)
CKD-EPI equation with 4-level
race or ethnicity coefficient
1.3 (2.2 to
0.6)
0.318 (0.295
to 0.343)
||
72.1 (68.7 to
75.7)
Matsuo et al.
91
Japan Hospitalized Japanese patients;
external validation set (women,
42%; mean age, 54 y; diabetes,
44 cases; donors, 10; KTRs, 2;
glomerulonephritis, 176 cases;
PKD, 0 cases; lupus, 3 cases)
350 Inulin (urine) 57.2 (34.7) (GFR
Z60, 41%; GFR
o60, 59%)
Hitachi enzymatic assay (Hitachi,
Tokyo, Japan) with excellent
agreement against Cleveland
Clinic
MDRD
Japanese-modified MDRD
(equation 1)
JSN-CKDI equation (equation 2)
MDRD with Japanese coefficient
(equation 3)
12.0
5.9
7.9
1.3
25.2
||
19.9
||
20.3
||
19.4
||
59 (54 to 64)
72 (67 to 76)
73 (69 to 78)
73 (59 to 78)
3-variable Japanese equation
(equation 4)
2.1 19.1
||
75 (70 to 79)
Horio et al.
88
Japan Japanese patients (women, 42%;
mean age, 54 y; diabetes, 22%;
donors, 3%; KTRs, 1%;
hypertension, 58%)
350 Inulin (urine) 45 (25) Enzymatic assay validated by
using the calibration panel of the
Cleveland Clinic
MDRD with Japanese coefficient
CKD-EPI with Japanese
coefficient
1.3
0.4
19.4
||
17.8
||
73 (69 to 78)
75 (70 to 79)
Yeo et al.
110
Korea Korean KTRs in the early
postoperative period (women,
43%; mean age, 42 y; diabetes,
16%)
102
51
Cr-EDTA (plasma) 76.8 (17.0) Rate-blanked, Toshiba-
compensated kinetic Jaffe assay
(Toshiba Medical Systems Tokyo,
Japan) using a Roche calibrator
(Roche Diagnostics, Indianapolis,
Indiana) traceable to IDMS
reference method
MDRD
Japanese-modified MDRD
0.33
17.95
12.57
z
11.06
z
94.1
68.6
van Deventer
et al.
111
South Africa Black South African patients
(women, 49%; median age, 47 y;
diabetes, 25%; donors, 7%; HIV,
20%; hypertension, 36%)
100
51
Cr-EDTA (plasma) 61.5 (49.6) Rate-blanked, Roche-
compensated, kinetic Jaffe assay
(Roche Modular analyzer; Roche
Diagnostics, Indianapolis,
Indiana) with calibration
traceable to IDMS, compared
with Roche enzymatic assay
(Creatinine Plus; Roche
Diagnostics, Indianapolis,
Indiana) at the Cleveland Clinic
with values adjusted by
regression
MDRD with race or ethnicity
factor**
MDRD without race or ethnicity
factor**
13.1 (5.5 to
18.3)
1.9 (0.8 to
4.5)
28.5
||
16.6
||
52
74
48 Kidney International Supplements (2013) 3, 19–62
chapter 1

Table 14 Continued
Measured GFR GFR estimation Results
Study Country Population Patients, N Reference standard
Value (SD), ml/min
per 1.73 m
2
SCr calibration and assay Equation
Bias (95% CI),
ml/min per
1.73 m
2
*
Precision
(95% CI)
w
P
30
(95% CI),
%
z
Teo et al.
112
Singapore Outpatients with CKD in the
nephrology clinic at National
University Hospital, Singapore
(women, 48%; Chinese, 41%;
Malay, 31%; Indian or other, 28%;
median age, 58 y; diabetes, 23%;
hypertension, 50%)
232
99
Tc-DTPA (plasma) 51.7 (27.5) (GFR
Z60, 69%; GFR
o60, 31%)
Siemens enzymatic assay
(Siemens Advia 2400; Siemens,
Munich, Germany) calibrated
with manufacturer-provided
materials traceable to
standardized creatinine (NIST
SRM 967) measured by using
IDMS
MDRD
CKD-EPI
3.0 (4.2 to
1.7)
1.2 (2.7 to
0.3)
12.2 (10.0 to
14.4)
y
12.1 (9.0 to
15.1)
y
79.7 (74.6 to
84.9)
82.8 (77.9 to
87.6)
Ma et al.
93
China Patients with CKD from 9 renal
institutes at university hospitals
located in 9 geographic regions
of China
230
99
Tc-DTPA (plasma) ND Hitachi kinetic Jaffe assay with
values adjusted by regression
analysis to Cleveland Clinic
Beckman CX3 assay (Beckman
Coulter, Fullerton, California)
Original MDRD equation,
calibrated to the Cleveland Clinic
creatinine measurements (study
equation 2)
ww
7.8 (21.5
to 1.8)
ND 66.1
Original MDRD equation,
calibrated to Cleveland Clinic
creatinine measurements, with
Chinese coefficient (study
equation 4)
ww
0.9 (9.6 to
7.4)
ND 77.8
Chinese equation (study
equation 6)
zz
0.8 (9.7 to
7.4)
ND 79.6
Praditpornsilpa
et al.
90
Thailand Thai patients with CKD who were
in stable condition
100
99
Tc-DTPA (plasma) 51.1 (28.4) Roche enzymatic assay (Roche
Diagnostics, Indianapolis,
Indiana) with values adjusted to
IDMS reference serum SRM 967
MDRD
CKD-EPI
MDRD equation with Thai racial
factor correction
Thai estimated GFR equation
11.9
10.9
10.3
7.2
8.8
z
7.8
z
8.5
z
6.3
z
62.7
68.0
73.3
90.0
CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; CI, confidence interval; Cr-EDTA, chromium-ethylenediamine tetraacetic acid; GFR, glomerular filtration rate; IDMS, isotope-dilution mass
spectrometry; JSN-CKDI, Japanese Society of Nephrology-Chronic Kidney Disease Initiatives; KTR, kidney transplant recipient; MDRD, Modification of Diet in Renal Disease; ND, not documented; NIST, National Institute of Standards
and Technology; P
30
, percentage of estimated GFR values within 30% of measured GFR; PKD, polycystic kidney disease; SCr, serum creatinine; SRM, standard reference material; Tc-DTPA, technetium-diethylenetriamine pentaacetic
acid.
* Computed as estimated GFR minus measured GFR. Positive numbers indicate overestimation and negative numbers indicate underestimation of measured GFR. Smaller absolute values indicate lesser bias.
wLower values indicate greater precision.
z
Higher values indicate greater accuracy. Among the 5 studies
90,91,93,110,112
that reported alternative measures of accuracy, results were consistent with P
30
in 3. Of the studies reporting results consistent with P
30
, Praditpornsilpa
et al.
90
reported P
10
and P
15
, Yeo et al.
110
reported P
10
, and Matsuo et al.
91
reported P
15
. Two studies
93,112
reported inconsistent results between P
15
and P
30
.
y
Evaluated as the interquartile range for the differences between estimated and measured GFR.
||
Evaluated as the root mean squared error for the regression of estimated GFR on measured GFR.
z
Evaluated as the SD of the differences between estimated and measured GFR.
**African American coefficient of the MDRD Study equation.
ww
Derived by using the original MDRD equation (with the 186 coefficient). For use with an SCr traceable to the SRM, the 186 coefficient should be replaced with the 175 coefficient from the reexpressed MDRD equation.
zz
Derived by using the original MDRD equation (with the 186 coefficient). For use with an SCr traceable to the SRM, the SCr should be replaced by SCr 0.95, which represents the calibration factor relating the SCr assay in the
Cleveland Clinic laboratory to the standardized SCr assay.
Reprinted with permission from Earley A, Miskulin D, Lamb EJ, et al.
85
Estimating equations for glomerular filtration rate in the era of creatinine standardization: a systematic review. Ann Intern Med 2012; 156(11): 785-795.
Kidney International Supplements (2013) 3, 19–62 49
chapter 1

RATIONALE
Cystatin C is licensed for use in some countries in
Europe and has been approved by the FDA as a measure
of kid ney function in the United States for nearly
10 years. In certain regions, notably Sweden and parts
of China, eGFR is routinely estimated by both creatinine
and cystatin C. As with creatinine, GFR estimates using
cystatin C are more accurate in estimating measured
GFR than the SCysC concentration alone. As with crea-
tinine, sources of error in GFR estimation from SCysC
concentration include non-steady state conditions, non-
GFR determinants of SCysC, measurement error at
higher GFR, and interferences with the cystatin C assays
(Table 15).
Pediatric Considerations
For Recommendation 1.4.3.6, this guideline is fully applicable
in pediatrics. See Recommendation 1.4.3.7 for details.
In terms of clinical settings where eGFR
cys
might be less
accurate, it should be noted that Schwartz et al. determined
that the only varia ble that explained the outlier values of
estimated GFR (in both univariate and multivariate for-
mulas) was heavier weight; race, high blood pressure,
albumin levels and use of steroids did not contribute.
115
Figure 14 | Association of CKD definitions with all-cause mortality and ESRD. CKD, chronic kidney disease; ESRD, end-stage renal
disease. Reprinted with permission from Peralta CA, Shlipak MG, Judd S, et al.
114
Detection of chronic kidney disease with creatinine, cystatin
C, and urine albumin-to-creatinine ratio and association with progression to end-stage renal disease and mortality. JAMA 2011; 305(15):
1545-1552. Copyright & (2011) American Medical Association. All rights reserved.
Table 15 | Sources of error in GFR estimating using cystatin C
Source of error Example
Non-steady state
K AKI
Non-GFR determinants of SCysC that differ from study populations in which
equations were developed
Factors affecting cystatin C generation
K Race/ethnicity other than US and European black and white
K Disorders of thyroid function
K Administration of corticosteroids
K Other hypothesized factors based on epidemiologic associations
(diabetes, adiposity)
Factors affecting tubular reabosrption of cystatin C None identified
Factors affecting extra-renal elimination of cystatin C Increased by severe decrease in GFR
Higher GFR
K Higher biological variability in non-GFR determinants relative to
GFR
K Higher measurement error in SCysC and GFR
Interference with cystatin C assay
K Heterophilic antibodies
Abbreviations: AKI, acute kidney injury; GFR, glomerular filtration rate, SCysC, serum cystatin C.
50 Kidney International Supplements (2013) 3, 19–62
chapter 1

1.4.3.7: We recommend that clinical laboratories that
measure cystatin C should (1B):
K measure serum cyst atin C using an assay
w ith calibration traceable to the interna-
tional standard reference material.
K report eGFR from serum cystatin C in
addition to the serum cystatin C concentra-
tion in adu lts and specify the equation
used whenever reporting eGFR
cys
and
eGFR
creat-cys
.
K report eGFR
cys
and eGFR
creat-cys
in adults
using the 2012 CKD-EPI cystatin C and
2012 CKD-EPI creatinine-cystatin C equa-
tions, respec tively, or an alternat ive cystatin
C-based GFR estimating equations if they
have been shown to improve accuracy of
GFR estimates compared to the 2012 CKD-
EPI cystatin C and 2012 CKD-EPI creati-
nine-cystatin C equations.
When reporting serum cystatin C:
K We recommend reporting ser um cysta-
tin C concentration rounded to the
nearest 100
th
of a whole nu mber when
expressed as conventional units (mg/l).
When reporting eGFR
cys
and eGFR
creat-cys
:
K We recommend that eGFR
cys
and
eGFR
creat-cys
be reported and rounded to
thenearestwholenumberandrelativetoa
body surface area of 1.73 m
2
in adults using
the units ml/min/1.73 m
2
.
K We recommend eGFR
cys
and
eGFR
creat-cys
levels less than 60 ml/min/
1.73m
2
should be reported as ‘ ‘decreased.’’
RATIONALE
As for SCr, reporting eGFR using cystatin C in addition to
cystatin C will facilitate clinician’s use of cystatin C for GFR
estimation. It is important to acknowledge that calibration of
assays is essential to interpretation of kidney function
measures. Cystatin C is measured by a variety of immu-
noassays and, as for creatinine, there can be variation among
methods in reported SCysC concentration but reported
analytic variation appears less common than with creatinine.
In June 2010 the Institute for Reference Materials and
Measurements (IRMM) released a reference material (ERM-
DA471/IFCC) for cystatin C measurement. Reagent manu-
facturers are in the process of recalibrating their assays
against this standard which will enable standardized report-
ing of cystatin C and eGFR results. This recommendation is
directed to labora tories with the intent to clarify the details of
such calibration and the use of specific equations so as to
facilitate international standardization.
Evidence Base
Numerous equations have been developed to estimate GFR.
Some equations include cystatin C as the only variable, while
others include, age, sex, or race, but the magnitude of
coefficients for these variables are smaller than in creatinine-
based equations, presumably reflecting less contribution of
muscle to cystatin C generation than to creatinine generation.
Equations without race are a potential advantage for cystatin
C-based estimating equations in non-black, non-white
populations.
For our review of GFR estimating equations, we only
considered equations that were developed using assays that
were traceable to the new reference methods and study
populations in which SCysC concentration was measured
using traceable assays. At this time, only the equations
developed by CKD-EPI are expressed for use with standar-
dized SCysC (Table 16), including equations developed in
CKD populations in 2008
116,117
and re-expressed for use with
standardized cystatin C in 2011, and equations developed in
diverse populations in 2012.
113
Equations using assays that
are not traceable to the the reference standard are listed in
Supplemental Table 3.
The 2012 creatinine-cystatin C equation is more
accurate than equations using creatinine or cystatin C
separately (Figure 15), and more accurate than the
2008 creatinine-cystatin C equation (Table 17). The average
of the GFR computed by the equations using creatinine
and cystatin C separately is similar to the GFR computing
using the creatinine-cystatin C equations. The 2012
cystatin C equation has similar accuracy to the 2009
creatinine equation described above but does not require
use of race, and may be more accurate in non-black,
non-white populations or in clinical conditions with
variation in non-GFR determinants of SCr. We antici-
pate the development of additional equations using cystatin
C in the future and recommend that they be compared with
the CKD-EPI 2012 cystatin C and creatinine-cystatin C
equations as well as with the CKD-EPI 2009 creatinine
equation.
Pediatric Considerations
For Recommendation 1.4.3.7 this set of statements would
need to be altered for application in pediatric practice in the
following way:
K Measure SCysC using an immunonephelometrically
determined method in which the assay is calibrated and
traceable to the international standard reference material.
K Report eGFR
cys
in addition to the SCysC concentration in
children.
K Report eGFR
cys
in children specifying the specific
equation used.
Based on their recent work comparing particle-enhanced
nephelometric to turbidometric immunoassays for cystatin C
in a pediatric population with significant reduction in GFR
(median GFR B45 ml/min/1.73 m
2
), Schwartz et al. demon-
strated less bias for the nepholometric value and that its
reciprocal showed a substantially improved correlation to the
iohexol GFR (0.87 versus 0.74) when compared to that of the
Kidney International Supplements (2013) 3, 19–62 51
chapter 1

Table 16 | Equations based on IDMS traceable creatinine and IFCC traceable cystatin C assays
Equation name Equation
N, Development and internal validation
population GFR measurement method SCr and/or SCysC method Reference
Cystatin C equations
CKD-EPI cystatin C (alone)
2008
Re-expressed 2011
76.7 x (-0.105 + 1.13 x standardized SCysC)
1.19
2,980 participants from pooled individual-level
patients data from the MDRD Study, African
American Study of Kidney Disease (AASK), and
Collaborative Study Group (CSG)
125
I-Iothalamate (U) [Mean GFR 51
±
26 ml/min/
1.73 m
2
]
Units for GFR in ml/min/1.73 m
2
SCr assays were calibrated to standardized SCr
values at the Cleveland Clinic Research Laboratory
(CCRL; Cleveland, OH). Results of the calibration
procedure for the MDRD Study, AASK, and CSG were
described previously. Samples for all studies had
been frozen at -701C until 2005 to 2006, when SCysC
was measured at the CCRL by using a particle
enhanced immunonephelometric assay (N Latex
Cystatin C; Dade Behring, Deerfield, IL). Interassay
coefficients of variation for the assay were 5.05%
and 4.87% at mean concentrations of 0.97 and
1.90 mg/l (72.7 and 142.3 nmol/l), respectively.
SCr in mg/dl
SCysC in mg/l
Stevens LA, et al.
117
Inker LA, et al.
116
CKD-EPI cystatin C 2008
Re-expressed 2011
[127.7 * (-0.105 + 1.13 x standardized
SCysC)
1.17
x age
0.13
x 0.91 [if female] x 1.06 [if
black]]
CKD-EPI creatinine-cystatin C
2008
Re-expressed 2011
[177.6 * SCr
0.65
x (-0.105 + 1.13 x standardized
SCysC)
0.57
x age
0.20
x 0.82 [if female] x 1.11
[if black]]
CKD-EPI cystatin C
2012
133 x min(SCysC/0.8, 1)
0.499
x max(SCysC/0.8,
1)
1.328
x 0.996
Age
x 0.932 [if female] where
SCysC is serum cystatin C, min indicates the
minimum of SCysC/0.8 or 1, and max indicates
the maximum of SCysC/0.8 or 1
5352 participants from 13 studies
(7 RS and 6 CP)
125
I-Iothalamate (U) [Mean GFR 68 (39) ml/min/
1.73 m
2
; 51% GFR Z60 ml/min/1.73 m
2
; 49% GFR
o60 ml/min/1.73 m
2
]
Units for GFR in ml/min/1.73 m
2
All assays were performed at the Cleveland Clinic
Research Laboratory (Cleveland, OH). We calibrated
SCr assays or measured serum SCr directly by the
Roche enzymatic method (Roche-Hitachi P-Module
instrument with Roche Creatininase Plus assay,
Hoffman-La Roche, Ltd., Basel, Switzerland), that had
been confirmed to be traceable to National Institute
Standardized Technology (NIST) creatinine standard
reference material 967. We calibrated SCysC assays
or measured SCysC directly on the Siemens Dade
Behring Nephelometer. We recently reported
standardization of this assay to the recently released
certified reference materials for cystatin C from the
International Federation for Clinical Chemists (IFCC)
Working Group for the Standardization of SCysC and
the Institute for Reference Materials and
Measurements (IRMM) at the University of
Minnesota.
SCr in mg/dl
SCysC in mg/l
Inker LA, et al.
113
CKD-EPI creatinine-cystatin C
2012
135 x min(SCr/k,1)
a
x max(SCr/k,1)
0.601
x
min(SCysC/0.8, 1)
0.375
x max(SCysC/0.8,
1)
0.711
x 0.995
Age
x 0.969 [if female] x 1.08
[if black] where SCr is serum creatinine, SCysC is
serum cystatin C, k is 0.7 for females and 0.9 for
males, a is -0.248 for females and -0.207 for
males, min(SCr/k,1) indicates the minimum of
SCr/k or 1, and max(SCr/k,1) indicates the
maximum of SCr/k or 1; min(SCysC/0.8,1)
indicates the minimum of SCysC/0.8 or 1 and
max(SCysC/0.8,1) indicates the maximum of
SCysC/0.8 or 1
Abbreviations: AASK, African American Study of Kidney Disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; CP, clinical population; GFR, glomerular filtration rate; IDMS, isotope-dilution mass spectrometry;
IFCC, International Federation for Clinical Chemists; IRMM, Institute for Reference Materials and Measurements; MDRD, Modification of Diet in Renal Disease; NIST, National Institute Standardized Technology, RS, Research study;
SCr, serum creatinine; SCysC, serum cystatin C; U, urinary clearance.
52 Kidney International Supplements (2013) 3, 19–62
chapter 1

turbidometric assay.
115
This demonstrates the importance of
using an assay calibrated and traceable to the international
standard reference material.
Numerous pediatric specific and derived eGFR
cys
formulas
have been published, the most current and recent by
Schwartz
115
who derives the newest available from a
validation set from the CKiD study and compares those
results to 3 other well-recognized formulas from the
literature, namely Zapitelli et al.,
118
Filler and Lepage,
119
and Hoek et al.
120
Their results demonstrate that the newest univariate
cystatin C formula derived from the CKiD cohort has
excellent accuracy with 82.6% of eGFR
cys
within 30% of the
true measured iohexol GFR and 37.6% within 10% of the
true measured iohexol GFR. Likewise the bias of 0.3% and
correlation of 0.85 are the best of all formulas reported to
date. The formula they use to obtain these values is: 70.69
(cystatin c)
-0.931
.
Of note, their final multivariate equation, when applied to
the validation set and using height/SCr, nepholemetric
cystatin C, BUN, sex, and an adjusted height term
demonstrated the best accuracy reported in pediatric studies
to date, 91% and 45% within 30% and 10% of the true GFR,
respectively; with a bias of only 0.2 and correlation of 0.92.
1.4.3.8: We suggest measuring GFR using an exogenous
filtration marker under circumstances where more
accurate ascertainment of GFR w ill impact on
treatment decisions. (2B)
RATIONALE
In clinical practice, there may be a requirement to measure
GFR when the need for a ‘truer’ more precise value is
identified (such as for organ donation or for dosing of toxic
drugs). The intention of this statement is to recognize that
specialty centers for kidney disease, usually tertiary referral
centers, should have the capacity to measure GFR using
exogenous filtration markers as a recognized specialist
service. We recognize that this ability does not currently
constitute the defi nition of specialty kidney referral centers
and that it may be problematic, but resources to ensure
accurate measurement ought to be made available. Given that
these specific measurements require levels of rigor and
reproducibility similar to those of laboratory calibration
issues, specialist centers would be the right place to suggest
that these facilities be made available.
Evidence Base
GFR is measured as the clearance of an exogenous filtration
marker. The ‘‘gold standard’’ method is the urinary clearance
of inulin during a continuous intravenous infusion. To
simplify the procedure there are a num ber of alternative
clearance methods and alternative filtration markers, with
minor differences among them.
79
For all measurement
methods, measured GFR should be reported as described
for eGFR.
Table 18 summarizes the strengths and limitations of
clearance methods and filtration markers for clearance
measurements. Thus measured GFR may also be associated
with error, and in evaluation of GFR estimating equations,
random error in GFR measurement is a source of some of the
imprecision in GFR estimating equations.
27,121
In principle,
the magnitude of random error in GFR measurements is
likely to be smaller than errors in GFR estimation using
creatinine and cystatin C due to conditions listed in Tables 11
and 15.
International Relevance
The calculation of eGFR using these equations usually
requires computer programming and some processes for
quality monitoring. Nonetheless the statements are here to
serve as ‘best practice’ recommendations so that these can be
Figure 15 | Performance of three equations for estimating
GFR. Panel a shows the median difference between measured
and estimated GFR. The bias is similar with the equation using
creatinine alone, the equation using cystatin C alone, and the
combined creatinine–cystatin C equation. Panel b shows the
accuracy of the three equations with respect to the percentage
of estimates that were greater than 30% of the measured
GFR (1 – P
30
). Whiskers indicate 95% confidence intervals. GFR,
glomerular filtration rate; P
30
, percentage of estimated GFR values
within 30% of measured GFR. From N Engl J Med, Inker LA,
Schmid CH, Tighiouart H, et al.
113
Estimating glomerular filtration
rate from serum creatinine and cystatin C. 367: 20-29. Copyright
& 2012 Massachusetts Medical Society. Reprinted with permission
from Massachusetts Medical Society.
Kidney International Supplements (2013) 3, 19–62 53
chapter 1

Table 17 | Performance comparison of cystatin C-based estimating equations in North American and European populations
Study description mGFR GFR estimate Results
Author Population (subgroup) N Reference
standard
mGFR (SD) Serum creatinine (SCr)
calibration and assay
detail
Cystatin C
calibration and
assay detail
Equations Bias ml/min/
1.73 m
2
*
Precisionw Accuracyz
Inker LA et al.
113
Multi
RS/CP: 5 studies (2 RS and 3 CP)
using equations developed in CKD
population [3% black, Mean age 50y,
53% DM]
1119 Exogenous
filtration
markers other
and urinary
iothalamate (U)
70 (41) Y (IFCC SRM 967) Y (IRMM) Re-expressed MDRD -6.3 (-7.8, -5.4) 19.4 (17.4,
21.1)
83%
CKD-EPI cystatin C 2011 -6.0 (-7.1, -4.9) 18.7 (17.5,
20.0)
84%
CKD-EPI creatinine-cystatin C
2011
-4.9 (-5.9, -4.2) 15.3 (14.0,
16.3)
92%
RS/CP: 5 studies (2 RS and 3 CP)
using equations developed in
diverse dataset [3% black, Mean age
50y, 53% DM]
CKD-EPI creatinine 2009 -3.7 (-4.6, -2.8) 15.4 (14.3,
16.5)
87%
CKD-EPI cystatin C 2012 -3.4 (-4.4, -2.3) 16.4 (14.8,
17.8)
86%
CKD-EPI creatinine-cystatin C
2012
-3.9 (-4.5, -3.2) 13.4 (12.3,
14.5)
92%
Average of CKD-EPI creatinine
2009 and CKD-EPI cystatin C
2012
-3.5 (-4.1, -2.8) 13.9 (12.9,
14.7)
92%
Stevens, LA et al.
117
France
CP: Clinical population in Paris,
France [29% women, 79% white, 8%
black, 13% other, Mean age 59y,
22% DM, 16%]
438
51
Cr-EDTA 34 (17) Y (IFCC SRM 967) Y (IRMM) Re-expressed MDRD -2 (-3, -1) 0.229
(0.210,
0.247)
84%
(83, 85)
CKD-EPI cystatin C (alone) 2011 3 (2, 3) 0.264
(0.239,
0.289)
73%
(72, 74)
CKD-EPI cystatin C 2011 2 (1, 2) 0.248
(0.223,
0.271)
79%
(78, 80)
CKD-EPI creatinine-cystatin C
2011
0 (0, 1) 0.193
(0.174,
0.211)
90%
(89, 91)
Abbreviations: CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; CP, clinical population; Cr-EDTA, chromium-ethylenediamine tetraacetic acid; DM, diabetes mellitus; GFR, glomerular
filtration rate; IFCC, International Federation of Clinical Chemistry; IQR, interquartile range; IRMM, International Reference Materials and Measurements; MDRD, Modification of Diet in Renal Disease; mGFR, measured glomerular
filtration rate; RMSE, root mean square error; RS, Research study; SCr, serum creatinine; SRM, standard reference material; U, urine; Y, yes
Footnotes:
*Bias is computed as estimated GFR minus measured GFR. A positive number indicates estimated GFR overestimates measured GFR. A negative number indicates estimated GFR underestimates measured GFR. A smaller absolute
value indicates lesser bias. A lower value for RMSE, IQR, or SD indicates greater precision.
wPrecision is evaluated as IQR for the differences between estimated and measured GFR.
zHigher values for P
30
indicate greater accuracy. Values in parenthesis are 95% confidence intervals.
54 Kidney International Supplements (2013) 3, 19–62
chapter 1

aspired to over time in those locations where these
recommendations are currently not able to be implemented.
The Work Group appreciated that not all laboratories have
capabilities to assay cystatin C. Different countries and
regions will have different availabilities for measurement of
GFR. The statement about GFR measurements mostly applies
to countries with tertiary care services such as kidney
transplantation and oncology.
Implications for Clinical Practice and Public Policy
It is important for clinicians to understand various methods
for estimating and measuring kidney function and the
situations in which specific methods may be superior in
clinical decision making about treatment and referral.
Standardized assays and robust equations are important
for epidemiological and planning purposes so that public
policy can be informed by more accurate estimates of CKD,
which may be possible with improved standardization of
both assays and equations.
In different parts of the world, different assays are used
and equations for estimating eGFR may differ. Thus,
appreciating and understanding local standards is important
for individual patients who may travel, and for comparative
research across countries or regions.
In the event that a clinician requires measurement of GFR
instead of an estimate, knowledge of these different tests and
availability of them is important. Situations in which
measurement would be required are likely quite infrequent
Table 18 | Strengths and limitations of GFR measurement methods and markers
Approach Strengths Limitations
Methods
Urinary clearance
Bladder catheter and continuous
intravenous infusion of marker
K Gold standard method K Invasive
Spontaneous bladder emptying
K Patient comfort
K Less invasive
K Possibility of incomplete bladder emptying
K Low flow rates in people with low levels of GFR
Bolus administration of marker
K Shorter duration K Rapidly declining plasma levels at high levels of GFR
K Longer equilibration time in extracellular volume expansion
24 h urinary collection
K Cumbersome
K Prone to error
Plasma clearance
K No urine collection required
K Potential for increased precision
K Overestimation of GFR in extracellular volume expansion
K Inaccurate values with 1-sample technique, particularly at
lower GFR levels
K Longer duration of plasma sampling required for low GFR
Nuclear imaging
K No urine collection or repeated
blood samples required
K Relatively short duration
K Less accurate
Markers
Inulin
K Gold standard
K No side effects
K Expensive
K Difficult to dissolve and maintain into solution
K Short supply
Creatinine
K Endogenous marker, no need
for administration
K Assay available in all clinical
laboratories
K Secretion can vary among and within individuals
Iothalamate
K Inexpensive
K Long half life
K Probable tubular secretion
K Requirement for storage, administration, and disposal of
radioactive substances when
125
I used as tracer
K Use of non-radioactive iothalamate requires expensive assay
K Cannot be used in patients with allergies to iodine
Iohexol
K Not radioactive
K Inexpensive
K Sensitive assay allows for low dose
K Possible tubular reabsorption or protein binding
K Use of low doses requires expensive assay
K Cannot be used in patients with allergies to iodine
K Nephrotoxicity and risk for allergic reactions at high doses
EDTA
K Widely available in Europe K Probable tubular reabsorption
K Requirement for storage, administration, and disposal of
radioactive substances when
51
Cr is used as tracer
DTPA
K Widely available in the US
K New sensitive and easy to
use assay for gadolinium
K Requirement for storage, administration, and disposal of
radioactive substances when
99m
Tc used as tracer
K Requires standardization for
99m
Tc
K Dissociation and protein binding of
99m
Tc
K Concern for NSF when gadolinium is used as the tracer
Abbreviations: DTPA, diethylenetriamine pentaacetic acid; EDTA, ethylenediamine tetraacetic acid; GFR, glomerular filtration rate; NSF, nephrogenic systemic fibros is.
Kidney International Supplements (2013) 3, 19–62
55
chapter 1

but include donor evaluation in kidney transplantation and
use of toxic drugs which have a narrow therapeutic range. We
acknowledge that drug development and clinical observation
programs may not define the various thresholds w ith
sufficient granularity to require greater accuracy than is
provided by eGFR
creat
. Guidance is evolving regarding kidney
function evaluation during drug development programs.
13
There are no direct implications for public policy for the
statement about GFR measurement.
Areas of Controversy, Confusion, or Non-consensus
The Work Group recognizes that no single creatinine-based
estimating equation will perform optimally in all clinical
circumstances and that there may be changes in the
performance of estimating equations over time and in
different regions. However, for the purpose of eGFR
reporting, it is important to select a single equation within
a region or country. At the writing of this guideline, in
North America, Europe, and Australia, the advantages of the
CKD-EPI equation at higher GFR make it more applicable
than the MDRD Study equation for general practice and
public health.
While cystatin C offers some advantages over SCr as the
basis of estimating equations, the cost of the assay and
potential lack of standardization across laboratories for this
‘newer’ test limi t our ability to recommend it as a preferred
or even usual second test after creatinine. We recognize that
these factors may lead to variations in implementation. The
recommendation to consider confirmatory or additional
testing if there is a need for more accurate determination of
GFR is important. That there are other laboratory markers to
estimate GFR (i.e., cystatin C) is stated here as there has been
accumulating data to support its use in these situations. We
have specifically mentioned cystatin C because of these data.
Clarification of Issues and Key Points
It is impor tant for clinicians to appreciate the need for
standardized assays and standardized equations for labora-
tory reporting of eGFR. Changes in laboratory assays or
calculation methods should be reported to clinicians in order
to avoid confusion when serially following individuals. This
is because values in an individual might indicate a worsening
or improvement in eGFR which may be attributable to
different assays or calculation methods, rather than a
reflection of true change.
When precise information about GFR is required, direct
measurement using reliable methods should be pursued.
Pediatric Considerations
For Recommendation 1.4.3.8 this guideline is fully applicable
in pediatrics.
1.4.4 Evaluation of albuminuria
1.4.4.1: We suggest using the following measurements
for initial testing of proteinuria (in descending
order of preference, in all cases an early
morning urine sample is preferred) (2B):
(1) urine albumin-to-creatinine ratio (ACR);
(2) urine protein-to-creatinine ratio (PCR);
(3) reagent strip urinalysis for total protein
with automated reading;
(4) reagent strip urinalysis for total protein
with manual reading.
1.4.4.2: We recommend that clinical laboratories report
ACR and PCR in untimed urine samples in
addition to albumin concentration or protein-
uria concentrations rather than the concentra-
tions alone. (1B)
1.4.4.2.1: The term microalbuminuria should
no longer be used by laboratories.
(Not Graded)
1.4.4.3: Clinicians need to understand settings that
may affect interpretation of measurements of
albuminuria and order confirmatory tests as
indicated (Not Graded):
K Confirm reagent strip positive albuminuria
and proteinuria by quantitative laboratory
measurement and express as a ratio to
creatinine wherever possible.
K Confirm ACR Z30 mg/g (Z3 mg/mmol) on
a random untimed urine with a subsequent
early morning urine sample.
K If a more accurate estimate of albuminuria
or total proteinuria is required, measure
albumin excretion rate or total protein
excretion rate in a timed urine sample.
RATIONALE
We recommend measurement of urinary albumin because it
is relatively standardized and because it is the single most
important protein lost in the urine in most chronic kid ney
diseases. Use of urinary albumin measurement as the
preferred test for proteinuria detection will improve the
sensitivity, quality, and consistency of approach to the early
detection and management of kidney disease.
By contrast, laboratory tests purporting to measure
urinary total protein are commonly flawed, often being
standardized against, and predominantly sensitive to, albu-
min. They have poor precision at low concentrations and
demonstrate poor between-laboratory agreement while being
insensitive, non-specific, and susceptible to a range of false-
positive and false-negative problems. There may occasionally
56 Kidney International Supplements (2013) 3, 19–62
chapter 1

be clinical reasons for a specialist to use PCR instead of
ACR to quantify and monitor significant levels of proteinuria
(e.g., in patients with monoclonal gammopathies).
Commonly used reagent strip devices measuring total
protein are insufficiently sensitive for the reliable detection of
proteinuria, do not adjust for urinary concentration, and are
only semi-quantitative. Furthermore, there is no standardization
between manufacturers. The use of such strips should be
discouraged in favor of quantitative laboratory measurements of
albuminuria or proteinuria. When used, reagent strip results
should be confirmed by laboratory testing (Figure 16).
The combination of reagent strips with automated reader
devices can improve inter-operator variability. More recently
launched reagent strip devices capable of producing albumin
or total protein results as a ratio to urinary creatinine require
further evaluation to provide evidence that they have
equivalent sensitivity and specificity to laboratory tests and
are economically advantageous.
Although the reference point remains the accurately timed
24-hour specimen, it is widely accepted that this is a difficult
procedure to control effectively and that inaccuracies in
urinary collection may contribute to errors in estimati on of
protein losses. In practice, untimed urine samples are a
reasonable first test for ascertainment of albuminuria.
An EMU (‘first pass’) sample is preferred since it correlates
well with 24-hour protein excretion, has relatively low
Opportunistic finding
of positive reagent
strip result for albumin
or total protein
Inspect urine
Repeat
reagent strip
positive?
Cloudy?
Measure urinary ACR
Yes
MSU for C&S
Treat infection if positive
Yes
Patient requires testing
for proteinuria as part of
CKD detectionor
surveillance program
No
ACR >300 mg/g
(>30 mg/mmol)?
Severely increased
proteinuria
ACR ≥30 mg/g
(≥3mg/mmol)?
Yes
No
Send 2 further EMU samples for ACR
within the next 2 months
Yes
EMU
ACR ≥30 mg/g
(≥3 mg/mmol) in at
least 1of 2
subsequent
samples?
Continue routine
observation
Refer for specialist
assessment e.g.,
possible biopsy
Hematuria?
Yes
Yes
No
Moderately increased
albuminuria
No
No
No
Figure 16 | Suggested protocol for the further investigation of an individual demonstrating a positive reagent strip test for
albuminuria/proteinuria or quantitative albuminuria/proteinuria test. Reagent strip device results should be confirmed using
laboratory testing of the ACR on at least two further occasions. Patients with two or more positive (Z30 mg/g or Z3 mg/mmol) tests on
early morning samples 1-2 weeks apart should be diagnosed as having persistent albuminuria. The possibility of postural proteinuria should
be excluded by the examination of an EMU. PCR measurement can be substituted for the ACR but is insensitive in the detection of
moderately increased albuminuria/proteinuria. Approximate PCR equivalent to an ACR of 30 mg/mmol is 50 mg/mmol. ACR, albumin-to-
creatinine ratio; C&S, culture and sensitivity; CKD, chronic kidney disease; EMU, early morning urine; MSU, mid-stream urine; PCR, protein-to-
creatinine ratio.
a
Consider other causes of increased ACR (e.g., menstrual contamination, uncontrolled hypertension, symptomatic urinary
tract infection, heart failure, other transitory illnesses, and strenuous exercise), especially in the case of type 1 diabetes present for less than
5 years. The presence of hematuria may indicate non-diabetic renal disease. This figure was published and adapted from Lamb EJ, Price
CP.
122
Kidney function tests, in Tietz Textbook of Clinical Chemistry and Molecular Diagnostics, eds Burtis CA, Ashwood E, Bruns DE, 5
th
edition, pp 669-708, 2012. Copyright Elsevier.
Kidney International Supplements (2013) 3, 19–62 57
chapter 1

intra-individual variability , and is required to exclude the diag-
nosis of orthostatic (postural) proteinuria. However, a random
urine sample is acceptable if no EMU sample is available.
The concentration of protein or albumin in a urine sample
will be affected by hydration (i.e., how diluted or concen-
trated a urine sample is). Creatinine excretion is considered
to be fairly constant throughout the day and it has become
customary to correct for urinary concentration by exp ressing
either the protein or albumin concentrations as a ratio to the
creatinine concentration in the same sample.
Timed urine collections may be used for confirmatory
purposes but are not required except in circumstances in
which untimed urine ACR is less accurate. It is worthwhile
noting that albumin and protein excretion display consider-
able biological variability and may be increased by a variety of
pathological and non-pathologic al factors. Consequently,
confirmation of increased excretion rates is recommended.
Evidence Base
Why is albumin measurement being recommended instead
of total protein?
Urine albumin measurement provides a
more specific and sensitive measure of changes in glomerular
permeability than urinary total protein.
123–125
There is
substantial evidence linking increased albuminuria to
outcomes of CKD
4,30
(e.g., CKD Prognosis Consortium
2–5
,
Nord-Trøndelag Health Study [HUNT 2]
125a
, Prevention of
Renal and Vascular Endstage Disease [PREVEND]
125b
). There
is also evidence that urinary albumin is a more sensitive test
to enable detection of glomerular pathology associated with
some other systemic diseases including diabetes, hyperten-
sion and systemic sclerosis.
126–129
In health, relatively small amounts of albumin (o30 mg/
24 hours) are lost in the urine. Because of this, and
additionally because total protein assays are imprecise and
insensitive at low concentrations, relatively large increases in
urine albumin excretion can occur without causing a
significant measurable increase in urinary total protein.
125
Total protein measurement is problematic in urine due to:
large sample-to-sample variation in the amount and
composition of proteins; high and varia ble concentrations
of non-protein interfering substances relative to the protein
concentration; and hig h inorganic ion content. All these
factors affect the precision and accuracy of the various
methods. Most laboratories currently use either turbidimetry
or colorimetry
130
to measure total protein and as with urine
reagent strip analysis, these methods do not give equal
analytical specificity and sensitivity for all proteins which can
contribute to diverse estimates of proteinuria preva-
lence.
131,132
Most methods tend to react more strongly with
albumin than with globulin and other non-albumin pro-
teins.
34,133–135
There are significant interferences causing
falsely high results.
136–138
. There is no reference measurement
procedure and no standardized reference material for urinary
total protein listed by the JCTLM. The variety of methods
and calibrants in use means that there is inevitably significant
between-laboratory variation.
139–141
Since a variable mixture
of proteins is measured, it is difficult to define a standardized
reference material.
How should albumin be measured and reported?
Albumin should be measured using immunological assays
capable of specifically and precisely quan tifying albumin at
low concentrations and of producing quantitative results over
the clinically relevant range. Currently urinary albumin is
predominantly measured by diagnostic laboratories using
turbidimetric assays.
130
At present there is no reference
measurement procedure or standardized reference material
for urine albumin listed by the JCTLM, although the NKDEP
and the International Federation of Clinical Chemistry and
Laboratory Medicine have recently established a joint
committee to address these issues.
142,143
At present, most
assays are standardized against a serum-based calibrant (CRM
470) distributed by the IRMM of the European Commission,
as has previously been recommended by KDIGO.
31
Albumin concentration should be reported as a ratio to
urinary creatinine concentration (mg/mmol or mg/g). A CR
results should be expressed to one decimal place (mg/mmol) or
whole numbers (mg/g). Both enzymatic and Jaffe assays are
suitable for the measurement of creatinine in urine. We suggest
that the term ‘ microalbuminuria’ no longer be used because it
can be misleading in suggesting that the albumin may be small or
different in some way . The proposed albuminuria categories
A1-3 are a more clinically meaningful way to express information
about categories within the continuum of albumin excretion.
Reagent strip point-of-care testing devices capable of
measuring low concentrations of albumi n are also available
producing both semi-quantitative and fully quantitative ACR
results. Reasonable analytical
144–147
and diagnosti c perfor-
mance has been demonstrated.
148–150
While studies of these
devices have been somewhat limited in size, they demonstrate
their potential to play a significant role in the care pathway of
patients suspected of having CKD.
Why are reagent strip devices for protein measurement
considered less accurate than laboratory measurement?
Reagen t strip devices for proteinuria detection hav e been in use
for more than 50 years. As discussed earlier, a positive reagent
strip result is also associated with outcomes of CKD. Such
devices have been used to support screening pro grams in some
countries,
151–153
although there appears to be no evidence
supporting such screening of unselected populations.
154
Although purporting to measure total protein, the reagent
pad is most sensitive to albumin.
155–157
There is evidence that
strips from different manufacturers perform differently at the
cutoff (‘ þ ’) concentration of 300 mg/l and degrees of ‘plus-
ness’ between different manufacturers don’t always corres-
pond to the same nominal concentration of protein in
urine.
124
Concentrated urines may give a color change in the
positive range of a reagent strip dev ice even though protein
loss remains normal and vice versa. False-positive results may
occur if the urine is alkalinized (e.g., due to urinar y tract
58 Kidney International Supplements (2013) 3, 19–62
chapter 1

infection) or in the presence of quaternary ammonium
compounds that alter the pH of the urine. The performance
of reagent strips is operator-dependent
158
and affected by the
presence of colored compounds such as bilirubin and certain
drugs (e.g., ciprofloxacin, quinine, and chloroquine).
159
Reagent strips cannot reliably distinguish between protein-
uria categories
124,157
and show relatively poor diagnostic
accuracy for proteinuria detection.
160,161
In the Australian
Diabetes, Obesity and Lifest yle (AusDiab) study, a reagent
strip reading of þ or greater had 58% and 99% sensitivity
for detecting ACR Z30 mg/g (Z 3 mg/mmol) and Z300 mg/
g(Z30 mg/mmol), respectively. 47% of individuals who
tested þ or greater had an ACR Z30 mg/g (Z 3 mg/mmol)
on laboratory testing.
162
Automated devices capable of reading the color changes of
reagent strips using reflectance spectrometry are available.
These reduce inter-operator variability and improve diag-
nostic accuracy.
150,158,163
A creatinine test pad has been added
to some reagent strip systems to enable a PCR to be reported
and so reduce the intra-ind ividual variation seen with
random urine collections. Such devices have been shown to
be suitable for ruling out significant proteinuria (4300 mg/
24 hours) in an outpatient setting.
149
Correcting for urinary dilution. Since creatinine excretion
in the urine is fairly constant throughout the 24-hour period,
measurement of ACR (or PCR) allows correction for
variations in urinary concentration.
164,165
ACR is a suitable
alternative to timed measurement of urine albumin
loss.
143,166–170
PCR on random or early morning untimed
samples shows good diagnostic performance and correlation
with 24-hour collection.
160,163,171–177
Expressing albumin as a ratio to creatinine reduces intra-
individual variability: lowest variability for the ACR has been
reported in EMU samples as opposed to other untimed
samples or timed collections.
142,178
In one study albumin
variability was reduced from 80% to 52% when expressed as
an ACR rather than an albumin concentration.
179
The
within-subject biological variation for urinary ACR in an
EMU has been reported to be 31%, compared to 36% for
urinary albumin concentration.
180
The same stud y reported
variability for ACR of 103% and 85% in random and timed
24-hour collections, respectively.
180
Intra-individual variation
for protein loss is also significantly reduced when reported as
a PCR compared to protein concentration in random urine
samples collected throughout the day (a mean reduction
from 97% to 39%).
179
Why and how should a finding of albuminuria be confirmed?
Given the high biological variation and other pathological
and physiological causes of albuminuria (Table 19),
143
repeat
testing to confirm albuminuria, ideally using an EMU and
laboratory testing, is recommended (Figure 16).
There has been extensive discussion in the literature about
the appropriate urine sample to use for the investigation of
protein loss. It is generally recognized that a 24-h our sample
is the definitive means of demonstrating the presence of
proteinuria. However, overnight, first void in the morning
(i.e., EMU), second void in the morning, or random sample
collections can also be used. In a systematic review random
urine PCR was shown to have better performance as a test for
ruling out significant proteinuria than as a ‘‘rule-in’’ test; the
authors suggested that positive PCR results may still require
Table 19 | Factors affecting urinary ACR
Factor Examples of effect
Preanalytical factors
Transient elevation in albuminuria Menstrual blood contamination
Symptomatic UTI
181
Exercise
182
Upright posture (orthostatic proteinuria)
41,183
Other conditions increasing vascular permeability (e.g., septicemia)
Intraindividual variability Intrinsic biological variability
180
Genetic variability
184
Preanalytical storage conditions Degradation of albumin before analysis
a
Non-renal causes of variability in creatinine
excretion
Age (lower in children and older people)
Race (lower in Caucasian than black people)
Muscle mass (e.g., lower in people with amputations, paraplegia, muscular dystrophy)
Gender (lower in women)
Changes in creatinine excretion Non-steady state for creatinine (AKI)
Analytical factors
Antigen excess (‘prozone’) effect Samples with very high albumin concentrations may be falsely reported as low or normal using some
assays
124
Abbreviations: ACR, albumin-to-creatinine ratio; AKI, acute kidney injury; UTI, urinary tract infection.
a
Samples for urinary albumin (or total protein) measurement may be analyzed fresh, stored at 41C for up to one week, or stored at -701C for longer periods. Freezing at -201C
appears to result in loss of measurable albumin and is not recommended. When analyzing stored samples, they should be allowed to reach room temperatureand
thoroughly mixed prior to analysis.
142
Kidney International Supplements (2013) 3, 19–62 59
chapter 1

confirmation with a 24-hour collection.
185
If an EMU is
unavailable, subsequent samples can give a reliable indication
of the 24-hour urine protein loss.
174
International Relevance
The recommendation to replace urinary total protein with
albumin as the test of choice in testing for proteinuria is
consistent with most,
1,31,130,186,187
but not all,
188,189
current
national and international guidance. It is accepted that cost
pressures may affect implementation of this recommendation
and may differ across the world.
Most international guidelines have also discouraged the
use of reagent strip analysis for proteinuria detec-
tion.
186,189–191
Nevertheless, in the present guideline we
acknowledge that these devices may have a role, par ticularly
in settings where access to laboratory services may be limited.
ACRs in North America tend to be repor ted in mg/g
whereas in other parts of the world usage of mg/mmol
predominates. This difference appears unlikely to be resolved
in the foreseeable future. When publishing data authors
should ensure either that both units are cited or that a
conversion factor is provided.
There is increasing adoption of the term ‘albuminuria’
instead of microalbuminuria by international and national
laboratory and some clinical organizations.
Implications for Clinical Practice and Public Policy
Direct reagent costs of total protein measurement are
generally lower than those of albumin measurement, which
requires antibody-based reagents. It is often considered that
reagent strip analyses are a cheaper option. Therefore some
health-care systems may struggle to justify the recommenda-
tions in this guideline.
Costs of diagnostic tests vary depending on local financial
agreements between hospitals and suppliers. In England,
the National Institute for Health and Clinical Exc ellence (NICE)
sampled a small random number of laboratories and estimated
the average cost of an ACR to be d2.16 whereas a PCR cost
d1.42.
186
It is ackno wledge d that incr eased use of A CR testing
may reduce the unit cost on the basis of economies of scale. In
Canada, laboratory analysis costs (Canadian dollars) of $2.81
for reagent strip, $11.67 for PCR, and $29.23 for ACR have been
cited.
192
In relation to albumin-specific reagent strips, a cost of
approximately $4 for a Micral test II (Roche Diagnostics)
compare d to $2 for a laboratory ACR has been reported.
193
The cost- and clinical-effectiveness of an approach
utilizing reagent strip testing followed by laboratory mea-
surement compared to an approach in which samples are
submitted directly to the laboratory (for either albumin or
protein measurement) has recently been evaluated in a health
economics model.
186
The model favored abandoning the use
of reagent strips for identification of proteinuria.
Areas of Controversy, Confusion, or Non-consensus
Some data suggest that ACR is a poorer predictor of 24-hour
total protein loss than PCR
194
and has no advantage over
PCR as a predictor of renal outcomes and mortality in
patients with CKD.
195,196
In the prediction of future
transplant rejection, PCR has been reported to have equal
utility to ACR,
192
although in a separate study ACR was
found to be a better predictor.
197
In the setting of preeclampsia, proteinuria is generally
defined as Z300 mg/24 hours or a PCR Z300 mg/g
(Z30 mg/mmol).
175
Currently, there is insufficient evidence
to substitute urine albu min measurement for total protein in
this setting.
172
Creatinine excretion is affected by a variety of non-renal
influences (Table 19) and it therefore follows that different
cutoffs for ACR (and PCR) may be required in different
individuals.
194,198
While age-related cutoffs have not gen-
erally been applied in clinical practice, clinicians should bear
this in mind when interpreting urine ACR data in older
individuals or those with very low body mass, as these will
impact the urine creatinine excretion.
While most guidelines agr ee that an ACR greater than
appro ximately 3 mg/mmol (30 mg/g) is pathological in the
setting of diabetes, in the non-diabetic population a higher
threshold has commonly been used to define proteinuria. In the
NICE guideline in England and Wales, proteinuria in non-
diabetic individuals was defined as Z30 mg/mmol (Z300 mg/g),
with higher level proteinuria being 470 mg/mmol (4700 mg/
g).
186
Confirmation of results lying between 30 and 70 mg/mmol
(300-700 mg/g) was recommended.
186
The present guideline
proposes a lower threshold definition for albuminuria for use in
both diabetic and non-diabetic individuals.
A study from Italy in type 2 diabetes has reported that,
although intra-individual biological varia tion of albuminuria
is large, a single sample (either ACR or timed collection) can
accurately classify patients into albuminuria categories,
negating the need for multiple collections.
178
Some data suggest that a significant proportion of
albumin present in urine may be non-immunoreactive,
199–202
although this finding has been questioned.
203,204
There is a substantial existing literature using the term
microalbuminuria and many existing guidelines use this term
especially in the context of diabetes and cardiovascular risk,
as its presence confers risk. Nonetheless, the Work Group
believes that it is important for this international guideline to
foster ‘best practices’ and clarity of communication, and
since the risk of adverse events is continuous throughout the
spectrum of albuminuria, we encourage adoption of the term
‘albuminuria’ with subsequent quantification of the level or
amount.
Pediatric Considerations
For Rec ommendation 1.4.4.1, this set of statements would need
to be altered for application in the pediatric practice as follows:
We suggest using the following measurements for initial
testing of proteinuria in children (in descending order of
preference):
(1) urine PCR, EMU sample preferred;
(2) urine ACR, EMU sample preferred;
60 Kidney International Supplements (2013) 3, 19–62
chapter 1

(3) reagent strip urinalysis for total protein with automat ed
reading;
(4) reagent strip urinalysis for total protein with manual
reading.
For Recommendations 1.4.4.2 and 1.4.4.3, this set of
statements would need to be altered for application in the
pediatric practice as follows:
Currently the urinary PCR should be favored over the
urine ACR in children. Unlike in adults where powerful
evidence exists in support of the use of measures of albumin
rather than total protein to predict adverse outcomes, this
level of evidence is currently lacking in children.
205
However,
current longitudinal trials such as CKiD
55
and European 4C
78
may eventually shed light on this issue.
In children the unde rlying conditions associated with the
diagnosis of CKD are also important considerations as to
which form of testing is most valuable. Unlike adults where
the majority of patients with CKD are attributed to an
underlying glomerular disease or hypertensive damage, the
vast majority of children have underlying developmental
abnormalities often referred to as CAKUT (congenital
anomalies of the kidney and urinary tra ct).
70
This relative
paucity of glomerular conditions makes the use of albumin
excretion a less sensitive test for diagnostic purposes as many
children will have underlying tubular conditions and hence
tend to excrete more Tamm-Horsfall protein and other low-
molecular-weight proteins that will not be captured by the
albumin-to-creatinine (or formal albumin excretion) assay.
For Recommendations 1.4.4.2 and 1.4.4.2.1 this guideline
is fully applicable in pediatrics. The recommendation that
clinical laboratories report ACR and PCR in untimed
urine samples in addition to albumin concentration or
proteinuria concentrations rather than the concentrations
alone is valid and useful in the pediatric population. As per
Recommendation 1.2.4, however, note should be made
that age-related normal values for urinary protein losses
must be considered when laboratories choose to report either
ACR or PCR.
Albuminuria in children, whether measured as an absolute
value per day, an excretion rate, or as an albumin to
creatinine ratio is fraught with more uncertainity than in
adults as they are known to vary across categories of age, sex,
height, weight, and Tanner staging.
206
In two recent reviews by Rademacher
206
and Tsioufis et al.,
205
both groups examined the results of all relevant studies on
normative values of AER or ACR. Rademacher’s paper in
particular provides detailed information on the mean AER
values (with SD) across a variety of studies, ages, sex, and race,
and provides a normative estimate for overnight AER of
between 2-6 mg/minute or a 95
th
perc enti le value from 4.5–28 mg/
minute. Similarly, they summarize results for A CR in normal
children and suggest that the mean for children older than 6
years would seem to fall between 8-10 mg/g (0.8-1.0 mg/mmol).
For Recommendation 1.4.4.3, this guideline is fully
applicable in pediatrics.
1.4.4.4: If significant non-albumin proteinuria is sus-
pected, use assays for specific urine proteins
(e.g., a
1
-microglobulin, monoclonal heavy or
light chains, [known in some countries as
‘‘Bence Jones’’proteins]). (Not Graded)
RATIONALE
Testing for tubular proteinuria using a total protein approach
almost certainly has very poor sensitivity for detecting
tubular disease. When an isolated tubular lesion is suspected
(Table 3), this is probably best investigated by measuring a
specific tubular protein (e.g., a
1
-microglobulin) using an
immunoassay approach.
Evidence Base
There have been concerns that replacing urinary total protein
measurement with albumin measurement may cause non-
albuminuric (effectively tubular and overproduction) pro-
teinuria to be missed. Low-molecular-weight proteinuria is a
defining feature in some uncommon kidney diseases (e.g.,
Dent’s disease).
207
However, for some of the reasons already
discussed, total protein assays will also be poor at detecting
tubular proteinuria. When investigating patients for tubular
proteinuria, it is advisable to use assays targeted at specific
tubular proteins.
In the AusDiab study , of those with proteinuria (2.4% of the
general population, defined as a PCR 423 mg/mmol [230 mg/
g]) 92% had albuminuria (defined as an ACR 43.4 mg/mmol
[34 mg/g]); 8% had an ACR within the refer ence range.
208
These
individuals were less likely to have diabetes than those with both
proteinuria and albuminuria, but no further information is
available as to the nature of the proteinuria in these individuals
or its likely significance. The authors speculate that these
individuals could have had light chain proteinuria or interstitial
nephropathies. Using albuminuria testing to identify proteinur -
ia had a specificity of 95%. The negative pr edictiv e value was
99.8% and the positive predictive value was 32.4%. The authors
concluded that testing for albuminuria rather than pr oteinuria
was supported.
As discussed above, quite significant increases in urinary
albumin loss have to occur before such an increase is
detectable on the background of a total protein assay. The
situation is even more extreme for tubular proteins which, in
health, are present in urine at lower concentrations than
albumin (e.g., normal daily losses of retinol binding protein,
a
1
-microglobulin and b
2
-microglobulin are 0.08, 3.6, and
0.1 mg/d, respectively).
209
This problem will be exacerbated
by the fact that the recognition of tubular proteins by some
total protein assays is poor.
210
In disease states c oncentrations of tubular proteins, at least
collectively, can reach levels detectable by total protein assays.
For example, among patients with tubulointerstitial disease
but without renal insufficiency, median concentrations of a
1
-
microglobulin were 37 mg/l, with concentrations up to 100 mg/l
being observed; higher conc entrations were seen in patients with
decreased GFR.
211
Among a group of patients with acute
Kidney International Supplements (2013) 3, 19–62 61
chapter 1

tubular necrosis requiring dialysis treatment, median a
1
-
microglobulin concentration was 35 mg/mmol of creatinine.
212
Howev er, although tubular proteinuria is characterized by a
relativ e increase in low-molecular-weight protein concentra-
tions, generally albumin still remains a significant component of
the total protein concentration. Indeed, it is thought that
tubulardiseaseresultsinanincreaseinalbuminlossasaresult
of decreased tubular reabsorption of filtered albumin. For
example, it has been estimated that when tubular absorption
fails completely, b
2
-microglobulin loss increases to 180 mg/24
hours (approximately 1800-fold normal) but there will also be
an increase in urinary albumin loss to about 360 mg/24 hours
(appro ximately 20-fold normal).
209
In a series of patients with
Dent’s disease, a classical tubular disor der, 21 of the 23 patients
demonstrating increased urinary a
1
-microglobulin and b
2
-
microglobulin loss also had increased urinary albumin loss:
those who did not had borderline increases in tubular protein
losses that would not have been detectable using a total protein
measurement approach.
207
The authors comment that in those
patients in whom proteinuria was marked (41g/d),urinary
albumin loss was also markedly incr eased. In some situations,
however, tubular proteinuria in the absence of albuminuria has
been reported (e.g., in some children with type 1 diabetes
213
and
in kidney scarring in reflux nephropathy
214
).
International Relevance
There is no reason to believe that there are significant
differences around the world with respect to incidence or
prevalence of conditions in which measurement of non-
albumin proteins would be required. The availability of
reliable tests for these alternative proteins, however, may be
different in different regions.
Implications for Clinical Practice and Public Policy
The incidence and prevalence of tubular disorders will vary
geographically with the clinical setting (e.g., adult or
pediatric practice) and factors such as occupational exposure.
Clinicians should agree with their local laboratories a suitable
approach to the detection of tubular proteinuria and
laboratories should be able to advise on suitable sample
handling procedures. It is acknowledged that many labora-
tories do not currently offer assays of tubular proteins.
In patients with suspected myeloma, monoclonal
heavy or light chains (known in some countries as Bence
Jones) protein should be sought in concentrated urine using
electrophoresis with immunofixation of any identified
protein bands in accordance with current myeloma guide-
lines.
215
Simultaneous albumin measurement is needed when
the possibility of immunoglobulin light chain (AL) amyloid
or light chain deposition disease is suspected.
Non-albumin proteinuria may also be suspected in
patients with di sorders of tubular function (see Table 3).
Areas of Controversy, Confusion, or Non-consensus
Testing for proteinuria using a urine albumin rather than
total protein first-line approach may occasionally miss cases
of tubular proteinuria but the significance of this problem is
probably overestimated and should be the subject of further
research.
Earlier guidance from KDOQI
1
suggested that proteinuria
in children should be detected with total protein rather than
albumin assays due to the higher prevalence of non-
glomerular diseases in this group of patients. For the reasons
outlined above, we do not think total protein assays are
suitable for this purpose and would ideally recommend
testing for albumin and for specific tubular proteins when
non-glomerular disease is suspected.
Pediatric Considerations
For Recommendation 1.4.4.4, this statement is fully applic-
able in pediatrics. In children the likelihood of any form of
overflow proteinuria such as seen in conditions of heavy or
light chain production is extremely low; however a significant
number of underlying genetic tubular disorders do exist and
protein electrophoresis can assist the practitioner in deter-
mining the presence of such a condition or the concurrent
finding of severe tubular injury in addition to a glomerular
condition.
DISCLAIMER
While every effort is made by the publishers, editorial board,
and ISN to see that no inaccurate or misleading data, opinion
or statement appears in this Journal, they wish to make it
clear that the data and opin ions appearing in the articles and
advertisements herein are the responsibility of the contri-
butor, copyright holder, or advertiser concerned. Accord-
ingly, the publishers and the ISN, the editorial board and
their respective employers, office and agents accept no
liability whatsoever for the consequences of any such
inaccurate or misleading data, opinion or statement. While
every effort is made to ensure that drug doses and other
quantities are presented accurately, readers are advised that
new methods and techniques involving drug usage, and
described within this Journal, should only be followed in
conjunction with the drug manufacturer’s own published
literature.
SUPPLEMENTARY MATERIAL
Supplemental Table 1: Search strategy.
Supplemental Table 2: Equations based on serum creatinine assays in
adults that are not traceable to the standard reference material.
Supplemental Table 3: Equations based on serum cystatin C assays in
adults that are not traceable to standard reference material.
Supplementary material is linked to the online version of the paper at
http://www.kdigo.org/clinical_practice_guidelines/ckd.php
62 Kidney International Supplements (2013) 3, 19–62
chapter 1
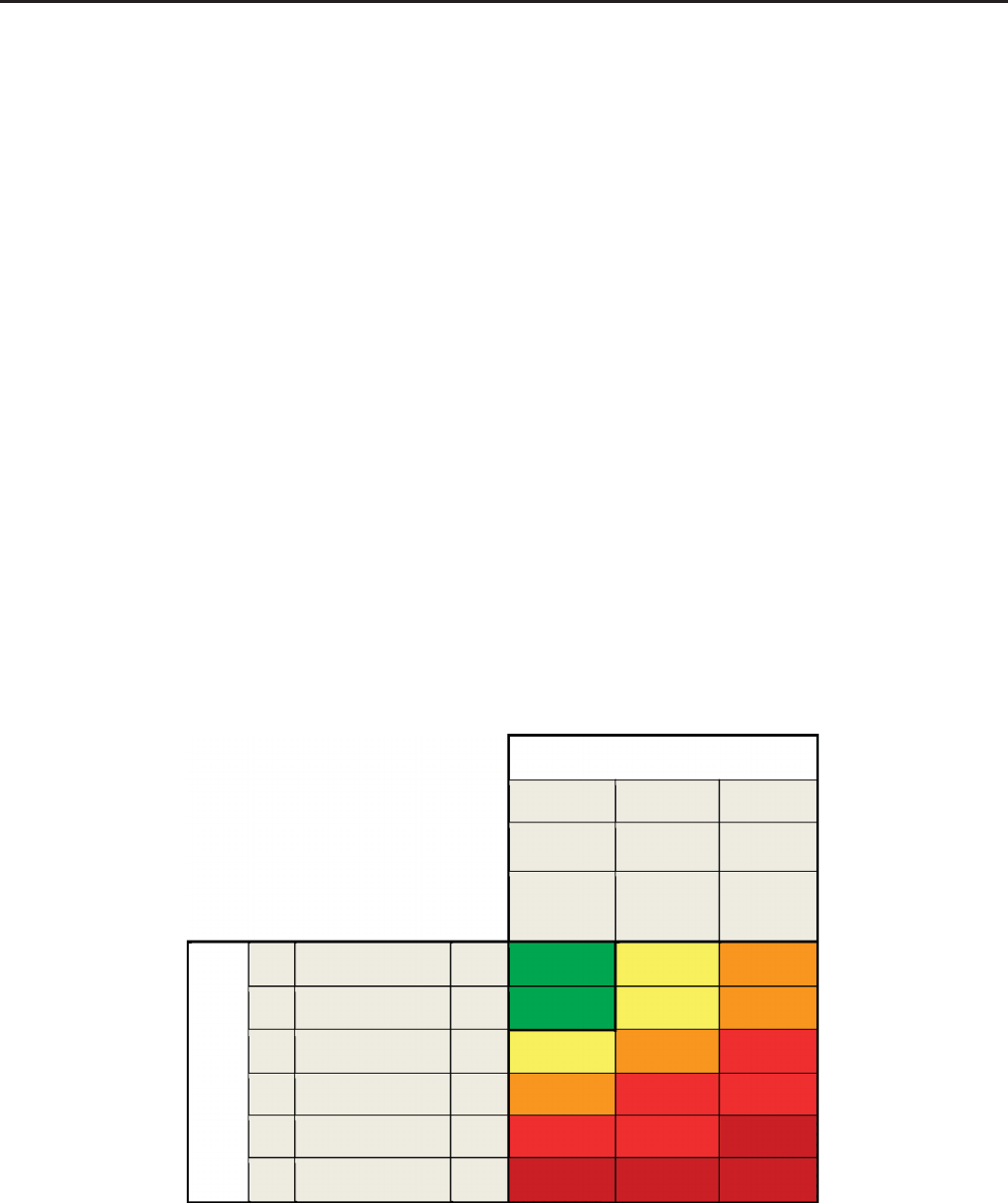
Chapter 2: Definition, identification, and prediction
of CKD progression
Kidney International Supplements (2013) 3, 63–72; doi:10.1038/kisup.2012.65
2.1: DEFINITION AND IDENTIFICATION OF CKD
PROGRESSION
2.1.1: Assess GFR and albuminuria at least annually
in people with CKD. Assess GFR and
albuminuria more often for individuals at
higher risk of progression, and/ or where
measurement will impact therapeutic deci-
sions (Figure 17). (Not Graded)
RATIONALE
The statement is worded this way to remind the practitioner
to use both GFR and albuminuria in order to assess
progression and is consistent with the definition offered in
Chapter 1 regarding definitions of CKD which include both
parameters. There is increasing evidence which supports that
both parameters are valuable. Lower GFR and greater
albuminuria are both associated with an increased rate of
progression and are synergistic.
Mor e frequent measures of eGFR and albuminuria should be
considered in patients with a lower GFR and greater
albuminuria as these people are more likely to progress.
Frequency of measurement should also be individualized based
on the patient history and underlying cause of kidney disease.
In specific conditions (e.g., GN or increased levels of
albuminuria), frequent (every 1–3 months) assessment may
guide therapeutic decisions. Regular monitoring of stable
patients may include more frequent monitoring than
annually, but will be dictated by underlying cause, history,
and estimates of GFR and ACR values obtained previously.
Evidence Base
There is variability in the presence of or rate of decline
of kidney function in those with CKD. The rate at which
http://www.kidney-international.org chapter 2
& 2013 KDIGO
Guide to Frequency of Monitoring
(number of times per year) by
GFR and Albuminuria Category
Persistent albuminuria categories
Description and range
A1 A2 A3
GFR categories (ml/min/1.73 m
2
)
Description and range
G1
Normal or high ≥90 1 if CKD 1 2
G2
Mildly decreased 60–89 1 if CKD 1 2
G3a
Mildly to moderately
decreased
45–59 1 2 3
G3b
Moderately to
severely decreased
30–44 2 3 3
G4
Severely decreased 15–29 3 3 4+
G5
Kidney failure <15 4+ 4+ 4+
Normal to
mildly
increased
Moderately
increased
Severely
increased
<30 mg/g
<3 mg/mmol
30–300 mg/g
3–30 mg/mmol
>300 mg/g
>30mg/mmol
Figure 17 | GFR and albuminuria grid to reflect the risk of progression by intensity of coloring (green, yellow, orange, red, deep
red). The numbers in the boxes are a guide to the frequency of monitoring (number of times per year). Green reflects stable disease, with
follow-up measurements annually if CKD is present; yellow requires caution and measurements at least once per year; orange requires
measurements twice per year; red requires measurements at 3 times per year while deep red may require closest monitoring approximately
4 times or more per year (at least every 1–-3 months). These are general parameters only based on expert opinion and must take into
account underlying comorbid conditions and disease state, as well as the likelihood of impacting a change in management for any
individual patient. CKD, chronic kidney disease; GFR, glomerular filtration rate. Modified with permission from Macmillan Publishers Ltd:
Kidney International. Levey AS, de Jong PE, Coresh J, et al.
30
The definition, classification, and prognosis of chronic kidney disease: a KDIGO
controversies conference report. Kidney Int 2011; 80: 17–28; accessed http://www.nature.com/ki/journal/v80/n1/full/ki2010483a.html
Kidney International Supplements (2013) 3, 63–72 63
this decline occurs also varies based on the underlying
population, cause of CKD, presence of albuminuria/protein-
uria, comorbidities and age. The Work Group searched the
literature for longitudinal studies that evaluated decline
in kidney function. As outlined in Table 20 the study
populations included healthy adults, those with comorbidity,
as well as a subgroup of adults aged 65 and older.
Data from the PREVEND study, a prospective, popula-
tion-based cohort study, provides important information
on decline in kidney function at the population level.
219
The PREVEND study evaluated 6894 people over a 4-year
period and reported loss in eGFR of 2.3 ml/min/1.73 m
2
/
4 years in the whole population, 7.2 ml/min/1.73 m
2
/4 years
in participants with macroalbuminuria (4300 mg/24 hours)
and 0.2 ml/min/1.73 m
2
/4 year in participants with impaired
renal function. The yearly decline in eGFR among a Japanese
general population over 10 years was slightly lower at
0.36 ml/min/1.73 m
2
/year.
220
The rate of eGFR decline was
approximately two times higher in participants with
proteinuria, and about 1.5 times higher among participants
with hypertension.
220
Among adults aged 65 and older,
progression (median follow-up 2 years) varied by sex and
presence of diabetes.
223
In general these studies suggest
progression rates of approximately 0.3 to 1 ml/min/1.73 m
2
/
year among participants without proteinuria or comor-
bidity and rates of approximately two to three times higher
among participants with proteinuria or comorbidity. The
somewhat surprising finding that eGFR had low rates of
progression among the group with impaired renal function
at baseline
219
has been shown in other studies
225
and may
relate to the statistical phenomenon of regression to the
mean. There is also a concern that it is hard to maintain
consistent calibration of the SCr assay over time and
progression results are highly sensitive to drift in the
creatinine assay.
Studies evaluating rate of decline in eGFR among
populations with CKD specifically are outlined in Table 21
and typically demonstrate a slightly more rapid rate of
decline in this subgroup, thus requiring more frequent
assessment of GFR and albuminuria.
Data from the MDRD Study during a mean 2.2 year
follow-up showed that the average rate of decline in GFR
ranged from 2.3 to 4.5 ml/min/year, depending on the
baseline GFR and protein/MAP treatment assignments.
227
Similarly, a more recent study of 4231 patients with GFR
categories G3a-G5 (GFRo60 ml/min/1.73 m
2
) referred to a
nephrologist showed a mean decline in GFR of 2.65 ml/min/
1.73 m
2
/year,
231
and variability in the rates of decline in this
referred cohort.
Note that there have been no studies which evaluate the
utility of more or less frequent moni toring in CKD cohorts.
International Relevance
Frequency of measurements of GFR and albuminuria may
vary by country and so are economic resources available
to support such testing, and the ability to implement
therapeutic strategies to address changes. Nonetheless, given
the availability of simple monitoring tools like urine reagent
Table 20 | Decline in kidney function in various populations (longitudinal studies only)
Reference Population N GFR decline
Healthy
Slack TK
216
Healthy kidney donors 141 0.40 ml/min/year
Rowe JW et al.
217
Healthy males 293 0.90 ml/min/1.73 m
2
/year (CrCl)
Lindeman RD
218
Healthy males 254 0.75 ml/min/year (CrCl)
Halbesma N et al.
219
PREVEND cohort (all participants) 6894 0.55 ml/min/1.73 m
2
/year
Imai E et al.
220
Annual health exam participants in Japan 120,727 0.36 ml/min/1.73 m
2
/year
Matsuchita K et al.
221
Atherosclerosis Risk In Communities Cohort 13,029 0.47%/year (median)
Kronborg J et al.
222
Healthy adults from Norway 4441 1.21 ml/min/1.73 m
2
/year (men)
1.19 ml/min/1.73 m
2
/year (women)
With comorbidity
Lindeman RD
218
Males with renal/urinary tract disease 118 1.10 ml/min/year (CrCl)
Lindeman RD
218
Males with hypertension 74 0.92 ml/min/year (CrCl)
Halbesma N et al.
219
PREVEND cohort – adults with macroalbuminuria (4300 mg/24 hours) 86 1.71 ml/min/1.73 m
2
/year
Halbesma N et al.
219
PREVEND cohort - Adults with impaired renal function
(5% lowest CrCl/MDRD GFR)
68 0.05 ml/min/1.73 m
2
/year
Imai E et al.
220
Annual health exam participants in Japan with hypertension 16,722 0.3 to 0.5 ml/min/1.73 m
2
/year
Imai E et al.
220
Annual health exam participants in Japan with proteinuria
(X1+ dipstick proteinuria)
2054 0.6 to 0.9 ml/min/1.73 m
2
/year
Older adults
Hemmelgarn B et al.
223
Males age 465 with diabetes 490 2.7 ml/min/1.73 m
2
/year
Hemmelgarn B et al.
223
Males age 465 without diabetes 2475 1.4 ml/min/1.73 m
2
/year
Hemmelgarn B et al.
223
Females age 465 with diabetes 445 2.1 ml/min/1.73 m
2
/year
Hemmelgarn B et al.
223
Females age 465 without diabetes 3163 0.8 ml/min/1.73 m
2
/year
Keller C et al.
224
Cardiovascular Health Study 4128 1.83 ml/min/1.73 m
2
/year (based on
cystatin C-based eGFR)
Abbreviations: CrCl, creatinine clearance; GFR, glomerular filtration rate; MDRD, Modification of Diet in Renal Disease; PREVEND, Prevention of Renal and Vascular End-Stage
Disease.
64 Kidney International Supplements (2013) 3, 63–72
chapter 2

strips, consideration may be given to implementation of thi s
assessment in hig h-risk groups.
Implications for Clinical Practice and Public Policy
Practitioners must incorporate underlying category of
GFR and albuminuria as well as cause of kidney disease
and individual patient characteristics in determining the
frequency of ongoing assessment. The implications for
practice include the incorporation of regular monitoring
of both GFR and albuminuria into clin ical care for patients
with CKD.
There are no immediate implications for public policy of
this statement.
Areas of Controversy, Confusion, or Non-consensus
There are many who would like more definitive guidance on
frequency of measurement according to specific categories of
risk. However this is not possible at the current time given
the lack of evidence to guide such statements and the extreme
number of individual circumstances that would mitigate any
proposed protocol.
We recommend further research to more accurately define
the frequency with which GFR and albuminuria measure-
ments should be performed based on their ability to inform
strategies which prevent adverse outcomes (e.g., progression
of kidney disease and death).
Clarification of Issues and Key Points
a) Assessment of both GFR and albuminuria should be
undertaken to evaluate progression.
b) More frequent assessment is required as kidney disease
progresses.
c) Not all individuals with CKD require close surveillance
and monitoring; clinical context remains an important
modifier for all recommendations.
d) While cause of CKD is an important predictor of
progression, it is the values of GFR and albuminuria
that are used to assess progression.
Pediatric Considerations
Curr ently there is no evidence as to the value of incr easing the
frequency of assessment of either GFR or proteinuria in
children with CKD. Eventually more complete longitudinal
data from the CKiD cohort
55
and hence better granularity of
individual and group rates of decline in GFR may provide the
data required to strengthen proof of this guideline in children.
Table 21 | Decline in kidney function in CKD populations
Study Study population N
Baseline GFR
ml/min/1.73 m
2
Mean Follow-up
years
GFR decline
Mean (SD) or
(95% CI) ml/min/1.73 m
2
/year
MDRD Study
Group
226
Study A: GFR 25-80 ml/min/1.73 m
2
28 Mean (SD)
37.1 (8.7)
1.2 3.7 (7.6)
Study B: GFR 7.5-24 ml/min/1.73 m
2
63 15.0 (4.5) 4.3 (4.7)
Klahr S et al.
227
Study 1: GFR 25-55 ml/min/1.73 m
2
Mean (SD) 2.2 years
- Usual protein, usual MAP 145 37.6 (9.0) 4.5 (3.7 – 5.3)
- Usual protein, low MAP 149 38.2 (8.6) 3.3 (2.5 – 4.1)
- Low protein, usual MAP 140 38.9 (8.8) 3.3 (2.5 – 4.2)
- Low protein, low MAP 151 39.7 (9.1) 2.3 (1.5 – 3.0)
Study 2: GFR 13-45 ml/min/1.73 m
2
- Low protein, usual MAP 62 18.7 (3.1) 4.9 (3.8 – 5.9)
- Low protein, low MAP 67 18.8 (3.3) 3.9 (3.2 – 4.7)
- Very low protein, usual MAP 61 18.3 (3.7) 3.6 (2.8 – 4.4)
- Very low protein, low MAP 65 18.4 (3.5) 3.5 (2.6 – 4.5)
Wright J et al.
228
African Americans with
hypertension and
GFR 20-65 ml/min/1.73 m
2
Mean (SD) 4 years Mean (SE)
- Low MAP 380 46.0 (12.9) 2.21 (0.17)
- Usual MAP 374 45.3 (13.2) 1.95 (0.17)
Eriksen B
229
GFR categories G3a and G3b
(GFR 30–59 ml/min/1.73 m
2
)
3047 Median (IQR)
55.1 (50.8 – 57.9)
Median 3.7 years Mean
1.03 ml/min/1.73 m
2
/year
Jones C et al.
230
Nephrology referrals with
GFR categories G3a-G5
(GFRo60 ml/min/1.73 m
2
)
726 Median (IQR)
29 (18-38)
Median (IQR)
2.9 years (1.3 – 4.1)
Median
0.35 ml/min/1.73 m
2
/year
Levin A et al.
231
Nephrology referrals with
GFR categories G3a-G5
(GFRo60 ml/min/1.73 m
2
)
4231 Median
33 ml/min/1.73 m
2
Median (IQR)
2.6 years
(1.6-3.6)
Mean
2.65 ml/min/1.73 m
2
/year
Abbreviations: CI, confidence interval; CKD, chronic kidney disease; GFR, glomeru lar filtration rate; IQR, interquartile range; MAP, mean arterial pressure; MDRD, Modification of
Diet in Renal Disease; SD, standard deviation; SE, standard error.
Kidney International Supplements (2013) 3, 63–72
65
chapter 2

As described in detail in the Pediatric Considerations for
Recommendation 1.3.1, there is good observational evi-
dence acknowledging the importance of level of GFR and
proteinuria at baseline on predicting rates of progression and
it may be that interventional opportunities would exist if
closer monitoring and earlier recognition of worsening in
these values for the individual were available. Future studies
may examine this.
2.1.2: Recognize that small fluctuations in GFR are
common and are not necessarily indicative of
progression. (Not Graded)
2.1.3: Define CKD progression based on one of more of
the following (Not Graded):
K Decline in GFR category (Z90 [G1], 60-89
[G2], 45-59 [G3a], 30-44 [G3b], 15-29 [G4],
o15 [G5] ml/min/1.73 m
2
). A certain drop in
eGFR is defined as a drop in GFR category
accompanied by a 25% or greater drop in eGFR
from baseline.
K Rapid progression is defined as a sustained decline
in eGFR of more than 5 ml/min/1.73 m
2
/yea r.
K The confidence in assessing progression is
increased with increasing number of serum
creatinine measurements and duration of
follow-up.
RATIONALE
This statement serves to inform clinicians that some consistent
definition of progression is required and should be imple-
mented based on an appreciation of baseline values for an
individual. There is considerable controversy as to what
constitutes normal progression of CKD. The potential for
biological and analytical variation associated with use of SCr
measurements should be taken into account as they represent
reversible fluctuations in GFR and are not necessarily
indicative of progression. Further, it is important to recognize
that the degree of precision with which progression is able to
be estimated is highly dependent upon two factors: the
number of SCr measurements used to define progression and
the duration of follow-up. Estimating risk of ESRD based on
extrapolation of the previous rate of change in GFR required
substantial information (Z4 measurements over Z3 years
in most cases) to add to the risk information in the most
recent GFR. Further, it should be recognized that some
reno-protective treatments (e.g., BP lowering and RAAS
antagonists) result in a slower rate of GFR decline long-
term but often lower GFR in a stepwise fashion as a result of
hemodynamic effects. Even substantial (5-25%) reductions in
GFR may be protective, complicating the interpretation of
progression in these individuals. Finally, underlying disease
activity should be considered when assessing patients for
progression of kidney dysfunction.
The importance of determining the rate of decline in
kidney fun ction over time is to identify individuals who are
progressing at a more rapid rate than anticipated, which is
associated w ith increased morbidity and mortality. Indivi-
duals who are ‘‘rapid progressors’’ should be targeted to
slow their progression and associated adverse outcomes.
A progressive decline in kidney function is influenced by
baseline GFR category and albuminuria category.
Evidence Base
Unfortunately few studies are available to guide us regarding
the optimal definition of ‘‘rapid progression.’’ Such studies
require serial follow-up of patients to calculate change in
GFR over time, with more frequent measurements and longer
duration of follow-up providing more accurate estimates.
The Work Group reviewed cohort stud ies of the general
population that have evaluated rapid progression of kidney
function (Table 22).
Approaches to define decline in kidney function included
absolute rate of loss
232,233,235
as well as percent change.
221,234
Studies consistently demonstrate that a more rapid rate of
loss of kidney function was associated with an increased risk
of adverse clinical outcomes including death and vascular
related events. These studies have been limited however by
relatively few patie nts with GFR levelso60 ml/min/1.73 m
2
,
few measurements of SCr, and relatively short duration of
follow-up to obtain accurate estimates of the rate of decline
in kidney function. The precision of the estimate of the slope
depends on a num ber of factors including the number of
measurements of kidney function, biological variability,
measurement error, and duration of follow-up. In general
at least three measures of kidney function are required to
permit an estimate of slope.
1
None of these studies assessed the impact of albuminuria
on rate of ‘‘rapid decline’’ in kidney function. However as
noted in Recommendation 2.1.1, the presence of proteinuria
has been associated with a faster rate of kidney function
decline compared with people without proteinuria.
236–238
Two of the largest prospective cohort studies have shown an
approximate two-fold increase in the rate of decline in GFR
in the presence of proteinuria.
219,220
Further evidence
regarding the potential adverse effects of albuminuria on
outcomes has been reported among patients with diabetes.
The AER is one of the best indicators of diabe tic nephropathy
risk in both type 1 and type 2 diabetes, and patients with
microalbuminuria have been reported to have 200 to 400%
higher risk for progression to proteinuria than patients with
normal albuminuria.
239,240
Long-term follow-up studies have
also demonstrated the increased risk of ESRD associated with
albuminuria among patients with both type 1 and type 2
diabetes.
241,242
Given the recognized limitations in defining rapid
progression, the Work Group aimed to provide options for
determination of progression, based on their clinical utility,
and ease of use. One approach included an assessment of
change in GFR category, combined with a minimal percent
change. A criterion requiring both a change in GFR category
(e.g., change from G2 to G3a) and percent change would
66 Kidney International Supplements (2013) 3, 63–72
chapter 2

Table 22 | Studies evaluating rapid progression (general population studies only)
Study Study population N
Categories for decline in
kidney function Outcome Follow-up Results (95% CI)
Al-Aly Z et al.
232
Veterans Affairs
- GFR categories G3a
and G3b (GFR 30–59
ml/min/1.73 m
2
) with
Z2eGFR
measurements
4171 No decline: eGFR 0 ml/min/yr
Mild decline: 0 to 1
Moderate decline: 1 to 4
Severe decline: 44
Death 5.7 yrs (median) HR (multivariate):
No decline: 1.15 (0.99-1.24)
Mild: Reference
Moderate: 1.10 (0.98-1.30)
Severe: 1.54 (1.30-1.82)
Shlipak et al.
233
Cardiovascular
Health Study
- Age 65þ
- eGFR
measurements at
baseline, years
3 and 7
4378 Rapid decline: eGFR
43 ml/min/1.73 m
2
/yr
Not rapid decline:
r3 ml/min/1.73 m
2
/yr
Incident:
HF
MI
Stroke
PAD
Subsequent
8 years after
enrollment in
year 7
HR (multivariate):
HF: 1.40 (1.20-1.65)
MI: 1.42 (1.14-1.76)
Stroke: 1.11 (0.89-1.37)
PAD: 1.67 (1.02-2.75)
Matsushita et al.
221
ARIC study
- eGFR
measurements at
baseline and 3 years
13,029 Quartiles of % annual
change in eGFR:
Q1 (-52.76 to -5.65)
Q2 (-5.65 to -0.47)
Q3 (-0.47 to -0.33)
Q4 (-0.33 to 42.94)
CHD & all
cause
mortality
Up to Jan 1, 2006
(Baseline 1987-89)
HR (multivariate):
CHD:
Q1: 1.30 (1.11-1.52)
Q2: 1.16 (1.00-1.35)
Q3: Reference
Q4: 1.04(0.90-1.22)
Mortality:
Q1: 1.22 (1.06-1.41)
Q2: 1.05 (0.92-1.21)
Q3: Reference
Q4: 1.10 (0.96-1.27)
Cheng et al.
234
Taiwanese civil
servant & school
teachers
7968 % decrease:
o20% decrease
Z20% decrease
All cause,
CVD,
CHD,
& stroke
mortality
Up to Dec 31,
2005
(Baseline 1989-94)
HR (multivariate):
All death: 1.45 (1.13-1.86)
CVD death: 2.48 (1.58-3.89)
CHD death: 2.14 (1.07-4.29)
Stroke death: 2.79 (1.45-5.36)
Rifkin et al.
235
Cardiovascular
Health Study
- Age 65þ
- eGFR
measurements at
baseline, years
3 and 7
4380 Rapid decline: eGFR
43 ml/min/1.73 m
2
/yr
Not rapid decline:
o3 ml/min/1.73 m
2
/yr
All cause,
& CVD
mortality
Mean follow-up
9.9 yrs
HR (multivariate):
All death: 1.73 (1.54-1.94)
CVD death: 1.70 (1.40-2.06)
Abbreviations: ARIC, Atherosclerosis Risk in Communities; CHD, coronary heart disease; CI, confidence interval; CKD, chronic kidney disease; CVD, cardiovascular disease; eGFR, estimated glomerular filtration rate; HF, heart failure;
HR, hazard ratio; MI, myocardial infarction; PAD, peripheral arterial disease; yr, year.
Kidney International Supplements (2013) 3, 63–72 67
chapter 2

ensure that small changes GFR from 61 to 59 ml/min/1.73 m
2
for example, which represents a change in category but a
minimal change in GFR, would not be misinterp reted to
represent progression. A change of o25% in a pair of GFR
estimates may reflect physiologic variation rather than true
progression.
Additional work to inform this definition has been
undertaken, using data from the Alberta Kidney Disease
Network (AKDN).
243
In this analysis 598,397 adults with at
least two out-patient measures of SCr spaced at least 6
months apart were included. Progression was defined as
‘‘certain’’ (rise or drop) if during the median follow-up time
of 2.4 years there was a change in GFR category combined
with a 25% or greater change in GFR from the baseline
measurement (constituting a certain rise or a certain drop).
Participants who changed GFR category but did not meet the
criterion of 25% change in GFR were categorized as
‘‘uncertain’’ rise or drop. The reference group was comprised
of participants who did not change GFR category over the
follow-up period. As outlined in Table 23, compared to
participants with stable eGFR, those w ith a certain drop had
an almost two-fold increase in the risk of all-cause mortality
(HR 1.89; 95% CI 1.83–1.95)
244
and a five-fold increase in the
risk of ESRD (HR 5.11; 95% CI 4.56–5.71).
245
Lesser degree
of risk was present for those with an uncertain drop
(reflecting a change in category only). It is worth noting
that once progression occurs, the last eGFR which has a lower
level often contains much of the information about risk of
ESRD and extrapolation of progression using information
from prior progression and the most recent eGFR is only
useful if the information about progression is precise and the
patient’s trajectory is linear.
The second approach to define progression takes into
account the rate of change in kidney function based on a
slope analysis. In this approach the rate of loss is defined by
both the absolute rate of change and the percent change, as
determined among a cohort of 529,312 adults who had at
least 3 outpatient SCr measurements over a four year period
(AKDN databases). Two indices of change in eGFR were
estimated: the absolute annual rate of change (categorized as:
increase, stable and -1, -2, -3, -4, and Z -5 ml/min/1.73 m
2
/
year decline); and the annual percentage change (categorized
as: increase, stable, -1 to -2, -3 to -4, -5 to -6, and Z-7
percent decline/year). The adjusted ESRD risk associated
with each category of change in eGFR was estimated, using
stable eGFR (no change in eGFR) as the reference. The results
were adjusted in two ways: for eGFR and covariates at the
time of the first eGFR measurement, and at the time of the
last eGFR measurement. As outlined in Table 24, the risk of
ESRD increased almost two-fold for every 1 ml/yr decline in
eGFR, when adjusted for covariates and eGFR at the time of
the first eGFR measurement. The risk remained significant,
but was less pronounced, when adjus tments were performed
at the time of the last eGFR measurement. This suggests
that extrapolation of kidney function beyond the last
measurement of eGFR is still informative, but identifies a
lesser risk. Similar results were obtained when change in
eGFR was defined by a percentage.
Table 23 | CKD progression and risk of all-cause mortality and
ESRD using baseline (first) eGFR
Definition of
progression
All-cause mortality
HR** (95% CI)
ESRD*
HR** (95% CI)
Certain rise 1.51 (1.46–1.56) 0.33 (0.26–0.42)
Uncertain rise 1.12 (1.08–1.16) 0.39 (0.30–0.51)
Stable (reference) Ref Ref
Uncertain drop 0.98 (0.95–1.01) 2.13 (1.84–2.47)
Certain drop 1.89 (1.83–1.95) 5.11 (4.56–5.71)
Abbreviations: CI, confidence interval; eGFR, glomerular filtration rate; ESRD, end-
stage renal disease; HR, hazard ratio.
Data from Turin et al.
244,245
*ESRD defined as requiring renal replacement therapy.
**Adjusted for age, gender, hypertension, diabetes, proteinuria, Charlson comor-
bidities and baseline (first) eGFR.
Table 24 | Association between absolute and percentage change in kidney function and risk of ESRD*, based on adjustment for
eGFR at the first and last measurement
Absolute rate of change
(over a median of 3.5 years)
Adjusted for eGFR at first
creatinine measurement HR** (95% CI)
Adjusted eGFR at last creatinine
measurement HR** (95% CI)
Increasing eGFR 0.64 (0.48–0.86) 1.20 (0.90–1.61)
Stable (0 ml/min/1.73 m
2
/year) Ref Ref
-1 ml/min/1.73 m
2
/year 2.05 (1.56–2.69) 1.45 (1.11–1.90)
-2 ml/min/1.73 m
2
/year 2.71 (2.08–3.53) 1.58 (1.21–2.06)
-3 ml/min/1.73 m
2
/year 3.98 (3.06–5.17) 1.63 (1.25–2.13)
-4 ml/min/1.73 m
2
/year 5.82 (4.45–7.61) 1.90 (1.45–2.48)
-5 ml/min/1.73 m
2
/year or more 12.49 (10.04–15.53) 1.70 (1.36–2.12)
Percentage Change:
Increasing 0.76 (0.55–1.07) 1.11 (0.80 – 1.55)
Stable Ref Ref
-1 to -2%/year 1.17 (0.81–1.68) 0.97 (0.67–1.40)
-3 to -4%/year 1.79 (1.25–2.56) 1.19 (0.83–1.71)
-5 to -6%/year 2.26 (1.55–3.29) 1.21 (0.83–1.78)
-7%/year or more 11.30 (8.53–14.97) 2.17 (1.60–2.93)
Abbreviations: CI, confidence interval; eGFR, estimated glomerular filtration rate; ESRD, end-stge renal disease; HR, hazard ratio.
Data from the Alberta Kidney Disease Network.
*ESRD defined as requiring renal replacement therapy.
**Adjusted for age, gender hypertension, diabetes, proteinuria, Charlson comorbidities and eGFR at the first or last measurement.
68 Kidney International Supplements (2013) 3, 63–72
chapter 2

With respect to the impact of changes in albuminuria over
time, a study from the Ongoing Telmisartan Alone and in
Combination with Rami pril Global Endpoint trial (ONTAR-
GET) investigators showed that a greater than or equal to
twofold increase in albuminuria from baseline to 2 years
associated with a nearly 50% higher mortality (HR 1.48; CI
1.32-1.66), while a greater than or equal to twofold decrease
in albuminuria associated with 15% lower mortality (HR
0.85; CI 0.74-0.98) compared with those with lesser changes
in albuminuria, after adjustment for baseline albuminuria,
BP, and other potential confounders. Increases in albuminur-
ia also significantly associated with renal outcomes, defined
as ESRD or doubling of SCr (HR 1.40; CI 1.11-1.78), while a
decrease in albuminuria was associated with a decrease of the
combined renal outcome (HR 0.73; CI 0.57-0.95).
246
However, preliminary analysis of cohort studies is limited
and sug gests that further research is required to more
accurately determine the change in albuminuria associated
with an increased risk of kidney disease progression.
There is accumulating evidence that the trajectories of
GFR decline are non-linear, and may take a number of
different courses over time. The longer an individual is
followed over time, the more likely they are to experience
non-linear change in trajectory.
247,248
The non-linearity
of a trajectory may be due to intercurrent illness, changes
in medication, intrinsic to the disease process, or any
combination of these factors. Li et al.
247
described individual
GFR progression trajectories over twelve years of follow-up
among participants in the African American Study of Kidney
Disease (AASK) (Figure 18). The authors reported that
41.6% of patients exhibited a greater than 90% probability
of hav ing a non-linear trajectory; in 66.1% the probability of
non-linearity was 450%.
International Relevance
Studies to date evaluating rapid progression of kidney disease
have been limited to North American (White and African
American), European, and Asian populations. Given the
differences in the prevalence of CKD by ethnic group, there
may also be variations in rate of progression by ethnicity, and
in particular ethnic groups with high rates of comorbid
conditions leading to ESRD such as the Aboriginal popula-
tion. Thus, the definition of rapid progression may vary
according to country or region. However, by using a general
definition of progression, which includes change of category
of eGFR or albuminuria or both, as well as a numeric change
over an established period of time, we believe that the
definition of progression can be used in all cohorts.
Implications for Clinical Practice and Public Policy
Practitioners should monitor the GFR and albuminuria at
regular intervals to identify rates of decline which exceed that
normally demonstrated. The rate of GFR decline may be
relatively constant over time in an individual; however the
rate of GFR decline is highly variable among people and over
long periods of observation, within individuals. Thus
clinicians are encouraged to evaluate changes in GFR or
albuminuria in the context of multiple observations over
time, and with attention to clinical events which may have
impacted the change. A number of factors influence
assessment of rate of progression including frequency and
duration of GFR and albuminuria measurements as well as
Figure 18 | Distribution of the probability of nonlinearity (top) with three example trajectories demonstrating different
probabilities of nonlinearity (bottom). Reprinted from Li L, Astor BC, Lewis J, et al. Longitudinal Progression Trajectory of GFR
Among Patients With CKD. Am J Kidney Dis 2012; 59: 504-512 with permission from the National Kidney Foundation.
247
; accessed
http://download.journals.elsevierhealth.com/pdfs/journals/0272-6386/PIIS0272638611017896.pdf
Kidney International Supplements (2013) 3, 63–72 69
chapter 2

factors related to the patient (e.g., baseline GFR, comor-
bidities, age etc.).
There are no implications for public policy at this time.
Areas of Controversy, Confusion, or Non-consensus
The practical issue in clinical practice and clinical trials is
how to define progression (as inferring true deterioration in
kidney function) with meaningful thresholds that are easy
to understand for the non-nephrologist. While changes in
proteinuria may signify change in clinical condition, there are
no data yet to suggest that change in proteinuria is itself
reliably associated with progression of CKD per se . This may
be confusing to practitioners, since a change in quantity of
proteinuria is an indication for referral.
We recommend research to confirm rates which can be
classified as slow, moderate, and rapid progression of kidney
disease. The rate to define ‘‘rapid progression’’ may vary
depending on the outcome considered, such as kidney failure
versus mortality for example. It will be important for
researchers to determine methods by which reproducible
classification systems for describing rates of progression can
be developed. There are increasing data to suggest the non-
linearity of progressive disease in many individuals. This
makes extrapolation risky and warrants continued assessment
of the slope on a regular basis.
Clarification of Issues and Key Points
Small fluctuations in GFR are common and should not be
misinterpreted to represent progressive decline in kidney
function. Many factors can cause a small change in GFR
including medications, volume status, measurement error,
and biological variability. Assessment of progressive decline
in GFR needs to take into account the number of
measurements considered and time period of assessment.
In pediatrics, information about utilty of serial creatinine
measurements over periods of time during which growth
(and muscle mass increase) is occurring, for the diagnosis of
progression or regression, remains problematic.
Pediatric Considerations
Applying strict GFR criteria in order to develop cutoff values
associated with ‘true’ progression in terms of any one
individual child is not currently possible. Conceptually the
movement from various levels of renal function downward,
in particular if that movement is associated with increasing
comorbidities or intensity of such, is a reasonable approach.
The most informative data available in children comes from
the longitudinal GFR data from CKiD .
72
Examination of the
whole cohort reveals an annual decline in GFR of 4.2%;
median GFR decline was 1.8 ml/min/1.73 m
2
(interquartile
range [IQR] 6.6 to 1.6); this can be expressed as a median
absolute decline in GFR of 4.3 ml/min/1.73 m
2
(IQR 11.9 to
1.1) and 1.5 ml/min/1.73 m
2
(IQR 5.0 to1.8).
Given that the lower IQR in each of the overall cohort and
both sub-groups is equal to or exceeds the suggested decl ine
of 5 ml/min/1.73 m
2
as stated here, we suggest that it is
reasonable to adopt this definition at least for the purpose of
classification as it relates to ‘rapid’ progression; note the
above values all relate to measured GFR.
Increasing numbers of any given measurement of an event
generally al low for greater precision and accuracy. However,
the simple repeated measurements of creatinine over time are
less likely to be valuable in children than in adults with CKD.
Unlike adults with static muscle mass and hence expected
stability in creatinine values, or adults with expected
declining muscle mass and hence expectations of declining
creatinine if renal function has remained stable, pediatric
populations have a situation of increasing muscle mass with
expectation of increasing creatinine in the otherw ise normal
child without CKD. In a child with CKD who is growing
therefore, and in particular one going through puberty, the
simple comparison of creatinine values over time will likely
not be sufficient to presume CKD progression or regression
has occurred. The two exceptions to this would be a) a series
of creatinine measurements demonstrating significant
increase over a short period of time wherein there is no
demonstrable or expected gain of muscle mass; b) values of
creatinine that over time demonstrate an increase to levels
above that which is expected of the child’s age and sex based
on population normative value for the lab and method of
measurement.
2.1.4: In people w ith CKD progression, as defined in
Recommendation 2.1.3, review current manage-
ment, examine for reversible causes of progression,
and consider referral to a specialist. (Not Graded)
RATIONALE
This statement intends to reassure patients and practitioners
that not all patients necessarily require referral to specialists,
but that this should be considered in the event that the
patient or clinician requires further guidance or prognostic
information. CKD progression, contextualized for the
individual circumstance, does not always require referral,
and earlier guidelines may not have been so overt in stating
this. Faster or unusual trajectories of progression should alert
the patient and physician to assess for potentially reversible
causes of progression.
Progressive kidney disease requires the need for more
aggressive assessment and treatment, which may include
referral to a nephrologist or specialist (if they are not
currently being managed by a nephrologist).
Evidence Base
Decline in GFR may not be constant, with acute decline
superimposed on CKD (see Chapter 2.2 for discussion of
factors associated with progression of a more chronic
nature). The most common risk factors identified for acute
decline in GFR for patients with established CKD include:
obstruction of the urinary tract; volume depletion; nonster-
oidal anti-inflammatory drugs (NSAIDs), including cyclo-
oxygenase-2 (COX 2) inhibitors; select antimicrobial agents
70 Kidney International Supplements (2013) 3, 63–72
chapter 2

such as aminoglycosides and amphotericin B; radiocontrast
agents; and angiotensin-converting enzyme inhibitors
(ACE-Is) and angiotensin-receptor blockers (ARBs).
249–252
Readers are also referred to the KDIGO Clinical Practice
Guideline for Acute Kidney Injury
7
which contains additional
relevant details regarding risk factors for rapid progression
and management strategies.
Rapid sustained decline in GFR could also be considered
an indication for referral once potentially reversible factors as
outlined above have been assessed and treated. The principles
guiding referral include additional input from a nephrologist
into management of CKD and preparation for RRT, such as
that which may be required with rapidly declining GFR. The
NICE guidelines for CKD also recommend referral for
‘‘rapidly declining GFR’’ although the definition of rapidly
declining is not provided.
186
Most studies assessing nephrol-
ogy referral have focused on early versus late referral, and not
considered the impact or implications of nephrology referral
in situations associated with rapid decline in GFR.
253
The
evidence that such referral will change outcomes is not clear
but given that nephrologists often have access to education
and specialized services, which are essential for optimal
preparation for RRT, referral to a specialist is recommended.
Additional discussion of when to consider a referral to a
nephrologist can be found in Chapter 5.
International Relevance
Risk factors for acute deterioration in kidney function may vary
slightly by country of origin, although the common categories
in general (e.g., medications, volume depletion and urinary
tract obstruction) would be relevant irrespective of country.
This set of statements serves to ensur e that attention to changes
in kidney function in those identified with kidney disease is part
of the usual care of these individuals.
Implications for Clinical Practice and Public Policy
Practitioner s should be aware of the common risk factors for
acute-on-chron ic kidney disease resulting in a rapid loss of
kidney function. Acute rapid deterioration in kidney function
should alert the practitioner to assess for these potentially
reversible causes of progression. This assessment would include
an evaluation for potential urinary tract obstruction as well as a
volume assessment and detailed medication review .
Areas of Controversy, Confusion, or Non-consensus
Readers are referred to the KDIGO Clinical Practice Guideline
for Acute Kidney Injury
7
for a detailed discussion of issues
related to definition, diagnosis, and treatment of rapid
progression of kidney dysfunction, as defined by AKI.
Clarification of Issues and Key Points
There are no specific numeric values to define ‘‘rapid loss’’ as
this varies within and between disease stat es, individuals, and
populations. The key point for practitioners is to serially
review individual trajectories of changes in kidney function
so as to determine stability or not, identify changes in
previous stable trajectories, and to be aware of the
importance of circumstances where AKI changes the course
of CKD. Further studies are required to determine the rate of
loss of GFR that constitutes rapid loss of kidney function. In
children, interpretation of serial changes in SCr requires
understanding of normative value ranges and drivers of
change in SCr which are different than in adults.
2.2: PREDICTORS OF PROGRESSION
2.2.1: Identify factors associated with CKD progres-
sion to inform prognosis. These include cause of
CKD, level of GFR, level of albuminuria, age,
sex, race/ethnicity, elevated BP, hyperglycemia,
dyslipidemia, smoking, obesity, history of
cardiovascular disease, ongoing exposure to
nephrotoxic agents, and others. (Not Graded)
RATIONALE
The rationale for estimating the risk of kidney failure is that
it may inform referral, care plans, and other therapeutic
strategies, including frequency of monitoring and follow-up.
Previous guidelines have not been able to suggest risk
equations or relative and absolute risks of specific outcomes
but with the data currently available, this is now possible.
There are several factors that influence the likelihood and rate
of CKD progression including GFR and albuminuria category,
the degree of albuminuria, the cause of kidney disease, on-
going exposure to nephrotoxic agents, obesity, hypertension,
age, race/ethnicity and laboratory parameters such as Hb
(hemoglobin), albumin, calcium, phosphate, and bicarbonate.
As some of these risk factors are modifiable they should be
actively identified and, if present, be treated as they may
impact long-term outcomes including cardiovascular condi-
tions, QOL, and progression of CKD.
It is not yet clear what the relative weight of each of these
factors is in predicting in an individual whether he/she will
have progressive CKD.
Evidence Base
As progression of CKD is defined as either a progressive
decrease in GFR or a progressive increase in albuminuria, we
should consider separately whether different factors would
predict these two components of CKD differently. Given the
limited evidence, this will not be discussed separately. It is
however clear that a subject with a lower GFR to start with
will progress more rapidly to a GFR o15 ml/min/1.73 m
2
,
just as a subject with already elevated albuminuria will
progress more rapidly to an ACR 4300 mg/g (430 mg/
mmol). Similarly, it is well-known that a subject with
membranous glomerulopathy is more likely to progress to
nephrotic syndrome, while a subject with adult polycystic
kidney disease is more likely to progress to ESRD.
Although there are many cross-sectional studies that describe
factorsassociatedwithalowGFRandfactorsassociatedwitha
high albuminuria, the number of studies evaluating which
Kidney International Supplements (2013) 3, 63–72 71
chapter 2

factors are associated with progressiv e decreases in GFR and
progressiv e increases in albuminuria are more limited. In
general, it can be argued that most of the above-mentioned
factors are associated with a more progressive rise in albuminuria
and a more progressive fall in GFR. Most recently , studies have
focused on the development of risk scores for identifying
progressiv e decreases in GFR and progressive increases in
albuminuria. It has not yet been established which prediction
formula could best be used. Some formulas use just simple
demographic and clinical measures, while others also include
laboratory tests. Some were developed for high-risk populations,
such as people with known underlying CVD ,
254
with CKD in
general,
255
or with specific causes of CKD , such as IgA
nephropath y,
256
diabetic nephropathy,
257
or renal artery steno-
sis.
258
Others developed a risk prediction model in the general
population.
259
This latter model included age, race, gender , and
in dichotomized version, the pr esence of anemia, hypertension,
diabetes, and CVD history. Mor e recently, two studies used more
accurate laboratory parameters in addition to demographic
characteristics. The first study was in patients with an eGFR of
15-60 ml/min/1.73 m
2
/year, and included age, gender , eGFR,
albuminuria, and serum calcium, phosphate, bicarbonate and
albumin.
260
The second study was in subjects from the general
population and included age, eGFR, albuminuria, measured
levels of BP and C-reactive protein (CRP).
261
The results from
these predictive models require validation in future studies but
they demonstrate the potential and the capabilities of developing
clinically meaningful classification of risk for individual patients.
Further research is required to establish whether prediction
formulas may differ for different ethnicities.
International Relevance
Studies describing factors asso ciated w ith lower GFR and
higher ACR have been described from all over the world. In
general, there is much overlap between these data. It may be
that in different parts of the world the relative weight of each
of the factors predicting progressive increases in albuminuria
or decreases in GFR may substantially differ.
Implications for Clinical Practice and Public Policy
It is important to realize that some factors predicting
progression of CKD are modifiable. This holds true for
lifestyle measures such as cessation of smoking and preven-
tion of obesit y. It also subtends to lowering of BP, lowering of
albuminuria and prevention of hyperglycemia. A further
factor that may be modifiable is the underlying cause of
CKD. As various causes may respond to targeted treatment,
finding the cause of CKD is the starting point of the work-up
of a subject with CKD. If this causal disease is modifiable, for
example by immunosuppressive treatment, then such
treatment is the first step to consider. Management of
patients with CKD and delay of progression are dealt with in
Chapter 3 and more fully in other guidelines (see KDIGO
Clinical Practice Guideline for Glomerulonephritis
8
and
KDOQI Clinical Practice Guideline for Diabetes and CKD:
2012 Update.
262
)
Areas of Controversy, Confusion, or Non-consensus
Whiletherearepredictionequationsavailableusingcombina-
tions of eGFR, albuminuria, cause of CKD, and some or all of
the laboratory abnormalities listed, they have not been used in
clinical practice to guide therapy as yet. Furthermore, while the
associated abnormalities clearly increase in severity with
worsening kidney function, normalizing them in some instances
has not changed progression to ESRD . The need for prediction
equations to take into account changes over time (trajectories)
and stability or instability of specific factors has been raised by
many. Nonetheless, the ability to determine progress ion versus
stability should be of value for patients and clinicians.
Clarification of Issues and Key Points
Clinicians should attempt to determi ne stability or progres-
sion of patients with CKD for the purposes of informing care.
Further research is required to determine which formula best
predicts who will have progressive increases in albuminuria
and progressive decreases in GFR. The key components of
prediction equations for ESRD may well be different than
prediction equations for cardiovascular events or death.
Pediatric Considerations
A more complete discussion of the evidence in children
supporting these factors as potentially related to risk of
progression, in addition to the pediatric specific risk of
growth/puberty, can be found in the Pediatric Considerations
for Recommendation 1.3.1.
DISCLAIMER
While every effort is made by the publishers, editorial board,
and ISN to see that no inaccurate or misleading data, opinion
or statement appears in this Journal, they wish to make it
clear that the data and opin ions appearing in the articles and
advertisements herein are the responsibility of the contributor,
copyright holder, or advertiser concerned. Accordingly, the
publishers and the ISN, the editorial board and their respective
employers, office and agents accept no liability whatsoever for
the consequences of any such inaccurate or misleading data,
opinion or statement. While every effort is made to ensure that
drug doses and other quantities are presented accurately,
readers are advised that new methods and techniques
involving drug usage, and described within this Journal,
should only be followed in conjunction with the drug
manufacturer’s own published literature.
72 Kidney International Supplements (2013) 3, 63–72
chapter 2

Chapter 3: Management of progression and
complications of CKD
Kidney International Supplements (2013) 3, 73–90; doi:10.1038/kisup.2012.66
DELAYING PROGRESSION OF CKD
The following section describes key recommendations and
guidance for people with CKD with respect to delaying
progression of CKD. General lifestyle recommendations are
provided as well as caveats given for those with diabetes.
Cardiovascular risk reduction including management of
hypertension, dyslipidemia, and hyperuricemia is further
addressed. Unless otherwise stated, the guidance is intended
to apply to adults with CKD.
For the practicing clinician, ideally working with a team of
health-care professionals, it is important to institute general
lifestyle modification practices in people with CKD so that
they may gain the benefit of these in addition to more
kidney-specific strateg ies. Often these general measures are
overlooked or disregarded in people with CKD, thus their
utility is underscored here.
3.1 PREVENTION OF CKD PROGRESSION
The management of progression of CKD is aimed at
addressing a multiplicity of factors known to be associated
with progression. There are general measures which have
been shown to address cardiovascular health and CKD
together, or each separately. Addressing CVD risk factors may
indirectly and directly impact CKD progression. Strategies
include general lifestyle measures which improve cardio-
vascular health, BP control, and interruptio n of the RAAS.
In addition, control of other metabolic parameters such as
blood sugar, uric acid, acidosis, and dyslipidemia may also be
important. This section deals with management of BP, RAAS
interruption, glycemic control and dietary/lifestyle mani-
pulations which have been examined in the context of
delaying progression of CKD.
BP and RAAS interruption
The following statements are excerpted and where
necessary, condensed, from the KDIGO Clinical Practice
Guideline for the Management of Blood Pressure in CKD.
10
3.1.1: Individualize BP targets and agents according to age,
coexistent cardiovascular disease and other comorbid-
ities, risk of progression of CKD , presence or absence
of retinopathy (in CKD patients with diabetes), and
tolerance of treatment as described in the KDIGO
2012 Blood Pressure Guideline. (Not Graded)
3.1.2: Inquire about postural dizziness and check for
postural hypotension regularly when treating CKD
patients with BP -lo wering drugs. (Not Graded)
3.1.3: Tailor BP treatment regimens in elderly patients with
CKD by carefully considering age, comorbidities and
other therapies, with gradual escalation of treatment
and close attention to adverse events related to BP
treatment, including electrolyte disorders, acute
deterioration in kidney function, orthostatic h ypoten-
sion and drug side effects. (N ot Graded)
3.1.4: We recommend that in both diabetic and non-
diabetic adults with CKD and urine albumin
excretion o30 mg/24 hours (or equivalent*) whose
office BP is consistently 4140 mm Hg systolic or
490 mm Hg diastolic be treated with BP-lowering
drugs to maintain a BP that is consistently r140 mm
Hg systolic and r90 mm Hg diastolic. (1B)
3.1.5: We suggest that in both diabetic and non-diabetic
adults with CKD and with urine albumin excre-
tion of Z30 mg/24 hours (or equivalent*) whose
office BP is consistently 4130 mm Hg systolic or
480 mm Hg diastolic be treated with BP-lowering
drugs to maintain a BP that is consistently r130 mm
Hg systolic and r80 mm Hg diastolic. (2D)
3.1.6: We suggest that an ARB or ACE-I be used in dia-
betic adults with CKD and urine albumin excretion
30-300 mg/24 hours (or equivalent*). (2D)
3.1.7: We recommend that an ARB or ACE-I be used in
both diabetic and non-diabetic adults with CKD
and urine albumin excretion 4300 mg/24 hours
(or equivalent*). (1B)
3.1.8: There is insufficient evidence to recommend com-
bining an ACE-I with ARBs to prevent progression
of CKD. (Not Graded)
3.1.9: We reco m m e nd tha t in children with CKD, BP-
lowering treatment is started when BP is consistently
abov e the 90
th
percentile for age, sex, and height. (1C)
3.1.10: We suggest that in children with CKD (particularly
those with proteinuria), BP is lowered to consistently
achieve systolic and diastolicreadingslessthanor
equaltothe50
th
percentile for age, sex, and height,
unless achieving these targets is limited by signs or
symptoms of hypotension. (2D)
http://www.kidney-international.org chapter 3
& 2013 KDIGO
*Approximate equivalents for albumin excretion rate per 24 hours—
expressed as protein excretion rate per 24 hours, albumin-to-creatinine
ratio, protein-to-creatinine ratio, and protein reagent strip results— are given
in Table 7, Chapter 1
Kidney International Supplements (2013) 3, 73–90 73

3.1.11: We suggest that an ARB or A CE-I be used in children
with CKD in whom treatment with BP-lowering
drugs is indicated, irrespective of the level of protein-
uria. (2D)
These statements are worded to maintain consistency with
the KDIGO Clinical Practice Guideline for the Management of
Blood Pressure in CKD,
10
where the full rationale and
evidence behind the statements may be found. In detailing
BP targets, we recognize that we have not made recommen-
dations or suggestions concerning lower limits of BP. The
risks of overtreatment should be specifically considered when
making decisions about BP lowering and this is encapsulated
in the first two guideline statements.
CKD and risk of AKI
3.1.12: We recommend that all people with CKD are
considered to be at increased risk of AKI. (1A)
3.1.12.1: In people with CKD, the recommendations
detailed in the KDIGO AKI Guideline
should be followed for management of
those at risk of AKI during intercur rent
illness, or when undergoing investigation
and procedures that are likely to increase
the risk of AKI. (Not Graded)
RATIONALE
Observational data suggest a strong association between pre-
existing CKD and AKI. The appreciation that CKD patients
may be more susceptible to AKI is the purpose of the above set
of statements. However, methodological issues such as how
CKD and AKI are defined in clinical studies and the statistical
adjustments for non-uniformity of comorbidities among
various studies may affect the validity of observed associations.
These statements would be applicable in pediatrics, though the
data are not available for this specific issue.
Evidence Base
CKD is designated as a risk factor for AKI because of the
epidemiological association between the two.
263,264
A number of
studies in a variety of settings report an association between
pre-existing CKD and AKI.
265–271
CKD is a potent predictor of
acute decline in kidney function following exposure to radio-
contrast,
272
major surgery ,
273
and other medical conditions.
274
Hsu et al.
14
compared the pre-hospitalization MDRD GFR
of 1764 adult members of the Kaiser Permanente Northern
California health-care system who developed dialysis-requir-
ing AKI during hospitalization with 600,820 individuals who
did not. Compared with a reference baseline GFR of Z60 ml/
min/1.73 m
2
, a baseline GFR of 45–59 ml/min/1.73 m
2
was
associated with an adjusted odds ratio (OR) of in-hospital
AKI of 1.66 (95% CI 1.40–1.97). For GFR values of 15–29 ml/
min/1.73 m
2
, the adjusted OR for in-hospital AK I was 20.42
(95% CI 17.40–23.96). The presence of diabetes, hyperten-
sion, and proteinuria increased the likelihood of developing
in-hospital AKI, with adjusted OR of 1.99 (95% CI
1.78–2.23), 1.55 (95% CI 1.37–1.76) and 2.84 (95% CI
2.52–3.19), respectively. The authors concluded that CKD is
the main risk factor for AKI during hospitalization. A
contrasting approach by Singh et al. defined AKI as dialysis-
requiring acute renal failure.
275
Because the clinical decision
to dialyze a patient is frequently influenced by a higher
overall SCr, presence of hemodialysis access, or consideration
of inevitable progression to ESRD, this definition of AKI
could bias toward capturing more AKI cases in CKD patients.
Moreover, in patients with advanced CKD, the progression of
CKD to ESRD may sometimes be difficult to separate from
acute-on-chronic renal failure. A cohort study by Lafrance
et al. followed a referred CKD population in British
Columbia for a median of 19.4 months after achieving a
GFR of r30 ml/min/1.73 m
2
. Forty-five percent had at least
one episode of AKI.
276
In another cohort study of 920,985
adults in Alberta, Canada with at least one outpatient
measurement of SCr and proteinuria and not requiring
chronic dialysis, risk of admission with AKI increased with
heavier proteinuria and reduced GFR.
16
International Relevance
The incidence of AKI in CKD populations may be different
around the world, or have different etiologies. It is not yet
clear what the recovery rates from AKI are in the CKD
population, and how these vary around the world dependent
on cause and duration of AKI.
Areas of Controversy, Confusion, or Non-consensus
Interpretation of published data examining the influence
of pre-existing CKD on the increased likelihood of AKI
is potentially confounded by a number of issues. These
include the comorbidities associated with CKD, influenced
by repeated exposure to various nephrotoxic insults or
in-hospital errors,
57,277
or primarily due to the altered
physiology in CKD. There are also methodological issues
such as how CKD and AKI are defined in clinical studies and
the varying statis tical adjustments for comorbiditie s.
A further important issue to clarify is whether pre-existing
CKD influences the outcome of AKI. Currently, there is no
single biomarker that can differentiate ‘acute’ from ‘chronic’
kidney disease and help to address this issue. Several large
observational and database studies report, surprisingly, lower
in-hospital mortality in patients w ith AKI superimposed on
CKD compared with controls.
278–283
Data from the Program
to Improve Care in Acute Renal Disease (PICARD) reveal
lower in-patient mortality and median length of stay in
intensive-care unit (ICU) subjects with acute-on-chronic
renal injury compared with non-CKD subjects with AKI,
though the post-discharge dialysis rates were higher in
subjects with pre-existing CKD.
284
Clarification of Issues and Key Points
AKI is relatively common in CKD populations and impacts
progression adversely. Clinicans should attempt to minimize
74 Kidney International Supplements (2013) 3, 73–90
chapter 3

avoidable episodes of AKI (see Chapter 4 for more details) as
part of a holistic approach to delaying progression.
RESEARCH RECOMMENDATIONS
Prospectively designed clinical studies with a clear and
uniform definition of CKD and AKI and adjusted for
comorbidities are needed to determine the:
K frequency of AKI events in a CKD population;
K outcome of AKI in patients with CKD;
K importance of proteinuria in addition to low GFR in the
risk of AKI.
Protein intake
3.1.13: We suggest lowering protein intake to 0.8 g/kg/day
in adults with diabetes (2C) or without diabetes
(2B) and GFR o30 ml/min/ 1.73 m
2
(GFR cate-
gories G4-G5), w ith appropriate education.
3.1.14: We suggest avoiding high protein intake (41.3 g/
kg/day) in adults with CKD at risk of progres-
sion. (2C)
RATIONALE
These statements are worded to reflect the potential benefits
and dangers of varying dietary protein intake (DPI) in people
with CKD. Excess dietary protein leads to the accumulation
of uremic toxins, conversely insufficient protein intake may
lead to loss of lean body mass, and malnutrition (the latter
more frequent in the elderly). The benefits of dietary protein
restriction include reduction of accumulation of metabolic
waste products that may suppress the appetite and stimulate
muscle protein wasting. The role of dietary protein restric-
tion in slowing progression of CKD is more controversial and
advanced CKD is associated with a protein wasting syndrome
which is directly correlated with morbidity and mortality.
Note that statements about reduction in dietary protein do
not apply to pediatric populations given issues related to
growth and nutrition.
Evidence Base
A number of systematic reviews and meta-analyses have
pooled the available RCT data.
285–289
Pedrini et al.
288
compared a low-protein diet (LPD), defined as a DPI of
0.4 to 0.6 g/kg/day, w ith a usual diet (5 RCTs, N ¼ 1413) over
a period of follow-up ranging between 18-36 months in
people with non-diabetic CKD and GFR o55 ml/min/1.73 m
2
.
Fouque et al.
285
updated this analysis to include 8 RCTs
in people with non-diabetic CKD (N ¼ 1524). DPI in
their low-protein group was between 0.3-0.6 g/kg/day and
follow-up ranged from 12-24 months (5 of 8 studies were in
people with GFR categories G4-G5 (GFR o30 ml/min/
1.73 m
2
). Roberston et al.
289
compared diabetic subjects
(8 studies in type 1 diabetes, N ¼ 322; 1 study in t ype 2
diabetes, N ¼ 263). DPI in the low-protein subjects was
0.3-0.8 g/kg/day and usual protein intake ranged from 1-2
g/kg/day. Mean follow-up ranged from 4.5 months to 4 years.
In all studies, compliance with a low DPI was poor. There was
no convincing or conclusive evidence that long-term protein
restriction delayed the progression of CKD.
The largest RCT to date was the MDRD Study.
227
The
MDRD Study compared the effects of LPD and BP control on
the progression of CKD in over 800 subjects split into
2 groups. Study A compared a DPI of 1.3 g/kg/day (usual
protein intake) with 0.58 g/kg/day (LPD) in 585 subjects with
a measured GFR of 25-55 ml/min/1.73 m
2
and the actual
DPIs were 1.11 and 0.73 g/kg/day, respectively. Study B
randomized 255 patients with a measured GFR 13-24 ml/
min/1.73 m
2
to DPIs of 0.58 g/kg/day (LPD) or 0.28 g/kg/day
supplemented by keto-aminoacids (denoted by VLPD-KA),
actual DPIs were 0.69 and 0.46 g/kg/day, respectively. In each
of the randomization groups ACE-Is were allowed and were
used by 32-44% of patients. Mean follow-up was 2.2 years
and the loss of GFR was estimated by the slope of
125
I-iothalamate clearance measured over 2 years. There was
no difference in GFR decline between groups in Study A and
in Study B. Although there was a somewhat faster decline in
GFR in the LPD group compared with the VLPD-KA group,
this was not significant.
A follow-up study of the original MDRD Study followed
those subjects recruited to Study B between 1989-1993 up
until the year 2000. Median duration of follow-up until
kidney failure, death, or administrative censoring was 3.2
years and median time to death was 10.6 years.
290
The
authors concluded that assignment to a very LPD did not
delay progression to kidney failure, but appeared to increase
the risk of death in the long-term. The chief limitation of this
follow-up study was the lack of measurements of DPI and
nutritional measurements during the course of the long-term
follow-up period and it is therefore not known how many
patients continued with the LPD or the VLPD-KA diets
after the study concluded.
There is some evidence to suggest that higher protein
diets above the recommended daily intake may accelerate
renal functional decline in people with early CKD. In a
study of 1624 women enrolled in the Nurses
0
Health Study,
Knight et al. described the effe ct of protein intake over an
11-year period in women with eGFR Z80 ml/min/1.73 m
2
(normal renal function) at baseline and those with eGFR
55-80 ml/min/1.73 m
2
.
291
DPI was measured twice during
the study period at intervals of 4 years using a semiquanti-
tative food-frequency questionnaire that inquired about
the average intake of specified foods and beverages during
the previous year. In women with normal renal function
at baseline high protein intake was not significantly
associated with change in eGFR. However in those with
eGFR 55-80 ml/min/1.73 m
2
at baseline, protein intake
was significantly associated with a change in eGFR of
1.69 ml/min per 1.73 m
2
(95% CI, 2.93 to 0.45 ml/
min per 1.73 m
2
) per 10 g increase in protein intake.
The effect was greatest in those with the highest intake of
non-dairy animal protein.
Dietary protein restriction of o0.80 g/kg/day appears to
offer no advantage and any dietary protein restriction should
Kidney International Supplements (2013) 3, 73–90 75
chapter 3

include careful monitoring of clinical and biochemical
markers of nutritional deficiencies. A high total protein
intake, particularly high intake of non-dairy animal protein,
may accelerate renal function decline in people with CK D
and should be avoided.
International Relevance
Studies on protein restriction have not been widely tested in
different ethnicities or within cultures with low baseline
proten intake or purely vegetarian diets. Thus, the applic-
ability of statements to all regions of the world is limited.
Implications for Clinical Practice and Public Policy
Clinicians should be aware of different sources of protein, and
if lowering of protein is recommended, education and
monitoring for malnutrition should be implemented. Appro-
priate dietary counseling for CKD patients may have health-
care resource implications, although as part of a combined
strategy to manage obesity, salt intake, and diabetes may be
considered cost-effective on a population basis in certain
countries. Avoidance of malnutrition is important.
Areas of Controversy, Confusion, or Non-consensus
Data are mixed on the value of protein restriction, the values
which are achievable in general populations, and the level of
GFR at which they should be instituted. Nonetheless, the
Work Group felt that on balance there is enough data to
support a reduction in dietary protein in selected individuals.
It is important to avoid this advice in those with evidence of
or at risk of malnutrition.
Pediatric Considerations
A Cochrane review addresses this issue in children
292
by
examining two RCTs with a total of 250 children to
determine the effect of protein restriction on a number of
variables. The RR of progression to ESRD in the low-protein
restricted versus normal group was 1.12 (95% CI 0.54-2.33).
At two years, progression of kidney disease was not
significant as measured by change in CrCl: mean difference
1.47 ml/min/1.73 m
2
(95% CI 1.19–4.14) or growth as
measured by mean weight difference: 0.13 kg (95% CI
1.10–0.84) or mean heig ht diffe rence: 1.99 cm (95% CI
4.84–0.86). The conclusion of the authors was that a low-
protein diet did not delay progression to kidney failure in
children, but it may be detrimental to growth.
Glycemic control
Diabetes is the leading cause of CKD worldwide. Diabetic
nephropathy occurs in 25–40% of patients with type 1 or type
2 diabetes within 20–25 years of disease onset and is an
independent risk factor for early death due to CVD. The
mortality rate in people with diabetes and urinary
ACR 430 mg/g (43 mg/ mmol) is more than twice that in
those with normal urinary albumin levels.
The National Kidney Foundation (NKF) KDOQI Clinical
Practice Guideline for Diabetes and CKD
293
has been updated
in 2012. The first three recommendations below are
reproduced verbatim from this guideline.
3.1.15: We recommend a target hemoglobin A
1c
(HbA
1c
)
of B7.0% (53 mmol/mol) to prevent or delay
progress ion of the microvascular complications of
diabetes, including diabetic kidney disease. (1A)
3.1.16: We recommend not treating to an HbA
1c
target of
o7.0% (o53 mmol/mol) in patients at risk of
hypoglycemia. (1B)
3.1.17: We suggest that targ et HbA
1c
be extended above
7.0% (53 mmol/mol) in indiv iduals with comor-
bidities or limited life expectancy and risk of
hypoglycemia. (2C)
3.1.18: In people with CKD and diabetes, glycemic control
should be part of a multifactorial intervention
strategy addressing blood pressure control and
cardiovascular risk, promoting the use of angio-
tensin-converting enzy me inhibition or angio-
tensin receptor blockade, statins, and antiplatelet
therapy where clinically indicated. (Not Graded)
RATIONALE
These statements are included to reflect the current evidence
that achieving a hemoglobin A
1c
(HbA
1c
) level of B7.0%
(53 mmol/mol) is able to prevent the microvascular compli-
cations of diabetes, although recognizing that the major risk
for patients attaining HbA
1c
levels o7.0% (o53 mmol/mol)
is hypoglycemia, and that this risk will be higher in people
with lower levels of kidney function.
Evidence Base
The evidence base for these statements is reviewed in the
NKF KDOQI Clinical Practice Guideline for Diabetes and
CKD: 2012 Update
293
and will not be reiterated in full here. It
should be noted that the evidence that intensive glycemic
control reduces the microvascular complications of diabetes
is based almost exclusively on prevention of development of
albuminuria (ACR 430 mg/g or 43 mg/mmol) and preven-
tion of increasing albuminuria. Evidence from the three most
recent studies, Action in Diabetes and Vascular Disease:
Preterax and Diamicron Modified Release Controlled Evalua-
tion (ADVANCE),
294
Action to Control Cardiovascular Risk
in Diabetes (ACCORD),
295
and the Veterans Affairs Diabetes
Trial (VADT) ,
296
is summarized in Table 25.
ADVANC E, ACCORD, or VADT did not show significant
benefits of more intensive glycemic control on creatinine-
based estimates of GFR. However, in the Diabetes Control
and Complications Trial (DCCT)/Epidemiology of Diabetes
Interventions and Complications (EDIC) follow-up study,
1.4% of participants in the previously intensive treatment
group and 3.6% of those in the previously conventional
treatment group developed SCr concentrations 42.0 mg/dl
(177 mmol/l) (P ¼ 0.01) and 0.6% versus 1.9% required
kidney replacement therapy (P o0.03).
297
For patients with
type 2 diabetes, intensive treatment in the United Kingdom
76 Kidney International Supplements (2013) 3, 73–90
chapter 3

Prospective Diabetes Study (UKPDS) was associate d with a
67% risk reduction for a doubling of plasma creatinine levels
at 9 years (0.71% of the intensive group and 1.76% of the
conventional group, P ¼ 0.027).
298
International Relevance
The incidence and prevalence of diabetes is rising around the
world and at rapid rates in developing countries. Glycemic
control is therefore one of the most important strategies
for delaying progression of CKD, irrespective of region or
country. It is recognized that not all hypog lycemic strategies
or treatments are available in all countries.
Implications for Clinical Practice and Public Policy
Practitioners should encourage glycemic control in all people
with CKD and diabetes, including referral to education
sessions and diabetic clinics where available. Public health
policies and diabetic strategies should include screening in
high-risk populations such as those with diabetes, as the
presence of CKD would confer higher risk of adverse events,
and could represent an opportunity for intensified interven-
tion with large implications for health-care.
Areas of Controversy, Confusion, or Non-consensus
R ecommendations on the use of specific medications for
glycemic control (e.g., metformin and glyburide) remain
variable depending on different perspectives. These are co ver ed
more fully in Chapter 4 on medication dosing and AKI.
Clarification of Issues and Key Points
Glycemic control improves outcomes in people with diabetes,
with or without CKD. Those with diabetes and CKD have
higher risk of adverse outcomes and therefore presumed
higher benefit from control. Many agents are renally excreted
and therefore adjustments to doses may be necessary as GFR
declines or if patients are acutely unwell (see Chapter 4 on
medication dosing).
Caveats in measuring glycemic control with HbA
1C
HbA
1C
may not be reflective of glucose control in people
with CKD who have reduced red cell life span, and thus
should be interpreted with caution. Review of blood sugar
daily logs may be more reliable.
Clinicians should be aware that HbA
1C
measurements that
inform glycemic control are based on an assumed red cell life
span of 90 days. In people with CKD the RBC life span is
shortened, even if receiving erythropoiesis-stimulating agents
(ESAs). As such, measurement may only reflect glycemic
control over a shor ter time period than the presumed 3
months and thus, HbA
1C
measurements may be falsely low.
Awareness of this may alter clinicians’ reliance on this
measurement as a long-term measure.
299–304
Ongoing
research intitiatives comparing HbA
1C
with glycated albumin
using continuous glucose monitoring suggest that glycated
albumin may provide a more reliable index of glycemic
control in people with advanced CKD.
Pediatric Considerations
It is recommended that the Guidelines of the American
Diabetes Association
305
or similar national guidelines for
diabetes management in children and adolescents, be
reviewed to address issues related to management of diabetic
children either with, or at risk of, CKD.
Salt intake
3.1.19: We recommend lowering salt intake to o90 mmol
(o2 g) per day of sodium (corr esponding to 5 g of
sodium chloride) in adults, unless contraindicated
(see rationale). (1C)
3.1.19.1: We recommend restriction of sodium intake
for children with CKD who have hypertension
(systolic and/or diastolic blood pressure 495
th
percentile) or prehypertension (systolic and/or
diastolic blood pressure 490
th
percentile and
o95
th
percentile), following the age-based
Recommended Daily Intake. (1C)
3.1.19.2: We recommend supplemental free water and
sodium supplements for children with CKD
and polyuria to avoid chronic intravascular
depletion and to promote optimal growth. (1C)
RATIONALE
In subjects with CKD, impaired excretion of sodium is often
present. High sodium intake increases BP and proteinuria,
induces glomerular hyperfiltration and blunts the response
to RAAS blockade. Lowering salt intake not only reduces BP,
but also lowers albuminuria. The importance of salt intake
in the general management of CKD patients cannot be
overemphasized, hence the need for specific statements here.
We appreciate that there are some conditions in which salt
Table 25 | Intensive versus normal glycemic control and albuminuria outcome
Study Intensified treatment versus normal treatment HbA
1C
goals Albuminuria outcome
ADVANCE
294
6.5% (48 mmol/mol) versus 7.3% (56 mmol/mol) 9% k in new ACR 3-30 mg/mmol (30-300 mg/g)
30% k in ACR progression to 430 mg/mmol (4300 mg/g)
ACCORD
295
6.3% (45 mmol/mol) versus 7.6% (60 mmol/mol) 21% k in new ACR 3-30 mg/mmol (30-300 mg/g)
32% k in ACR progression to 430 mg/mmol (4300 mg/g)
VADT
296
6.9% (52 mmol/mol) versus 8.4% (68 mmol/mol) 32% k in new ACR 3-30 mg/mmol (30-300 mg/g)
37% k in ACR progression to 430 mg/mmol (4300 mg/g)
Abbreviations: ACCORD, Action to Control Cardiova scular Risk in Diabetes; ACR, albumin-to-creatinine ratio; ADVANCE, Action in Diabetes and Vascular Disease: Preterax and
Diamicron Modified Release Controlled Evaluation; HbA
1C
, hemoglobin A
1C
; VADT, Veterans Affairs Diabetes Trial.
Kidney International Supplements (2013) 3, 73–90
77
chapter 3

restriction may be harmful, and hence the qualifier ‘‘unless
contraindicated.’ These conditions include salt losing ne-
phropathies and those prone to hypotension and volume
contraction who do not have heart failure.
Evidence Base
A systematic review of 16 studies addressing salt intake and
kidney disease set out to establish whether variations in
dietar y sodium consumption influence renal outcomes in
people with CKD.
306
Despite marked heterogeneity, the
review suggested that increased salt intake was associated
with worsening albuminuria and an increased likelihood of
reduction of GFR. Although the quality of the studies
included was insufficient to support the authors’ hypothesis
that increased salt intake is nephrotoxic, the results were
strong enough to suggest modest dietary avoidance of salt
should be encouraged in people with CKD, especially those
with hypertension and/or proteinuria. In a randomized,
double-blind, placebo-controlled trial of salt reduction in 40
Afro-Caribbean hypertensives, a salt-restricted diet (approxi-
mately 5 g daily) significantly reduced 24-hour urinary
protein excretion by 19% and led to a fall in systolic and
diastolic BP of 8 mm Hg and 3 mm Hg, respectively.
307
The
fall in urinary protein excretion correlated with a reduction
in urinary sodium excretion, not with BP reduction.
Individuals with metabolic syndrome may be especially
sensitive to the effects of sodium intake. Hoffman and
Cubeddu examined the role of salt intake in increased BP in
109 subjects with metabolic syndrome.
308
Salt restriction
from an average usual intake of 8.2 g/day to nearly 2.3 g/day
reduced the percentage of hypertensive patients from 23.8%
to 8.2%. In a six-month prospective controlled trial, 110
patients with GFR categories G4 or G5 (GFRo30 ml/min/
1.73 m
2
) followed a low-sodium (circa 1 g/day) diet as part of
either a LPD (0.6 g/kg/day) or very low-protein diet (VLPD)
supplemented with essential amino acids (0.35g/kg/day), or a
free diet.
309
BP fell significantly in the VLPD group from
143
±
19/84
±
10 to 128
±
16/78
±
7mm Hg (Po0.0001),
despite reduction of antihypertensive drugs. The improved
BP correlated with decreased urinary sodium and the authors
concluded that the antihypert ensive effect was due to
reduction of salt intake independent of actual protein intake.
Finally, a randomized controlled crossover trial in 52 non-
diabetic subjects with CKD compared the effects of a low-
sodium diet (target 50 mmol [1.15 g] sodium per day) versus
a regular sodium diet (target 200 mmol [4.60 g] sodium per
day) on lowering of proteinuria through RAAS blockade.
310
The reduction of proteinuria by the addition of a low-sodium
diet to angiotensin-converting enzyme inhibition was
significantly larger (Po0.001) than the addition of angio-
tensin receptor blockade to angiotensin-converting enzyme
inhibition and similarly the reduction of systolic BP by the
addition of a low-sodium diet was significantly larger (P ¼
0.003) than the addition of angiotensin receptor blockade.
The authors concluded that sodium restriction to a level
recommended in guidelines was more effective than dual
renin-angiotensin blockade for reduction of proteinuria and
BP in non-diabetic nephropathy.
International Relevance
Salt intake has been identified as an important driver of high
BP in all countries and in adults, thus this recommendation
is of international relevance and applicability. The relevance
and caveats to children in different situations around the
world merits further review.
Implications for Clinical Practice and Public Policy
In most developed countries, a reduction in salt intake can be
achieved by a gradual and sustained reduction in the amount
of salt added to foods by the food industry. In other countries
where most of the salt consumed comes from salt added
during cooking or from sauces, a public health campaign is
needed to encourage consumers to use less salt. The range of
sodium intake associated with improved long-term outcomes
is easily achievable and may have the potential to greatly
improve health outcomes in patients with CKD around the
world.
Areas of Controversy, Confusion, or Non-consensus
While salt restriction is of benefit on a population-basis, and
in most people w ith hypertension and CKD, there may be
individual conditions in which salt restriction might be
detrimental (e.g., those with salt wasting tubular disorders;
those either hypotensive in the abs ence of heart failure or
those prone to volume contraction). Thus while this general
statement applies to the majority of people, individualization
is required based on clinical circumstance. This may be
particularly relevant to children.
Pediatric Considerations
The suggested relative salt restrictions for children with CKD
and/or hypertension are age-based with consideration given
to both average intakes and upper limit values. The
recommended upper limit for sodium intake from which a
reduced intake could be derived is provided in Table 26.
It must also be recognized that children with CKD often
have underlying tubular conditions that predispose to
numerous electrolyte losses, including sodium. For these
children a supplemental rather than restricted sodium intake
will be required.
Table 26 | Recommended Daily Intake of sodium for healthy
children
Age Upper Limit
0-6 months No data
7-12 months No data
1-3 years 1500 mg
4-8 years 1900 mg
9-13 years 2200 mg
14-18 years 2300 mg
78
Kidney International Supplements (2013) 3, 73–90
chapter 3

Hyperuricemia
3.1.20: There is insufficient evidence to support or refute
the use of agents to lower serum uric acid
concentrations in people with CKD and either
symptomatic or asymptomatic hyperuricemia in
order to delay progression of CKD. (Not Graded)
Hyperuricemia is common in people with CKD and is
defined by urate concentrations above 7.0 mg/dl (420 mmol/l)
as measured by automated enzymatic (uricase) laboratory
methods. Concentrations obtained with colorimetric
methods are approximately 1 mg/dl (60 mmol/l) lower.
The Work Group believe it impor tant to acknowledge the
accumulating body of evidence describing the association of
hyperuricemia with CKD and adverse cardiovascular out-
comes, and thus list hyperuricemia as a potential contributor
to progression. However, at the time of current writing, there
is not a reliable body of evidence from which to recommend
treatment of hyperuricemia for the specific goal of delaying
progression of CKD. Thus, the wording of this ungraded
statement is very deliberate.
RATIONALE
Observational data had implicated uric acid in the progres-
sion of CKD suggesting that adverse outcomes in people with
CKD may be improved by specific uric acid lowering therapy.
Small studies using appropriate RCT design have shown
reduced left ventricular mass, improved endothelial function,
and reduced progression of CKD in people with either
symptomatic or asymptomatic hyperuricemia and CKD.
Thus, the specific statement is intended to ensure and foster
RCTs to properly assess the risk and benefits of uric acid
lowering strategies in people with CKD.
Evidence Base
Published data implicate elevated serum uric acid (SUA)
concentrations in the progression of CKD.
311–315
Reduction
of SUA by allopurinol has been reported to delay progression
of CKD in people with both diabetic and nondiabetic
CKD.
316,317
Treatment of asymptomatic hyperuricemia has
also been reported to improve kidney function even in
subjects with normal levels of GFR.
318,319
Both GFR and
endothelial function significantly improved in asymptomatic
hyperuricemic subjects randomly assigned to 300 mg/day of
allopurinol in comparison to placebo.
318
A separate double-
blind, placebo-controlled, parallel-group study in 67 people
with CKD (GFR 30-60 ml/min/1.73 m
2
) and left ventricular
hypertrophy (LVH) randomly assigned subjects to treatment
with allopurinol (300 mg/day) or placebo for 9 months.
320
In comparison to placebo, the allopurinol-treated subjects
had significant reductions in left ventricular mass assessed
by magnetic resonance imaging (MRI), and improvements
in endothelial function assessed by flow-mediated dilation
of the brachial artery and in central arterial stiffness
assessed by pulse-wave analysis. Another study randomized
70 subjects with known hyperuricemia or SUA concentra-
tions Z 7.0 mg/dl (Z420 mmol/l) to treatment with either
allopurinol monotherapy (100-200 mg/day) or a combina-
tion of allopurinol and a citrate preparation (3 g/day).
321
SUA concentrations were decreased in both groups but to a
significantly lower level by combination treatment. GFR
assessed by CrCl increased in the combination therapy group
but remained unchanged in those treated with allopurinol
alone. Other uric acid lowering agents have also been
reported to improve outcomes in people with CKD. In an
8-week, placebo-controlled group comparison of rasburicase
and placebo, a single 4.5 mg dose of rasburicase significantly
lowered SUA and resulted in a significant improvement in
kidney function assessed by CrCl.
322
In a post hoc analysis of
1342 patients with type 2 diabetes mellitus and nephropathy
participating in the Reduction of Endpoints in Non-Insulin-
Dependent Diabetes Mellitus with the Angiotensin II
Antagonist Losartan (RENAAL) trial, Miao et al. examined
the relationship between change in SUA concentration after 6
months of treatment with losartan and doubling of SCr or
ESRD.
323
Baseline SUA was 6.7 mg/dl (400 mmol/l) in placebo
and losartan-treated subjects. During the first 6 months,
losartan lowered SUA by 0.16 mg/dl (9.5 mmol/l) [95% CI
0.30-0.01; P ¼ 0.031] as compared with placebo. The risk of
doubling of SCr or ESRD was decreased by 6% (95% CI 10%-
3%) per 0.5-mg/dl (30-mmol/l) decrement in SUA during the
first 6 months. This effect was independent of other risk
markers, including albuminuria.
International Relevance
There are no data to support or refute the importance of
hyperuricemia in different geographical reasons or ethnic
groups. Further study is needed.
Implications for Clinical Practice and Public Policy
There is insufficient evidence to recommend the use of uric
acid lowering agents in asymptomatic individuals for the
specific purpose of delaying CKD progression. Further large
trials are required to better understand the potential benefit
of uric acid lowering for this purpose.
Lifestyle
3.1.21: We recommend that people with CKD be encour-
aged to undertake physical activity compatible with
cardiovascular health and tolerance (aiming for at
least 30 minutes 5 times per week), achieve a
healthy weight (BMI 20 to 25, according to country
specific demographics), and stop smoking. (1D)
RATIONALE
People with CKD have self-reported reduced physical
functioning and are not as aerobically fit as the general
population. Frailty, impaired physical performance, disabil-
ity, and ger iatric syndromes are common among older adults
even with mild kidney disease. Reduced physical functioning
and inactivit y are associated with increased mortality and
poor QOL. Obesity is associated with increased morbidity,
Kidney International Supplements (2013) 3, 73–90 79
chapter 3

mortality, and reduction in life expectancy and leads to an
increase in the incidence of diabetes, hypertension, and
dyslipidemia. Associations between smoking and CKD
suggest that smoking increases the risk of kidney failure
and that smoking cessation decreases that risk . Thus, this
statement reflects the importance of ensuring lifestyle
recommendations.
Evidence Base
CKD patients have a reduced exercise capacity and impaired
physical functioning.
324–326
Moreover, reduced physical
activity is asso ciated with increased mortality and poor
QOL in people with CKD.
327–329
Regular exercise leads to
increased exercise capacity, decreased morbidity, and im-
proved health-related QOL.
330–332
Exercise may reduce
cardiovascular risk through its beneficial effects on BP,
triglycerides, high-density lipoprotein cholesterol (HDL-C),
insulin resistance, and glycemic control. In ESRD, exercise
has been shown to improve arterial stiffness, BP, cardior-
espiratory function, and QOL.
333–339
Less data are available
on the beneficial effects of exercise on early CKD. However, as
cardiovascular risk gradually increases with both a lower GFR
and a higher ACR, it is expected that exercise will also help to
prevent progressive CVD in less severe CKD. Indeed, in
subjects with GFR categories G3a-G4 (GFR 15–59 ml/min/
1.73 m
2
), long-term exercise training improved physical
impairment, arterial stiffness, and health-related QOL.
340
It
has therefore been argued that exercise training is imperative
in CKD patients
341
and that support programs including self-
monitoring , verbal reinforcement, and motivation should be
applied
342–344
in an attempt to prevent the high cardiovas-
cular risk in CKD. A prospective study compared the benefits
of 6 months of regular walking in 40 predialysis patients with
GFR categories G4-G5 (GFRo30 ml/min/1.73 m
2
) (20 in an
exercising group and 20 patients who continued with usual
physical activity for comparison). Improvements noted after
1 month were sustained to 6 months in the 18 of 20 who
completed the exercise study. These included improvements
in exercise tolerance (reduced exertion to achieve the same
activity), weight loss, improved cardiovascular reactivity,
avoiding an increase in BP medication and improvements in
quality of health and life and uremic symptom scores as
assessed by questionnaire.
345
In the absence of diabetes, hypertension or other
cardiovascular risk factors there has been a lack of evidence
to support a causal link between obesity and CKD.
Observational studies suggest that obesity is an independent
risk factor for CKD.
346–348
The ev idence in population studies
is conflicting; some studies have failed to link obesity with
decreased GFR,
349,350
possibly because BMI in isolation is a
poor measure, while others sug gest that CKD is indepen-
dently associated with BMI.
351
It has been known for some
time that obesity is associated with secondary focal and
segmental glomerulosclerosis,
352
yet significant associations
between obesity and CKD in large observational studies such
as The Framingham Heart Study disappear after adjustment
for age, gender, and cardiovascular risk factors.
353
However,
systematic review and meta-analysis of weight loss interven-
tions in CKD have shown weight loss to be associated with
significant decrease in proteinuria and systolic BP with no
further decrease in GFR in people with CKD during a mean
follow-up of 7.4 months.
354
A further systematic review drew
very similar conclusions. Weight loss interventions were
associated with decreased proteinuria and albuminuria by
1.7 g (95% CI 0.7-2.6 g) and 14 mg (95% CI 11-17 mg),
respectively (Po0.05).
355
Each 1-kg weight loss was asso-
ciated with 110 mg (95% CI 60-160 mg; Po0.001) decrease
in proteinuria and 1.1 mg (95% CI 0.5-2.4 mg; P ¼ 0.011)
decrease in albuminuria, respectively, independent of reduc-
tion in BP.
Multiple studies docum ent a clear association between
smoking and renal damage in the general population,
patients with diabetes, and hypertensive patients.
356
Smoking
is causally linked to cardiovascular events in the general
population and is also associated with an increased risk for
cardiovascular events in patients with CKD.
357–360
Studies
investigating the beneficial effects of smoking cessation on
kidney function have all been positive.
361–365
International Relevance
Exercise, weig ht loss and smoking cessation in CKD are
equally important in all countries and thus this recommen-
dation is of international relevance and applicability.
Implications for Clinical Practice and Public Policy
Implementation of this recommendation has no public
health cost but has the potential to make far-reaching public
health gains both in terms of population health and health-
care economics.
Additional dietary advice
3.1.22: We recommend that individuals with CKD receive
expert dietary advice and information in the
context of an education program, tailored to
severity of CKD and the need to intervene on salt,
phosphate, potassium, and protein intake where
indicated. (1B)
RATIONALE
International Relevance
The importance of lifestyle recommendations and dietary
counseling cannot be overstated. It is however recognized
that within the context of different countries, health-care
systems and jurisdictions, the degree to which these can be
implemented is variable. These recommendations serve as a
‘best practice’ suggestion.
3.2 COMPLICATIONS ASSOCIATED WITH LOSS OF KIDNEY
FUNCTION
People with CKD are prone to develop a variety of compli-
cations which reflect loss of endocrine or exocrine function of
80 Kidney International Supplements (2013) 3, 73–90
chapter 3

the kidneys. The incidence and prevalence of these complica-
tions increase with severity of CKD as defined predominantly
by GFR categories (Table 27).
It is beyond the scope of this guideline to describe each
of the complications and the proposed treatment options
for them in detail as guidance for these conditions can
be found in other documents. However, for the purpose of
completeness, the key complications and management
recommendations for people with CKD are addressed in
this section.
In addition to these complications, we have described
strategies to delay progression of CKD which are in part
predicated on the identification and management of the
clinical, metabolic, and hematologic complications. Note that
not all people with CKD will have all of the complications
and complications may not occur at the same rate or to the
same degree in individuals with the same categories of GFR
or albuminuria. Nonetheless knowledge of the common
complications and treatment options is important in the care
of CKD.
Management of Complications
Anemia in CKD.
Anemia is an important complication of
CKD because it contributes significantly to the heavy
symptom burden of CKD. It has a major impact on the lives
of people with CKD but it is potentially reversible with
appropriate treatment. The guideline statements included
here are those we consider to be the key considerations for
people with non-dialysis CKD. Interested readers are referred
to the KDIGO Clinical Practice Guideline for Anemia in
CKD
11
for comprehensive guidance on this topic.
Definition and identification of anemia in CKD
3.2.1: Diagnose anemia in adults and children 415 years
with CKD when the Hb concentration is o13.0 g/
dl (o130 g/l) in males and o12.0 g/dl (o120 g/l) in
females. (Not Graded)
3.2.2: Diagnose anemia in children with CKD if Hb
concentration is o11.0 g/dl (o110 g/l) in children
0.5-5 years, o11.5 g/dl (115 g/l) in children 5-12
years, and o12.0 g/dl (120 g/l) in children 12-15
years. (Not Graded)
RATIONALE
These statements reflect the need to measure Hb in
people with CKD in order to detect if anemia is present,
given its association with poor outcomes and its use in
prediction models. Anemia is a common occurrence in CKD
patients, though variable in its time of presentation and
severity within individuals. Thus, the guidelines stress that
evaluation and treatment of anemia in people CKD should be
undertaken as in other individuals, and emphasize that
anemia due to CKD is a diagnosis of exclusion. Further-
more, the guidelines stress that laboratory values used for
diagnosis do not imply therapeutic thresholds or targets.
For details, consult KDIGO Clinical Practice Guideline for
Anemia in CKD.
11
Evidence Base
Conventionally anemia is defined as a Hb concentration
lower than the established cutoff defined by the WHO.
368
Different biological groups have different cutoff Hb values
below which anemia is said to be present, ranging from
11 g/dl (110 g/l) for pregnant women and for children
between 6 months and 5 years of age, to 12 g/dl (120 g/l)
for non-pregnant women, and to 13 g/dl (130 g/l) for men
(Table 28).
International Relevance
Globally the prevalence of anemia is 24.8% of the population,
much of this is linked to nutritional deficiency exacerbated by
Table 27 | Prevalence of CKD complications by GFR category*
derived from CKD cohorts
Complication
GFR category (ml/min/1.73 m
2
)
Reference
Z90 60-89 45-59 30-44 o30
Anemia
1
4.0% 4.7% 12.3% 22.7% 51.5%
366
Hypertension
2
18.3% 41.0% 71.8% 78.3% 82.1%
366
25(OH) Vit D deficiency
3
14.1% 9.1% 10.7% 27.2%
367
Acidosis
4
11.2% 8.4% 9.4% 18.1% 31.5%
366
Hyperphosphatemia
5
7.2% 7.4% 9.2% 9.3% 23.0%
366
Hypoalbuminemia
6
1.0% 1.3% 2.8% 9.0% 7.5%
366
Hyperparathyroidism
7
5.5% 9.4% 23.0% 44.0% 72.5%
366
Abbreviations: CKD, chronic kidney disease; GFR, glomerular filtration rate.
*Note that modification of prevalence according to albuminuria categories data is
not yet available to inform this table adequately, though there are limited data to
suggest increasing prevalence of hypoalbuminemia, hypertension, anemia, and
acidosis as albuminuria category increases
1
Defined as hemoglobin levels o12 g/dl (120 g/l) for women; o13.5 g/dl (135 g/l)
for men
2
Defined as a systolic blood pressure Z140 mm Hg, diastolic blood pressure
Z90 mm Hg, or self-reported use of antihypertensive medication
3
Less than 15 ng/ml [37 nmol/l] (as defined in Study for the Evaluation of Early
Kidney disease [SEEK])
4
Defined as serum bicarbonate less than 21 mEq/l
5
Defined as serum phosphate Z4.5 mg/dl (Z1.5 mmol/l)
6
Defined as serum albumin less than 3.5 g/dl (35 g/l)
7
Defined as PTH levels Z70 pg/ml; (Z7.4 pmol/l)
Table 28 | Hemoglobin thresholds used to define anemia
Age or gender group Hemoglobin threshold: g/dl (g/l)
Children
6 mo-5 yr 11.0 (110)
5-12 yr 11.5 (115)
12-15 yr 12.0 (120)
Non-pregnant females 415 yr 12.0 (120)
Pregnant females 415 yr 11.0 (110)
Men 415 yr 13.0 (130)
Abbreviations: mo, month; yr, year.
Reproduced with permission from World Health Organization. Worldwide pre-
valence of anaemia 1993-2005: WHO global database on anaemia.
368
In: de Benoist
B, McLean E, Egli I and Cogswell M (eds), 2008; accessed: h ttp://whqlibdoc.who.int/
publications/2008/9789241596657_eng.pdf.
Kidney International Supplements (2013) 3, 73–90
81
chapter 3

infectious disease. However, altitude, race, and smoking will
each also have an impact on Hb concentration. Hb
concentration can be expected to increase by about 0.6 g/dl
(6 g/l) in women and 0.9 g/dl (9 g/l) in men for each 1000 m
of altitude above sea level.
369
Hb concentrations also vary
between ra ces, with African-American individuals consis-
tently showing concentrations 0.5 to 0.9 g/dl (5 to 9 g/l)
lower than those of whites or Asian populations.
370–372
Smoking is associated with elevated carboxyhemoglobin
levels, which are associated with a compensatory increase
in total Hb concentration. Hence, the US Centers for Disease
Control and Prevention have recommended a downwards
adjustment of 0.3 g/dl (3 g/l) for smokers.
373
Thus, specific
Hb levels expected for different levels of eGFR should be
contextualized within the normal distribution of the
population.
Implications for Clinical Practice and Public Policy
The recommended thresholds for diagnosis and evaluation
of anemia should not be interpreted as being thresholds
for treatment of anemia but simply for the identification of
this complication. Practice preferences with respect to
treatment strategies should be directed according to local
resources.
Evaluation of anemia in people with CKD
3.2.3: To identify anemia in people with CKD measure Hb
concentration (Not Graded):
K when clinically indicated in people with GFR
Z60 ml/min/1.73 m
2
(GFR categories G1-G2);
K at least annually in peo ple with GFR 30-59 ml/
min/1.73 m
2
(GFR categories G3a-G3b);
K at least twice per year in people with GFR
o30 ml/min/1.73 m
2
(GFR categories G4-G5).
RATIONALE
The recommendation that pati ents be evaluated at least
annually rests on observation s from clinical trials that (in the
absence of ESA therapy) the natural history of anemia in
patients with CKD is a gradual decline in Hb concentration
over time.
374–376
The exact frequency of monitoring of Hb
concentration will be influenced by kidney function, under-
lying disease process, the initial Hb concentration, and the
rate of change in Hb concentration. The latter w ill also be
influenced by whether or not anemia is being treated and
what type of treatment is undertaken. The statem ents
specifically address the need to measure Hb concentrations
at a minimum and are not intended to deter the clinician
from more frequent measurements as required for individual
circumstances.
The initial evaluation of anemia in CKD is geared towards
the exclusion of causes other than those directly related to
kidney disease (relative iron and erythropoietin deficiency);
see KDIGO Clinical Practice Guideline for Anemia in CKD
11
for details.
Evidence Base
Anemia as defined above is found in people with CKD in
increasing proportions as GFR declines (Table 27). This is
due to a number of reasons, including loss of erythropoietic
hormone efficacy, production, substrate deficiency (most
notably iron), and other conditions w hich may contribute to
the lack of effective erythropoiesis. For a full description of
the evidence behind this statement please refer to the KDIGO
Clinical Practice Guideline for Anemia in CKD.
11
International Relevance
Frequency of Hb concentration monitoring and the initial
evaluation of anemia should not differ by country, except
that causes of anemia other than those related to CKD do
differ. Nutritional deficiencies in resource-poor areas are
common, particularly iron deficiency, which is frequently
exacerbated by infectious diseases.
Implications for Clinical Practice and Public Policy
Anemia is associated with increased morbidity, mortality,
and consumption of health-care resources. The major health
consequences include poor pregnancy outcome, impaired
physical and cognitive development, increased risk of
morbidity in children, and reduced work productivity in
adults.
Treatment of anemia in CKD. The last 30 years have seen a
major transition in approach to the treatment of anemia in
people with CKD beginning with the introduction of
erythropoietin therapy into clinical practice and the sub-
sequent resurgence of interest in iron therapies. The promise
of early intervention with anemia treatment in non-dialysis
CKD suggested by observational study has been tempered
with the realit y of risks of adverse cardiovascular outcomes in
RCTs. Nevertheless treatment of anemia with iron and ESAs
has an important positive role to play in the lives of people
with CKD. For the treatment of anem ia in people with CKD,
we suggest clinicians refer to the KDIGO Clinical Practice
Guideline for Anemia in CKD for further detai ls.
11
The key points for practitioners to remember include the
following:
1. Work-up for anemia in CKD should include assess-
ment of secondary causes including iron deficiency.
2. Iron replacement is often effective in anemia of CKD as
initial therapy and routes of administration (intrave-
nous or oral) will be determined by clinicians, patient
preferences, and local available resources.
3. ESA therapy is not recommended in those with active
malignancy, or recent history of malignancy.
4. In most people with CKD, ESAs should not be used to
intentionally increase the Hb concentration above 11.5
g/dl (115 g/l)
5. For pediatric patients, the selection of Hb concentra-
tion at which ESA therapy is initiated should be
individualized after taking into account the poten-
tial benefits (e.g., improvement in QOL, school
82 Kidney International Supplements (2013) 3, 73–90
chapter 3

attendance/performance, and avoidance of transfusion)
and potential harms.
3.3 CKD METABOLIC BONE DISEASE INCLUDING
LABORATORY ABNORMALITIES
Changes in bone mineral metabolism and alterations in calcium
and phosphate homeostasis oc cur early in the course of CKD
and progress as kidney function declines (Table 27). These
changes are grouped under the umbrella term Chronic Kidney
Disease-Mineral and Bone Disorder (CKD-MBD) which
includes renal osteodystrophy and extraskel etal (vascular) calci-
fication related to abnormalities of bone mineral metabolism.
R enal osteodystroph y is the component of CKD-MBD that is
identified and quantified through bone biopsy histomorpho-
metry and includes osteitis fibrosa (hyperparath yroidism),
osteomalacia, and adynamic bone disease.
The evidence on which existing recommended guideline
treatment targets for serum concentrations of calcium,
phosphate, and parathyroid hormone (PTH), and the
strategies to achieve these targets, is exclusively observational
and thus problematic for that reason. Furthermore, very little
of the evidence is derived from patients with non-dialysis CKD.
Nevertheless we feel it is important to include here some of the
key statements relating to mineral metabolism abnormalities in
patients with non-dialysis CKD from the KDIGO Clinical
Practice Guideline for the Diagnosis, Evaluation, Prevention,
and Treatment of Chronic Kidney Disease-Mineral and Bone
Disorder (CKD-MBD) published in 2009.
9
These statements
will be qualified where new data since the publication of
the CKD-MBD guidance have become available.
3.3.1: We recommend measuring serum levels of calcium,
phosphate, PTH, and alkaline phosphatase activity
at least once in adults with GFR o45 ml/min/
1.73 m
2
(GFR categories G3b-G5) in order to
determine baseline values and inform prediction
equations if used. (1C)
RATIONALE
As kidney function declines, abnormalities of serum calcium,
phosphate, and circulating hormones related to CKD-MBD
progress. These include PTH; 25-hydroxyvitamin D
(25(OH)D), 1,25-dihydroxyv itamin D (1,25(OH)
2
D), and
other vitamin D metabolites; fibroblast growth factor-23
(FGF-23); and growth hormone. At the tissue level there is
down regulation of vitamin D receptors and resistance to the
actions of PTH. Immunohistochemical abnormalities in bone
also occur early and generally precede changes in mineral
homeostasis. Extraskeletal calcification may result from
deranged mineral and bone metabolism and from the
therapies used in an attempt to correct these abnormalities.
Associations between disorders of mineral metabolism and
CVD have widened the focus of CKD-MBD to include
abnormal mineral metabo lism, abnormal bone, and extra-
skeletal calci fication. Once baseline values have been
obtained, the subsequent frequency of testing will be
determined on an individual basis by the actual value and
any intervention that may be introduced.
Evidence Base
There are obse rvational studies which describe the abnorm-
alities of each of these parameters at relatively high values of
eGFR in general and high-risk population cohorts.
367,377,378
Importantly, abnormalities of calcium and phosphate appear
to occur relatively later in the course of CKD than do
abnormalities in value s of 1,25(OH)
2
D, 25(OH)D, and PTH.
Thus, the recommendation is to evaluate these parameters
relatively early in the trajectory of CKD, as an assessment of
burden of illness. In dialysis patients, the highest risks for
mortality have been reported with combinations of hig h
serum phosphate and calcium together with either high
PTH (RR 3.71; 95% CI 1.53-9.03; P ¼ 0.004) or low PTH
(RR 4.30; 95% CI 2.01-9.22; Po0.00 1) compared with the
combination of high PTH with normal serum calcium and
phosphate which had the lowest mortality and was used as
the index category.
379
The importance of examining combi-
nations of parameters of mineral metabolism is likely to be
no different in patients with less severe CKD, but this has not
been tested in non-dialysis populations.
There are also racial differences in the parameters of
mineral metabolism. In a multicenter cohort of 227 black and
1633 non-black patients with early CKD, blacks had similar
1,25(OH)
2
D levels compared with non-blacks but signifi-
cantly lower levels of 25(OH)D with higher levels of calcium,
phosphate, and PTH, and were significantly more likely to
have hyperphosph atemia than non-blacks.
380
In multi-
variable analyses adjusted for age, gender, eGFR, BMI, and
diabetes, blacks had significantly lower 25(OH)D and hi gher
PTH levels than non-blacks. Examining the relationships
between 25(OH)D and PTH in 8415 adult participants (25%
black and 24% Mexican-American) in NHANES 2003–2004
and 2005–2006, and the relationship between 25(OH)D and
bone mineral density (BMD) in 4206 adult par ticipants (24%
black and 24% Mexican-American) in the 2003–2004
NHANES sample, Guitierrez et al. found signific ant racial
differences.
381
Blacks and Mexican-Americans had signifi-
cantly lower 25(OH)D and higher PTH concentrations than
whites (Po0.01 for both). Bone mineral density significantly
decreased (Po 0.01) as serum 25(OH)D and calcium intake
declined among whites and Mexican-Americans, but not
among blacks (P ¼ 0.2).
International Relevance
The association between black race and Hispanics and
secondary hyperparathyroidism, independent of known risk
factors, suggests that novel mechanisms may contribute to
secondary hyperparathyroidism in non-whites with CKD.
Testing for these parameters would therefore be informed by
the demographics of the population. In different countries
and regions, the ability to measure these parameters may
Kidney International Supplements (2013) 3, 73–90 83
chapter 3

vary, thus the authors of the guideline statements appreciate
that implementation of regular measurements of all thes e
parameters may not be possible in all jurisdictions.
Implications for Clinical Practice and Public Policy
Given there remains no clear recommendation as to
‘expected’ values in CKD nor consensus on thresholds
regarding treatment, the testi ng of PTH, and vitamin D
parameters would lead to substan tial costs to the health-care
system. Abnormal values lead to repeat testing. There are no
data to suggest how effective or useful repeated moni toring of
abnormal values is, nor what an acceptable interval of
monitoring should be to inform care. Laboratory testing for
phosphate and calcium is relatively inexpensive, but treat-
ment and ongoing monitoring may be exp ensive. At the
current time, recommendations for testing frequency may be
problematic for clinical practice.
Areas of Controversy, Confusion, or Non-consensus
The inter-relationship of calcium, phosphate, and PTH, and
the potential impact of vitamin D on these mineral
metabolites and extraskeletal calcification remains an area
of research and debate among clinicians. Newer research
examining the role of FGF-23, an important molecule in
phosphate, PTH, and vitamin D homeostasis, has caused
many to question the previous focus on PTH values as
misplaced. The questions of whether vitamin D therapies are
toxic in some or all patients and what values of phosphate are
pathologic have yet to be resolved.
Pediatric Considerations
Application of guidelines related to bone health, growth, and
CKD in children is extremely complex. Numerous issues
arise, including age-related variation in normative values,
comparisons across age, sex, size, and the need to account for
pubertal changes etc., when one considers the options and
targets for evaluation and treatment.
It is recommended that in the application of any of these
specific guidelines, the reader carefully reviews the publica-
tions as they relate to pediatrics, star ting with the following
two documents and then accessing the most currently
available pediatric CKD resources for the topic(s).
Recommended primary pediatric CKD-MBD resources:
K KDOQI Clinical Practice Guideline for Nutrition in
Children with CKD: 2008 Update
382
K KDIGO Clinical Practice Guidelines for the Diagnosis,
Evaluation, Prevention, and Treatment of CKD-MBD
9
3.3.2: We suggest not to perform bone mineral density
testing routinely in those with eGFR o45 ml/min/
1.73 m
2
(GFR categories G3b-G5), as information
may be misleading or unhelpful. (2B)
RATIONALE
While there is an appreciation that BMD is measured in
many elder ly, this statement is intended to highlight for the
clinician the fact that the information gained from BMD in
those with reduced GFR may be false, leading to either
under- or over-treatment. Although fractures rates and
fracture-related mortalit y are elevated in CKD, bone
densitometry does not reliably predict fracture risk in
patients with GFR o45 ml/min/1.73 m
2
and neither does it
predict the type of renal osteodystrophy. Thus, BMD
measurements do not provide the information usually sought
from such testing, which is usually the basis of interventions.
Evidence Base
Decreased bone mass and changes in bone microarchitectu re
occur early in CKD and worsen with progression of disease
such that patients with CKD are at increased risk of bone
fracture.
383
Bone strength is determined by the density and
quality of the bone. Dual-energy x-ray absorp tiometry
(DXA) scann ing measures density of bone but is not able
to determine bone quality (cortical and trabecular micro-
architecture). Studies using high-resolution peripheral quan-
titative computed tomography (HR-pQCT) demonstrate
abnormalities in the cortical and trabecular microarchitec-
ture of patients with early CKD compared with healthy
control subjects.
384
Although abnormalities of both DXA and
HR-pQCT associate with fractures in patients with CKD,
receiver operator characteristic curve analysis sug gests that
neither technique is predictive of fracture (area under the
curve o0.75), although this improved for patients with
longer duration of CKD.
385
In a cross-sectional study, the
combination of these imaging techniques with markers of
bone turnover improved prediction of fracture.
386
Implications for Clinical Practice and Public Policy
A major component of fracture risk is related to fall risk, thus
reduction in fall risk may be achieved through establish-
ment of falls prevention programs. Such programs include
medication review; prevention of postural hypotension;
cardiac pacing, where appropriate; home hazard assessment
and modifications; muscle strengthening and retraining; and
treatment of vitamin D deficiency.
Areas of Controversy, Confusion, or Non-consensus
The combination of measurements of bone thickness, BMD
of femoral neck, and a history of fracture may be useful to
identify CKD patients who might benefit from fracture
prevention strategies. Prospective studies are needed to assess
the utility of these parameters for fracture prediction in the
CKD population.
Treatment of CKD-MBD
Disturbances of calcium, phosphate, vitamin D, and PTH
develop early during the course of CKD and are associated
with adverse outcomes. Studies of these and other markers of
bone mineral metabolism have improved our understanding
of disease mechanisms governing adverse outcomes of CKD-
MBD but clinical studies have yet to indicate whether or not
manipulation of these markers improves patient-level
84 Kidney International Supplements (2013) 3, 73–90
chapter 3

outcomes. In making recommendations for therapeutic
targets for mineral metabolism abnormalities, we have been
careful not to reach beyond the evidence.
3.3.3: In people w ith GFR o45 ml/min/1.73m
2
(GFR
categories G3b-G5), we suggest maintaining serum
phosphate concentrations in the normal range
according to local laboratory reference values. (2C)
3.3.4: In people w ith GFR o45 ml/min/1.73m
2
(GFR
categories G3b-G5) the optimal PTH level is not
known. We sug gest that people with levels of intact
PTH above the upper normal limit of the assay
are first evaluated for hy perphosphatemia, hypo-
calcemia, and vitamin D deficiency. (2C)
RATIONALE
Higher serum phosphate concentrations are associated with
mortality and experimental data suggests that serum
phosphate concentration is directly related to bone disease,
vascular calcification and CVD. Serum phosphate, calcium,
and PTH concentrations are all inter-related in patients with
CKD. Randomized studies linking manipulation of these
parameters to clinical outcomes are lacking but systematic
review indicates that earlier phosphate control may help
reduce the early clinical consequences of CKD-MBD. Simi-
larly there is insufficient evidence that any specific phosphate
binder significantly impacts patient-level outcomes.
Evidence Base
Systematic review of serum concentrations of calcium,
phosphate, and PTH and the risk of death and CVD in
people with CKD showed that the ris k of death increased
18% for every 1 mg/dl (0.33 mmol/l) increase in serum
phosphate concentration (RR 1.18; 95% CI 1.12-1.25).
387
There was no association seen with either PTH or serum
calcium and all-ca use mortality (Figure 19). Of the 327,644
subjects included in the review only 16,247 were not receiving
dialysis and of these only 8990 were people with GFR
o60 ml/min/1.73 m
2
not receiving RRT. In these subjects the
risk of all-cause mortality for each 1 mg/dl (0.33 mmol/l)
increase in serum phosphate concentration was very similar
(RR 1.29; 95% CI 1.12-1.48). As with all subjects included
there was no evidence of an association between serum
calcium concentration and all-cause mortalit y in people with
CKD not receiving RRT (RR 1.02; 95% CI 0.81-1.29). Dat a
for associations of calcium, phosphate, and PTH with
cardiovascular death were only available in one of the studies
included.
In the Multi-Ethnic Study of Atherosclerosis (MESA)
study, the associations of serum phosphate concentrations
with vascular and valvular calcification were examined in 439
people with GFR o60 ml/min/1.73 m
2
. Each 1 mg/dl
(0.33 mmol/l) increase in serum phosphate concentration
was associated with a 21% (P ¼ 0.002), 33% (P ¼ 0.001 ), 25%
(P ¼ 0.16), and 62% (P ¼ 0.007) greater prevalence of
coronary artery, thoracic, aortic valve, and mitral valve
calcification, respectively.
388
The strength of the associations
did not differ by age, race, or diabetes. Adjustment for serum
concentrations of PTH and 1,25(OH)
2
D did not alter the
strength of the associations.
Factors affecting gastrointestinal phosphate absorption
include 1,25(OH)
2
D, food content, phosphate bioavailability
and phosphate binders (natural and prescribed). Sources of
dietar y phosphate are protein-rich foods, including dairy
products, meat, and fish as well as legumes, nuts, chocolates
and inorganic phosphate additives such as those found in
carbonated drinks. In a non-vegetarian Western diet, over
half the dietary intake of phosphate comes from animal
protein. Although the phosphate content of plant-derived
phosphate is higher than animal derived, its bioavailability in
terms of gastrointestinal absorption is lower.
389
Inorganic
phosphate additives have the highest bioavailability. A
number of clinical studies detail benefit from dietar y
phosphate and protein control in terms of secondary
hyperparathyroidism and progression of CKD in people
with moderate CKD.
390
Few studies have evaluated the
impact of dietary phosphate restriction on bone disease or
vascular calcification and only one has addressed survival. In
people on hemodialysis, a post hoc analysis suggested that
more restrictive prescribed dietary phosphate was associated
with poorer indices of nutritional status and a greater need
for nutritional supplementation.
391
There was a stepwise
trend toward greater survival with more liberal phosphate
prescription, which reached statistical significance among
subjects prescribed 1001 to 2000 mg/d and those with no
specified phosphate restriction, raising concerns about
protein energy malnutrition with dietary phosp hate restric-
tion. The means used to achieve phosphate restriction may
therefore be important.
Table 29 details the relative cost comparisons of phosphate
binders currently in clinical use for which there is observa-
tional or study trial data demonstrating their efficacy. Data
concerning comparative patient-level outcomes such as
mortality are not available.
There are a number of agents available for phosphate
binding which are listed in the table ranked in order of
relative cost, appreciating that both availability and specific
costs are country- and era-specific.
A Cochrane meta-analysis considered 60 RCTs or quasi-
RCTs (7631 participants) that assessed the effects of various
phosphate binders in adults with CKD.
392
The autho rs
concluded that all available phosphate-binders reduced
serum phosphate concentrations in comparison to placebo
but that data to date do not support superiority of novel
non-calcium binding agents for patient-level outcomes such
as all-cause mortality and cardiovascular end points in CKD.
International Relevance
Availability of different phosphate binders differs around the
globe. Thus, recommendations as to specific agents are not
possible within the context of these statements. Similarly,
dietar y phosphate intake may be different around the world,
Kidney International Supplements (2013) 3, 73–90 85
chapter 3

rendering this problem of greater or lesser significance in
different jurisdictions. Measurement of specific hormones
(PTH, vitamin D) is expensive and may not inform care
sufficiently to warrant the expense at this time.
Implications for Clinical Practice and Public Policy
Existing data support prevention of hyperphosphatemia
and associated secondary hyperparathyroidism in CKD. In
the absence of hypercalcemia, there is no indication to
prescribe phosphate-binders that are less cost-effective than
calcium-based agents. Current data are insufficient to make
recommendations about target levels of serum calcium or
PTH concentrations that should be achieved in order to
reduce mortality or cardiovascular morbidity in people with
CKD not requiring dialysis. Assay variability of PTH
and vitamin D remains problematic and this issue is
beyond the scope of this document. The practitioner and
health-care administrators are advised to appreciate this
problem in developing targets for care or thresholds for
treatment.
Areas of Controversy, Confusion, or Non-consensus
As per comments above, the data to support levels of
laboratory values for interventions, types of interventions,
and target values remain problematic. Thus recommenda-
tions for therapy remain similarly problematic and practice
varies depending on location and resource availability. Likely
correlation of symptoms with blood values and addressing
laboratory abnormalities within that context is a pragmatic
approach at the current time. The non-specialist is asked to
seek advice from local experts for best advice for specific
individuals.
The KDIGO guidelines on this subject have not been
updated at the time of the writing of this CKD guideline.
Increased Serum
Phosphorus Better
Decreased Serum
Phosphorus Better
No. of
Participants
Phosphorus
No. of
Cohorts
Relative Risk
(95% CI) Per
Unit Increase
All-cause mortality
4651
Adequate adjustment
3 1.35 (1.16-1.57)
87 694
Partial adjustment
10 1.16 (1.09-1.23)
92 345
All studies combined
13 1.18 (1.12-1.25)
Cardiovascular mortality
17 326
Adequate adjustment
elbamitse toN1
5881
Partial adjustment
2 1.14 (1.05-1.24)
23
207
All studies combined
3 1.10 (1.06-1.13)
0.5 1.0 2.0
Relative Risk (95% CI)
0.5 1.0 2.0
Increased
Serum Parathyroid
Hormone Better
Decreased
Serum Parathyroid
Hormone Better
No. of
Participants
Parathyroid hormone
No. of
Cohorts
Relative Risk
(95% CI)
All-cause mortality
17 326
Adequate adjustment
elbamitse toN1
83
732
Partial adjustment
3 1.01 (0.99-1.03)
101 058
All studies combined
4 1.01 (1.00-1.02)
Cardiovascular mortality
17 326
Adequate adjustment
elbamitse toN1
5041
Partial adjustment
elbamitse toN1
22
367
All studies combined
2 1.05 (0.99-1.11)
Relative Risk (95% CI)
0.5 1.0 2.0
Increased Serum
Calcium Better
Decreased Serum
Calcium Better
No. of
Participants
Calcium
No. of
Cohorts
Relative Risk
(95% CI)
All-cause mortality
22 367
Adequate adjustment
2 1.07 (0.91-1.24)
83 789
Partial adjustment
6 1.09 (0.99-1.20)
106
156
All studies combined
8 1.08 (1.00-1.16)
Cardiovascular mortality
17
326
Adequate adjustment
elbamitse toN1
5041
Partial adjustment
elbamitse toN1
22
367
All studies combined
2 1.15 (1.08-1.23)
Relative Risk (95% CI)
Risks of all-cause mortality, cardiovascular mortality, and nonfatal cardiovascular events are shown per 1-mg/dL
increase in serum levels of phosphorus, 100-pg/mL increase in serum parathyroid hormone, and 1-mg/dL in-
crease in serum calcium. Summary estimates are not reported when only a single cohort contributed data. CI
indicates confidence interval.
Figure 19 | Summary estimates for risks of all-cause mortality and cardiovascular mortality associated with levels of serum
phosphorus, PTH, and calcium. PTH, parathyroid hormone. Reprinted with permission from Palmer SC, Hayen A, Macaskill P, et al. Serum
levels of phosphorus, parathyroid hormone, and calcium and risks of death and cardiovascular disease in individuals with chronic kidney
disease: a systematic review and meta-analysis. JAMA 2011; 305(11): 1119-1127.
387
Copyright & (2011) American Medical Association. All
rights reserved. Accessed http://jama.jamanetwork.com/data/Journals/JAMA/18301/jrv15003_1119_1127.pdf
86 Kidney International Supplements (2013) 3, 73–90
chapter 3

We have attempted to balance current knowledge with
published guidance statements in non-CKD populations.
Clarification of Issues and Key points
Clinical trials comparing strategies such as vitamin D
replacement, dietar y phosphate restriction, phosphate bin-
ders, and calcimimetics with placebo are needed to address
patient-level outcomes such as mortality and cardiovascular
morbidity in people with CKD.
Measurement of vitamin D l evels is problematic and
expensive and is not advocated here.
Vitamin D supplementation and bisphosphonates in people
with CKD
3.3.5: We suggest not to routinely prescribe vitamin D
supplements or vitamin D analogs, in the absence of
suspected or documented deficiency, to suppress
elevated PTH concentrations in people with CKD
not on dialysis. (2B)
RATIONALE
This statement is intended to highlight the lack of robust data
to support either the measurement or the treatment of
vitamin D deficiency in non-dialysis CKD populations. The
statement asks the clinician to more fully evaluate the
individual situation. The internationally accepted definition
of vitamin D deficiency is a blood concentration o20 ng/ml
(o50 nmol/l). Low 25(OH)D levels are common in patients
with non-dialysis dependent CKD; concentrations of
o15 ng/ml (o37 nmol/l) occur in at least 12-15% of patie nts
with CKD and are more prevalent at lower GFR levels, in
institutionalized subjects, at extremes of age, and in certain
racial groups. Deficiency of 25(OH)D increases fracture
risk and is associated with increased mortality. As CKD
progresses, levels of 1,25(OH)
2
D progressively fall and are
closely associated with increasing PTH concentrations. In
vitamin D-deficient subjects supplementation with vitamin D
increases BMD and muscle strength, reduces risk for fractures
and falls, and reduces PTH. In the absence of deficiency,
treatment with vitamin D and related compounds has not
been shown to improve either mortality or cardiovascular
outcomes.
Evidence Base
There is a substantial amount of data to support 25(OH)D
deficiency in general and CKD populations
367,393
which is
likely multifactorial. In addition to 25(OH)D deficiency,
note has been made that there is a progressive increase
in prevalence of 1,25(OH )
2
D deficiency with lower GFR
category, which occurs earlier than 25(OH)D deficiency
(Figure 20).
No relationship between 25(OH)D levels and 1,25(OH )
2
D
levels was apparent but there was a strong association
between 1,25(OH)
2
D deficiency and PTH concentration. Of
particular note, a higher urinary ACR was associated with
lower levels of 1,25(OH)
2
D at GFR values of o60 ml/min/
1.73 m
2
.
Table 29 | Phosphate binding agents in routine clinical practice and their ranked cost
Agent Dose/day Clinical experience and evidence base
Ranked
cost*
Aluminium hydroxide 1.425-2.85 g Extensive clinical experience in CKD and ESRD, no RCT comparison versus placebo.
Aluminium accumulates in bone and neural tissue with long-term use, avoids
calcium
1
Calcium citrate 1.5-3 g Limited trial evidence in ESRD. Reduction in phosphate and elevation in calcium
dose-dependent
2
Magnesium carbonate 0.7-1.4 g (plus calcium
carbonate 0.33-0.66 g)
Short-term RCT evidence in ESRD, less hypercalcemia 3
Calcium acetate and
Magnesium carbonate
combination
Calcium acetate 435 mg
plus
Magnesium carbonate
235 mg, 3-10 tablets daily
Short-term RCT evidence in ESRD, less hypercalcemia 3
Calcium carbonate 3-6 g Extensive clinical experience in CKD and ESRD, limited RCT evidence versus placebo.
Reduction in phosphate and elevation in calcium both dose-dependent
4
Calcium acetate 3-6 g Extensive clinical experience in ESRD; RCT evidence comparing to other binders.
Reduction in phosphate and elevation in calcium dose-dependent but less than with
calcium carbonate
4
Lanthanum carbonate 3 g Extensive prospective cohort evidence, RCT evidence compared to other phosphate
binders. Potential for accumulation in bone and other tissues, avoids calcium
5
Sevelamer-HCl 4.8-9.6 g Extensive prospective cohort evidence in ESRD; RCT evidence compared to other
phosphate binders; surrogate and patient-centered outcomes, avoids calcium
6
Sevelamer carbonate 4.8-9.6 g RCT evidence compared to other phosphate binders; equivalency studies compared
to sevelamer-HCl, avoids calcium
6
Abbreviations: CKD, chronic kidney disease; ESRD, end-stage renal disease; RCT, randomized controlled trial.
*The average annual cost of aluminium hydroxide in the UK, for example, is d51/year equivalent to US$84/year. The cost of lanthanum and sevelamer in the UK is 38-42 times
higher and the cost of calcium and magnesium-based binders 5-7 times higher than aluminium hydroxide (All drug costs derived from 2011 British National Formulary list
prices).
Kidney International Supplements (2013) 3, 73–90
87
chapter 3

The potential importance of these deficiencies has been
illustrated in a number of studies examining the relationship
between low levels of 25(OH)D and 1,25(OH)
2
D and all-cause
and cardiovascular mortality in a different cohorts.
393,394
Despite the associations with mortality, systematic review of
published data to date on vitamin D supplementation in
patients with CKD not on dialysis has only shown an
improvement in biochemical end points. A series of publica-
tions
395,396
have attempted to summarize the efficacy of
vitamin D therapy on biochemical, bone, cardiovascular, and
mortality outcomes in people with CKD and not requiring
dialysis. No formulation, route, or schedule of vitamin D
compound was found to alter the mortality risk or need for
dialysis although vitamin D compounds significantly lowered
serum PTH concentrations. None of the studies assessed
reported outcomes related to CVD, bone disease, or mortality.
International Relevance
The importance of vitamin D deficiency has been addressed in
both general and CKD populations. Specific populations have
been identified as more likely to be vitamin D deficient
depending on cultural and e n vironmental factors; estimates of
worldwideprevalenceofvitaminDdeficiencyrangefrom
25-60%. CKD populations within high-risk areas may be parti-
cularly vulnerable. The interplay between loss of kidney func-
tion and exacerbation of vitamin D deficiency is not known.
Implications for Clinical Practice and Public Policy
Vitamin D supplementation improves biochemical end
points similar to active vitamin D analogs with a lower
burden of costs and side effects. Measurement of serum
25(OH)D is expensive and in the CKD population with
vitamin D deficiency, simple vitamin D replenishment is all
that is indicated until new evidence becomes available. Except
for education or research purposes, there is no need to
measure vitamin D levels in general practice.
Areas of Controversy, Confusion, or Non-consensus
The appropriate dose and formulation of vitamin D, target
range for treatment, frequency, route of administration, and
safety at different severities of CKD remain to be determined.
3.3.6: We suggest not to prescribe bisphosphonate treat-
ment in people with GFR o30 ml/min/1.73 m
2
(GFR categories G4-G5) w ithout a strong clinical
rationale. (2B)
RATIONALE
The risk-benefit ratio of bisphosphonates has not been well
studied in CKD populations. Indications for bisphosphonate
therapy include osteoporosis, corticosteroid therapy, malig-
nant disease and Paget’s disease. In people with CKD and
GFR categories 460 ml/min/1.73 m
2
with osteoporosis and/
or at high risk of fractures, and in people with GFRs between
30-60 ml/min/1.73 m
2
with normal PTH, osteoporosis
and/or at high risk of fracture, treatment should be the same
as for the general population (although dose modification
may be necessary, see Table 30).
In people with CKD at lower categories of GFR
(o30-35 ml/min/1.73 m
2
), the correct diagnosis of osteo-
porosis becomes increasingly complex and other forms of
renal osteodystrophy requiring alternative management
strategies to osteoporosis require exclusion before treatment
with bisphosphonates is considered. In people with adynamic
renal bone disease, more likely at lower GFR categories, there
is a lack of evidence of either harm or benefit of bisphos-
phonates on bone strength or vascular calcification.
Evidence Base
Bisphosphonates increase BMD, reduce bone turnover, and
reduce the risk of fragility fractures. The bioavailability of
intravenous bisphosphonate formulations is 100% but the
bioavailability of oral formulations is only 1-5%. Approxi-
mately 50-80% of available bisphosphonate is taken up by
bone and the remaining 20-50% is excreted in urine without
being metabolized. Although oral bisphosphonates have not
been shown to adversely affect kidney function in people
with GFRs as low as 15 ml/min/1.73 m
2
,inpost hoc analysis
of clinical trial data their safety and efficacy below
GFRs of 30 ml/min/1.73 m
2
have not been well-validated
and intravenous bisphosphonates have been implicated
in nep hrotoxicity, especially when rapidly infused
(Table 30).
9,397,398
Thus, from a patient’s safety perspective,
the statement serves to limit exposure of those with abnormal
kidney function to these agents.
International Relevance
Given cost and clinical practice variation, the use of
bisphosphonates varies around the world. Thus, this state-
ment may be less applicable in different jurisdi ctions.
100
80
60
40
20
0
(n =61) (n=117) (n =230) (n =396) (n =355) (n =358) (n =204) (n =93)
80
79− 70
GFR level (ml/min)
Patients (%)
69− 60 59 − 50 49 − 40 39 − 30 29 − 20
< 20
Prevalence of 1,25(OH)
2
D
3
and 25(OH)D
3
deficiency
and intact PTH by GFR
25(OH)D
3
<15 ng /ml (<37 nmol /l)
(<57 pmol /l)
(>6.9 pmol /l)
1,25(OH)
2
D
3
< 22 pg /ml
Intact PTH > 65 pg/ml
Figure 20 | Prevalence of deficiency of 1,25(OH)
2
D
3
, 25(OH)D
3
,
and secondary hyperparathyroidism by GFR intervals. GFR,
glomerular filtration rate; PTH, parathyroid hormone. Adapted by
permission from Macmillan Publishers Ltd: Kidney International.
Levin A, Bakris GL, Molitch M, et al.
367
Prevalence of abnormal
serum vitamin D, PTH, calcium, and phosphorus in patients with
chronic kidney disease: results of the study to evaluate early
kidney disease. Kidney Int 2007; 71: 31-38; accessed http://
www.nature.com/ki/journal/v71/n1/pdf/5002009a.pdf
88 Kidney International Supplements (2013) 3, 73–90
chapter 3

Implications for Clinical Practice and Public Policy
Given the widespread use of bisphosphonates in developed
countries, especially in older women who are also likely to
have some degree of kidney dysfunction, the cessation of
bisphosphonates in that group may be problematic. There is
a need to monitor clinical practice and understand the
implications of this recommendation for large populations,
who may or may not be deriving benefit from these agents.
Areas of Controversy, Confusion, or Non-consensus
Despite these concerns , primary care data from the USA suggest
that prescription of bisphosphonates is common in people with
CKD.EveninthesubsetwithGFRo30 ml/min/1.73 m
2
,
bisphosphonate use was no less prevalent than in the non-CKD
population.
399
P eople with CKD in this study were nearly seven
times more likely to receiv e a bisphosphonate than active
vitamin D . Similar uncertainty about the efficacy and safety of
such treatment for people with CKD was demonstrated in a
studyofpeopleunderthecareofaUKrenalunit.
400
Half of
all people prescribed bisphosphonates had a GFR o30 ml/min/
1.73 m
2
and yet 50% of people with higher GFRs and
documented osteoporosis were not prescribed bisphosphonates.
Clarification of Issues and Key Points
Further study is requir ed to elucidate whether or not
bisphosphonates, through reduction of bone turnover in
people with pre-existing low bone turnover states, would be
beneficial or harmful both for bone and vascular calcification.
3.4 ACIDOSIS
The prevalence and severity of metabolic acidosis in people
with CKD progressively rises as GFR falls (Table 27).
Adaptations in acid excretion by the kidneys initially prevent
a fall in serum bicarbonate concentration but as GFR
continues to decline below 40 ml/min/1.73 m
2
, metabolic
acidosis commonly develops.
3.4.1: We suggest that in people with CKD and serum
bicarbonate concentrations o22 mmol/l treatment
w ith oral bicarbonate supplementation be given to
maintain serum bicarbonate within the normal
range, unless contraindicated. (2B)
RATIONALE
Serum bicarbonate concentrations less than 22 mmol/l are
associated with increased risk of CKD progression and
increased risk of death. Conversely, high serum bicarbonate
concentrations greater than 32 mmol/l are associated with
increased risk of death irrespective of the level of kid ney
function. Small studies of alkali supplementation have shown
reduction in progression of CKD and improved nutritional
status in people with CKD. This statement is therefore
worded to reflect the potential benefits of oral bicarbonate
supplementation to maintain serum bicarbonate concentra-
tions within the normal range, but the word ‘suggest’ in the
statement reflects the lack of robust evidence base on which
to support thi s statement.
Evidence Base
Chronic metabolic acidosis is associated with increased
protein catabolism, uremic bone disease, muscle wasting,
chronic inflammation, impaired glucose homeostasis, im-
paired cardiac function, progression of CKD, and increased
mortality.
401–410
The suggestion that treatment of acidosis
may be beneficial was first made by Richard Bright,
411
later
Table 30 | Summary data for bisphosphonates and CKD
Agent Indications
Dose, frequency and route of
administration
Special considerations for CKD and clinical trial
notes
Alendronate Postmenopausal osteoporosis
Corticosteroid use
10 mg daily, oral
70 mg weekly, oral
GFR o35 ml/min/1.73 m
2
: not recommended
No reported adverse events specific to CKD
Clodronate Malignancy-related bone disease 1.6-3.2 g daily, oral GFR o10 ml/min/1.73 m
2
: contraindicated
GFR 10–30 ml/min/1.73 m
2
: reduce dose by 50%
Etidronate Postmenopausal osteoporosis
Corticosteroid use
Paget’s disease
400 mg daily for 14 days, oral
5-10 mg/kg daily for up to 6 months
Mild renal impairment: reduce dose
Moderate or severe renal impairment: avoid
No data in CKD
Ibandronate Malignancy-related bone disease
Postmenopausal osteoporosis
150 mg monthly, oral
3 mg every 3-months intravenous
GFR o30 ml/min/1.73 m
2
: not recommended
No reported adverse events specific to CKD
Pamidronate Malignancy-related bone disease
Paget’s disease
15-60 mg single dose, intravenous
30 mg weekly for 6 weeks, intravenous
GFR o30 ml/min/1.73 m
2
: avoid
AKI reported
Risedronate Postmenopausal osteoporosis
Corticosteroid use
Paget’s disease
5 mg daily, oral
35 mg weekly, oral
GFR o30 ml/min/1.73 m
2
: contraindicated
No reported adverse events specific to CKD
Tiludronate Paget’s disease 400 mg daily for 3 months CrCl o30 ml/min: contraindicated
No data in CKD
Zoledronate Malignancy-related bone disease
Postmenopausal osteoporosis
Paget’s disease
4-5 mg single dose, intravenous GFR o30 ml/min/1.73 m
2
: avoid
GFR o60 ml/min/1.73 m
2
:
graded dose reduction
No data in CKD, AKI reported in non-CKD
Abbreviations: AKI, acute kidney injury; CKD, chronic kidney disease; CrCl, creatinine clearance; GFR, glomerular filtration rate.
Kidney International Supplements (2013) 3, 73–90
89
chapter 3

taken up enthusiastically by Arthur Osman
412
and further
explored by Lyon et al. who conducted a form of crossover
study of supplementation with bicarbonate in 17 people with
moderate kid ney failure and suggested that treatment with
alkali might preserve kidney function.
413
Rustom et al.
described a reduction in proximal renal tubular catabolism
and markers of tubular injury after oral NaHCO
3
in 11
people with mild/moderate renal impairment (mean
51
Cr-EDTA GFR 46.2
±
6.0 ml/min/1.73 m
2
) and proteinuria
(3.2
±
0.8 g/24 hours).
414
They suggested that oral sodium
bicarbonate may protect the proximal renal tubule and help
delay kidney disease progression. Mathur et al. randomized
40 subjects with mild to moderate CKD to oral bicarbonate
(1.2 mEq/kg of body weight) or placebo for 3 months and
suggested that correction of metabolic acidosis signific antly
attenuates the rise in blood urea.
415
A larger study randomly
assigned 134 adults with moderate to severe CKD (CrCl 15 to
30 ml/min/1.73 m
2
) and serum bicarbonate 16 to 20 mmol/l
to either supplementation with oral sodium bicarbonate
(1.82
±
0.80 g/day) or standard care for 2 years.
416
Primary
end points were rate of kidney function decline, the
proportion of subjects with rapid decline of kidney function
(43 ml/min/1.73 m
2
/yr), and ESRD (CrCl o10 ml/min
[o0.17 ml/s]). Secondary end points were DPI, normalized
protein nitrogen appearance, serum albumin, and mid-arm
muscle circumference. Serum bicarbonate increased signifi-
cantly in the bicarbonate group but despite the associated
sodium load there was no difference in BP control,
prescription of antihypertensives and loop diuretics, or
hospitalization for heart failure compared with the control
group. The decline in CrCl was significantly slowed in the
bicarbonate group compared w ith controls (5.93 versus
1.88 ml/min/1.73 m
2
,Po0.0001), rapid progression was less
prevalent (9 versus 45%, RR 0.15; 95% CI 0.06-0.40;
Po0.0001), and fewer subjects developed ESRD (6.5 versus
33%, RR 0.13; 95% CI 0.04-0.40; Po0.001). Nutritional
parameters also improved significantly with bicarbonate
supplementation. In a non-random ized study the effects of
supplementation of 1 mmol of bicarbonate equivalent per kg
body weight per day for 2 years using oral sodium citrate in
30 subjects with hypertensive nephropathy was compared
with 29 control subjects.
417
There were no statistically
significant differences in baseline demographic data, systolic
BP, venous bicarbonate levels, or parameters of kidney
function between the two groups. All subjects were receiving
angiotensin-converting enzy me inhibition throughout the
study and BP was controlled to similar levels in both groups
in a 6-month run-in period prior start of study. In those
treated with oral sodium citrate, urine end othelin-1 excretion
and N-acetyl-b-D glucosaminidase were each significantly
lower while the rate of GFR dec line was significantly slower
after 24 months of treatment compared to the control group.
International Relevance
Alkali supplementation appears to be a promising low-cost,
high-benefi t adjunct treatment for patients with CKD and
may be accessible to all populations. It is not known how
prevalent acidosis is among different communities or
countries around the world.
Implications for Clinical Practice and Public Policy
The use of sodium bicarbonate or calcium carbonate as a
source of alkali should not present financial hardships in
most practices or jurisdictions.
Areas of Controversy, Confusion, or Non-consensus
A potential concern regarding treatment of patients with
CKD with sodium bicarbonate or sodium citrate is the
possible associated sodium loading. A crossover study of
200 mmol/day (17 g/day) of sodium bicarbonate versus
200 mmol/day (12 g/day) of sodium chloride in 10 subjects
with severe CKD (CrCl 2.5-16.8 ml/min [0.04-0.28 ml/s]) on
an extremely low-sodium diet showed that sodium chloride
but not sodium bicarbonate produced an increase in weight
and BP.
418
A subsequent follow-up study suggested that if
dietar y sodium chloride intakes are near maximum tolerance,
then sodium bicarbonate supplementation should be accom-
panied by reductions in sodium chloride intake to maintain
sodium balance.
419
Given the lack of large and long-term
trials, many are sti ll not convinced that this is a reasonable
recommendation, and is thus controversial. We would like to
ensure that individual clinicians are aware of the controversy
rather than making no statement at all.
DISCLAIMER
While every effort is made by the publishers, editorial board,
and ISN to see that no inaccurate or misleading data, opinion
or statement appears in this Journal, they wish to make it clear
that the data and opinions appearing in the articles and
advertisements herein are the responsibility of the contributor,
copyright holder, or advertiser concerned. Accordingly, the
publishers and the ISN, the editorial board and their respective
employers, office and agents accept no liability whatsoever
for the consequences of any such inaccurate or misleading
data, opinion or statement. While every effort is made to
ensure that drug doses and other quantities are presented
accurately, readers are advised that new methods and
techniques involving drug usage, and described within this
Journal, should only be followed in conjunction with the drug
manufacturer’s own published literature.
90 Kidney International Supplements (2013) 3, 73–90
chapter 3

Chapter 4: Other complications of CKD: CVD,
medication dosage, patient safety, infections,
hospitalizations, and caveats for investigating
complications of CKD
Kidney International Supplements (2013) 3, 91–111; doi:10.1038/kisup.2012.67
This section addresses a series of important topics which
impact the outcomes of people with CKD. Specifically CVD,
interpretation of tests for CVD, infections, vaccinations and
hospitalizations, medications, and patient safety are ad-
dressed. This section is designed to help the practitioner
appreciate the specific nuances related to testing and drug
administration in people with CKD and to alert all clinicians
to the fact that many common tests may not be as useful in
people with CKD to diagnose or evaluate therapies. It was
beyond the scope of the guideline and evidence review to
thoroughly evaluate all tests with respect to sensitivity and
specificity in different groups of people with CKD but future
research may consider this as a useful direction.
4.1 CKD and CVD
Population-based studies have demonstrated an increased risk
of death and cardiovascular mortality as GFR falls below 60
ml/min/1.73 m
2
or when albumin is detected on urinalysis.
This is not explained by an increase in traditional risk factors.
There are CKD-specific risk factors associated with more
advanced CKD which drive the high rates of mortality and
morbidity even at young ages. People with CKD are more
likely to experience a cardiovascular event than to progress to
ESRD, have a worse prognosis with higher mortality after acute
myocardial infarction (MI), and have a higher risk of recurrent
MI, heart failure and sudden cardiac death. Management of
modifiable cardiovascular risk factors, such as improved BP
and diabetes control, also reduces CKD progression.
4.1.1: We recommend that all people with CKD be
considered at increased risk for cardiovascular
disease. (1A)
RATIONALE
This statement is worded in this way to reflect the strong and
independent associations between GFR and albuminuria
categories and risk of CVD in people with CKD and applies
to both adult and pediatric populations.
Evidence Base
Large cohort studies have demonstrated the strong and inde-
pendent associations between CVD (acute coronary syndrome
[A CS], stroke, heart failure and sudden cardiac death) and
CKD by category of eGFR, after adjusting for known CVD risk
factors, history of CVD events, and proteinuria. In those with
an eGFR of 45-59 ml/min/1.73 m
2
, risk is increased by 43%
and in those with eGFR below 15 ml/min/1.73 m
2
,riskis
increased by 343%.
58
Although people with GFR category G5
(GFRo15 ml/min/1.73 m
2
) are at the highest risk of a CVD
event, there will be more eve nts in people with GFR categories
G3a-G3b (GFR 30–59 ml/min/1.73 m
2
) because of the much
higher prevalence at these categories.
420
These events occur at a
younger age in people with CKD suggesting that CKD
promotes CVD at an accelerated rate.
421
The prognosis after
an acute event is related to level of GFR with a significant rise
in mortality when eGFR falls below 45 ml/min/1.73 m
2
.
422–424
Albuminuria is associated with duration and severity of
hypertension; an adverse lipid profile with higher levels of
total cholesterol, triglycerides and lipoprotein(a) and low
HDL-C levels;
425
and abnormalities of coagulation. The
presence of higher levels of proteinuria increases the risk of
mortality and MI independently of level of eGFR.
426
Many
studies have demonstrated low levels of urinary albumin to
be associated with the increased risk of CVD in people with
diabetes independent of renal function; however population
studies of non-diabetic individuals have confirmed that even
small amounts of albuminuria are associated with increased
CVD risk. In the Third Copenhagen study, in people with
microalbuminuria, risk of coronar y heart disease was
increased independently of age, sex, renal function, diabetes,
hypertension, and plasma lipids.
427
The Chronic Kidney
Disease Prognosis Consortium demonstrated that in general
practice cohorts there was an increase in cardiovascular
mortality when ACR is higher than 30 mg/g (3 mg/mmol).
4
Analysis of data from the Heart Outcomes Prevention
Evaluation (HOPE) study demonstrated that any degree of
albuminuria is a risk factor for cardiovascular events in
individuals with or without diabetes.
428
The lack of a
threshold of albuminuria for cardiac risk was also confirmed
in the HUNT 2 Study
429
and the Losartan Intervention For
Endpoint Reduction in Hypertension (LIFE) study in
patients with LVH.
430
Albuminuria and low eGFR were
synergistic cardiovascular mortality risk factors in the
HUNT 2 study and using both ACR and eGFR improved
http://www.kidney-international.org chapter 4
& 2013 KDIGO
Kidney International Supplements (2013) 3, 91–111 91

cardiovascular risk stratification at all age levels, but
particularly in persons 70 years and over.
429
In the MDRD Study cohort of patients with GFR
categories G3a-G4 (GFR 15–59 ml/min/1.73 m
2
), cystatin C
level was strongly associated with all-cause and cardiovas-
cular mortality particularly in the elderly.
431
Analysis of data
from MESA and the Cardiovascular Health Study (CHS)
demonstrated that the worst prognosis of CVD, heart failure,
and progressive CKD was found in those subjects with CKD
diagnosed using the cysta tin C-based equation.
432
LVH is common with CKD, is known to reflect target
organ damage, and is associated with increased cardio-
vascular mortality in CKD.
360
It is also important to consider
the role of CKD-specific risk factors particularly in patients
with more severe CKD (GFR o30 ml/min/1.73 m
2
) and
correct them where possible. Anemia has been thought to
have a par ticular role in the early development of CVD in
people with CKD. Although treatment of anemia is
associated with improved well-being and greater exercise
capacity, the results of several RCTs of anemia correction
have suggested that complete correction of anemia is not
advisable in people with lower GFR (o60 ml/min/1.73 m
2
).
A meta-analysis of 9 RCTs comparing different target Hb
levels suggests an increase in mortality and worse BP control
with the higher treatment targets, independent of the GFR
category.
433,434
Those patients with LVH at baseline, a sign of
target organ damage and which was present in 47% of the
Cardiovascular Risk Reduction by Early Anemia Treatment
with Epoetin Beta Trial (CREATE) cohort, had significantly
worse cardiovascular outcomes when treated to the higher
targets.
435
Abnormalities of mineral metabolism with low 1,25 -
dihydroxyvitamin D and PTH were found to occur early in
CKD in the Study for the Evaluation of Early Kidney disease
[SEEK] study, however serum calcium and phosphate were
usually normal until eGFR fell below 40 ml/min/1.73 m
2
.
367
An association between elevated serum phosphate and
cardiovascular mortality was demonstrated in a prospective
study of people with CKD and this was also thought to be
associated with low vitamin D levels.
436
Pediatric Considerations
While the majority of data for, and evidence of, the high risk
of cardiovascular morbidity and mortality in the pediatric
renal population stems from the ESRD (dialysis and
transplant) populations,
437–440
there is recognition that many
if not all of the factors responsible for such outcomes will
develop much earlier in this course of CKD. In fact, the
current American Heart Association’s guidelines for reduc-
tion of cardiovascular risk in pediatrics stratified the child
with CKD to the highest risk category.
441
Based on data from the CKiD trial, Wilson et al.
442
demonstrated extremely high prevalence rates for four
traditional cardiovascular risk factors (hypertension, dyslipi-
demia, abnormal glucose metabolism, and obesity) in a
subgroup of this cohort of 586 children with iohexol GFR
proven mild to moderate CKD. In this cross-sectional
analysis of 250 children who had data on all variables of
interest, median GFR of 45.2 ml/min/1.73 m
2
(IQR 34.6-
58.2), the prevalence of hypertension was 46%, dyslipidemia
was 44%, abnormal glucose metabolism was present in 21%,
and obesity in 15%. Hypertrig lyceridemia was common, seen
in 33%. In aggregate, only 26% of the cohort had no risk
factors present, 39% had at least one and 22% had two, 11%
had three and 2% had all four risk factors present. The
prevalence of all these factors remained elevated even when
the population was restricted to those defined as lean, BMI
o85
th
. In this group, the presence of 2 or 3 cardiovascular
risk factors was still seen in 20% and 2%, respectively. Within
the lean group 12% demonstrated abnormal glucose
metabolism. Finally, multivariate regression analysis of their
data demonstrated that glomerular disease as a cause of CKD
and the presence of nephrotic range proteinuria were
associated with much higher odds of having cardiovascular
risk factors, 1.96 (95% CI 1.04-3.72) and 2.04 (95% CI 0.94-
4.43), respectively.
It is also well-recognized that carotid intimal media
thickness and pulse wave velocit y changes, as potential
surrogate measures for vascular damage in children, can be
increased in in children with CKD.
443,444
For fur ther detailed discussions of the pathophysiology
and complexity of CVD in the pediatric CKD population,
two recent review papers from Mitsnefes
445
and
Shroff et al.
446
are suggested. Both papers identify and
discuss the pathophysiology of both traditional and CKD-
specific cardiovascular risk factors as seen in this population.
4.1.2: We recommend that the level of care for ischemic
heart disease offered to people with CKD shoul d
not be prejudiced by their CKD. (1A)
4.1.3: We suggest that adults with CKD at risk for
atherosclerotic events be offered treatment with
antiplatelet agents unless there is an increased
bleeding risk that needs to be balanced against the
possible cardiovascular benefits. (2B)
RATIONALE
These statements are worded in this way to reflect the
growing body of evidence that suggests that treatment of
traditional risk factors in CKD patients is of benefit, and that
attempts to modify or preclude usual investigations and
therapies are not necessary in CKD patients. Inasmuch as
CKD populations have been understudied, but likely
inadvertently included in larger population studies,
the rationale for risk factor modification is similar to that
in the general population. Modification of risk factors
includes:
1. Smoking cessation
2. Exercise
3. Weight reduction to optimal targets
4. Lipid modification recognizing that the risk reduction
associated with statin therapy in adults w ith CKD is
92 Kidney International Supplements (2013) 3, 91–111
chapter 4

relatively constant across a broad range of baseline low-
density lipoprotein cholesterol (LDL-C) levels
5. Optimal diabetes control HbA
1C
o7% (53 mmol/mol)
6. Optimal BP control to o140/90 mm Hg or o130/80
mm Hg in those with CKD and depending on the degree
of proteinuria (see Recommendations 3.1.4 and 3.1.5)
7. Aspirin is indicated for secondary prevention but not
primary prevention
8. Correction of anemia to individualized targets (see
KDIGO Clinical Practice Guideline for Anemia in CKD
11
)
Evidence Base
The maintenance of cardiovascular health ultimately
impacts ‘kidney health’ given the links between cardiac
function, renal perfusion, atherosclerosis, and glomerulo-
sclerois. The details of this complex biology are beyond
the scope of these guidelines, but can be found in key
references below.
There is evidence in the general population that smoking
cessation is associated with a reduction in cardiovascular risk.
In CKD there is evidence that smoking is associated with
progression of renal disease, though no specific data exist that
support cessation of smoking and delay of progression.
447
People with CKD have reduced levels of muscle strength
and aerobic activity. In those not on dialysis, exercise training
is shown to improve functional capacity and BP.
448
Weight loss in obese CKD patients can reduce rate of
decline, proteinuria, and BP.
354
The Study of Heart and Renal Protection (SHARP)
449
is
the largest RCT in people with CKD to date. The results
demonstrate that a lipid-lowering strategy which included
fixed dose simvastatin and ezetimibe resulted in a 17%
reduction in atherosclerotic events, as compared to placebo.
The cohort enrolled included those with eGFR under 60 ml/
min/1.73 m
2
, age greater than 40 years, and was an
international study including more than 9000 subjects from
around the globe. The lipid-lowering strategy was effective
and safe. Lipid targets have not been established specifically
for people w ith CKD. Treatment strategies should be
implemented in accordance with current recommendations
for high-risk populations.
At the time of this writing , the KDIGO Clinical Practice
Guideline for Lipid Management in Chronic Kidney Disease
was under preparation for public review. In brief, the key
aspects of the draft recommendations include treating those
at high risk for atherosclerotic disease with lipid-lowering
therapies, regardless of LDL levels, in those 50 years of age
and above. Since this Guideline has not yet been finalized,
interested readers should refer to the final document when it
is formally released in 2013.
The benefits of aspirin in people with CKD and
hypertension was demonstrated in a post hoc analysis of the
Hypertension Optimal Treatment (HOT) trial.
450
Jardine
et al. reported that among every 1000 persons with eGFR
o45 ml/m in/1.73 m
2
treated for 3.8 years, 76 major
cardiovascular events and 54 all-cause deaths will be
prevented while 27 excess major bleeds will occur. They
concluded that an increased risk of major bleeding appears to
be outweighed by the substantial benefits. Clopi dogrel is used
as an alternative to aspirin but CKD has been shown to be
associated with an increase in platelet reactivity and there is
resistance to clopid ogrel in people with CKD, diabetes, and
CVD.
451
The Clopidogrel for Reduction of Events During
Observation (CREDO) trial concluded that clopidogrel in
mild or moderate CKD may not have the same beneficial
effect as it does in people without CKD. Subjects with normal
renal function who received 1 year of clopidogrel had a
marked reduction in death, MI, or stroke compared with
those who received placebo (10.4% versus 4.4%, P o0.001),
whereas those with mild and moderate CKD did not have a
significant difference in outcomes with clopidogrel therapy
versus placebo (mild: 12.8% versus 10.3%, P ¼ 0.30;
moderate: 13.1% versus 17.8%, P ¼ 0.24). Clopidogrel use
was associated with an increased RR of major or minor
bleeding, but this increased risk was not different based on
renal function.
452
International Relevance
Although it is clear and mentioned in many guidelines that
CKD is associated with an increase in CVD risk, it is not
included in many assessment tools and there is a deficiency in
ethnicity and regional specific tools.
The assessment tool from US Third Report of the National
Cholesterol Education Program (NCEP III) does not include
CKD. The Fourth Joint Task Force of the European Society of
Cardiology suggest that both an eGFR of less than 60 ml/
min/1.73 m
2
and albuminuria increase risk but do not
quantify the risk or include CKD in their Systematic
COronary Risk Evaluation (SCORE) assessment tool.
453
In
the UK, the QRISK
s
2 online tool includes CKD as ‘yes’ or
‘no’ and therefore does not allow for level of eGFR or
proteinuria.
454
The Joint British Societies Guidelines regards
proteinuria a s a sign of target organ damage, which conveys a
risk of at least 20% in 10 years.
455
A study of cardiovascular risk estimation in Chinese adults
in the USA–People’s Republic of China Collaborative Study of
Cardiovascular and Cardiopulmonary Epidemiology (USA-
PRC Study) did not include CKD as a risk factor at all.
456
It is
also important to remember that in this population stroke
was the predominant CVD. A review of 25 risk assessment
tools identified only 2 derived from an Asian population.
457
However, links between GFR categories and CVD events are
evident in the Asian population, for example in the Japanese
Gonryo study.
458
A prospective study in a general Japanese
population demonstrated links between lower GFR, high
creatinine levels, and proteinuria with cardiovascular mor-
tality particularly stroke.
459
In Chinese patients who were at
least 50 years old and either had existing CVD or were at high
risk, 34% had an eGFR o60 ml/min/1.73 m
2
and eGFR o45
ml/min/1.73 m
2
was found to be an independent predictor of
all-cause and cardiovascular death.
460
In a population-based
study of 2353 people aged over 40 years in Beijing, an eGFR
Kidney International Supplements (2013) 3, 91–111 93
chapter 4

o90 ml/min/1.73 m
2
was associated with increased CVD
risk, and for each CKD category stroke was more prevalent
than MI.
461
In India the increasing prevalence of type 2 diabetes is
driving an increase in CKD and both are associated with
increased cardiovascular risk. In CKD patients attending one
clinic in North India, 28% had diabetes, 27% were overweight,
and 92% had hypertension. Metabolic syndrome as defined by
the International Diabetes Federation (IDF) 2007 guidelines
was present in 39% and more frequent in women.
462
Implications for Clinical Practice and Public Policy
A full CVD risk assessment in a person with CKD should
include an estimate of GFR and a quantitative assessment of
albuminuria. Cystatin C may be helpful at risk stratifying
those at intermediate risk.
The key aspect of these recommendations is to ensure that
people with CKD are not deprived of treatment strategies
known to be effective in general populations.
Areas of Controversy, Confusion, or Non-consensus
Many of the traditional cardiovascular risk assessment tools
do not adjust for the presence of early CKD. Any tool should
also adjust for level of eGFR because of increasing risk
associated with lower levels of kidney function. The
Framingham risk equation underestimates true events in a
CKD population, but no validated different tools exist at
present which better quantify cardiovascular or mortality risk
in CKD populations.
Proteinuria is considered to be a sign of target organ
damage and thus associated with high cardiovascular risk.
However, assessment of risk should also include the presence
of albuminuria. Few studies exist which demonstrate that
targeting lowering of proteinuria results in reduced cardio-
vascular risk, though a number of studies which have
targeted interruption of the RAAS (and in which lowering
of urine protein has also been shown) have demonstrated a
benefit.
An appropriate risk assessment tool should be available to
assess risk in people with CKD. Addition of the presence of
CKD to conventional risk factors in the Reykjavik population
increased discrimination but did not increase risk to the same
degree as smoking or diabetes.
463
In the Framingham Heart
Study, GFR category 3b (GFR 30–44 ml/min/1.73 m
2
)was
associated with CVD but not equivalent to previous CVD.
464
Lifestyle modification has not been studied in large trials
in people with CKD but smoking cessation, achievement of
optimum weight, regular exercise, and salt restriction should
be seen as reasonable aims.
Pediatric Considerations
It is presently unclear whether children w ith CKD and
elevated lipid levels will benefit from lipid-lowering strategies
with no prospective trials ongoing in this area. It is hoped
that data from both CKiD
55
and the 4C trials
78
may be able to
address these shortcomings.
However, given that these children demonstrably have
elevated risk of cardiovascular and atherosclerotic disease (see
Recommendation 4.1.1), that there are data supporting the
use of a number of statins in the pediatric population,
465
and
that the adult ev idence for benefit of statins in a subset of the
SHARP trial
449
with a GFR o60 ml/min/1.73 m
2
exist, it
would seem reasonable to consider the use of such drugs in
children with CKD and elevated lipids.
Recommendations from 2006
441
for use of dietary
modifications, followed by statins in children older than 8
years of age with persistent elevation in LDL-C levels, have
been endorsed by the American Academy of Pediatrics.
However, at the time of publication of these guidelines, the
KDIGO Dyslipidemia Work Group has suggested that statin
therapy may not be appropriate.
Age- and renal function-adjusted doses of such drugs
should be carefully considered prior to any therapy being
initiated (see Recommendation 4.4.1).
Regarding diabetes control, treatment in keeping with
national and international diabetes recommendations is
prudent. Note is made that there are specific caveats with
respect to drugs and side effects that are important (see
Recommendations 3.1.15-3.1.18).
There is no literature in the area of antiplatelet agents in
atherosclerotic disease in the subset of children with CKD
and the suggestion to offer antiplatelet agents does not apply
to pediatric practice.
4.1.4: We suggest that the level of care for heart failure
offered to people with CKD should be the same as
is offered to those without CKD. (2A)
4.1.5: In people with CKD and heart failure, any
escalation in therapy and/or clinical deterioration
should prompt monitoring of eGFR and serum
potassium concentration. (Not Graded)
RATIONALE
This statement is worded this way to make clinicians aware
that people with CKD and heart failure should receive at least
equal benefit from heart failure therapy as those without
CKD, but that heart failure therapies (in particular increase
of RAAS blockade and diuretic therapy) may lead to
significant changes in GFR and serum potassium concentra-
tions. This does not imply that such therapy should be
avoided but only that clinicians are cognizant of this
possibility, monitor it, and understand it in the context of
individual risks and benefits.
Heart failure is a complex clinical syndrome which can be
caused by any structural or functional cardiac disorder that
impairs the pump function of the heart and has a high
mortality. Within the general population, the commonest
causes of heart failure are ischemic heart disease causing left
ventricular systolic dysfunction, and hypertensive heart
disease with left ventricular hypertrophy and diastolic
dysfunction. Thus people with CKD are at increased risk of
both. Evidence-based medication for heart failure impacts on
94 Kidney International Supplements (2013) 3, 91–111
chapter 4

GFR and prescribers need to be alert to this. Changes in GFR
are often transient and the downward trajectory GFR is not
sustained if all other aspects of care and clinical status are
stable. Monitoring and close follow-up are required.
Evidence Base
In an outpatient setting, normal GFR is unusual in heart
failure. A study from the Alberta Heart Function Clinic found
that in people with an ejection fraction of r35%, GFR
categories G3a and G3b (GFR 30–59 ml/min/1.73 m
2
)were
present in 40% and 16% of people were GFR categories G4 or
G5 (GFRo30 ml/min/1.73 m
2
).
466
This study demonstrated a
1% increase in mortality associated with each 1 ml/min fall in
CrCl demonstrating the clear link between kidney function
and outcome for people with CKD and heart failure. Meta-
analysis of 16 studies and over 80,000 people with heart failure
by Smith et al. showed that renal impairment was present in
63% and was associated with increased mortality across the
range of kidney function, with an adjusted HR of 1.56 (95% CI
1.53-1.60).
467
In the Candesartan in Heart Failure-Assessment
of Reduction in Mortality and Morbidity (CHARM) Program
with candesartan in heart failure, 36% of subjects had eGFR
o60 ml/min/1.73 m
2
which was associated with increased
mortality and hospital admission regardless of ejection
fraction.
468
The Digitalis Intervention Group (DIG) identified
a GFR of 50 ml/min as a risk threshold for increased mortality
risk in heart failure.
469
Smith et al. demonstrated that 1-year
mortality after admission for heart failure was 29% in people
with normal kidney function but 52% in those with moderate
to severe renal impairment.
470
Anand et al. also demonstrated
an independent association between the presence of positive
reagent strip proteinuria present in 8% of subjects and
increased mortality in heart failure.
471
In the Alberta Provincial Project for Outcome Assessment
in Coronary Heart Disease (APPROACH) study of people
with heart failure related to coronary artery disease (CAD),
39% had CrCl o60 ml/min (o1 ml/s). This study also
demonstrated that the level of kidney function was a better
indicator of poor outcome than cardiac anatomy.
472
In
people admitted to hospital with heart failure, increasing SCr
concentration is associated with a longer hospital stay and
worse outcome.
473
LVH is present in 40% of people with CKD,
474
progressively increasing as kidney function declines.
475
Ha et al. demonstrated LVH in 87% of people w ith
predialysis CKD.
476
Parfrey et al. in a study of 432 people
who were predialysis, found that only 16% had a normal
echocardiogram while 41% had concentric LVH, 16% systolic
dysfunction, and 28% left ventricular dilatation.
477
LVH is
related to increased work load caused by arterial stiffness and
hypertension, and at lower levels of GFR, volume overload.
Alterations in electrolyte balance, anemia, bone metabolism,
uremia, oxidative stress, inflamm ation, and other inflamma-
tory mechanisms all play a role.
478
In the majority of people with CKD, the myocardium
develops concentric hypertrophy
476
with interstitial fibrosis
and CKD-associated cardiomyopathy, leading to left ventri-
cular stiffness.
474
This results in diastolic heart failure; people
with CKD have higher mortality from diastolic heart failure
than systolic heart failure.
479
Diastolic hear t failure is more
common in people with CKD who are older females with
hypertension and/or diabetes.
480
Left ventricular hypertrophy is particularly associated with
anemia and a fall in Hb to less than 12.8 g/dl (128 g/l) is
associated with left ventricular growth in people with early
CKD.
481
Lower Hb levels are associated with increased risk of
death and hospitalization in heart failure.
482
Other cardio-
vascular conditions, for example MI and atrial fibrillation, are
more common in CKD and may exacerbate heart failure.
It is important to note that in a study of an older
population (age 464 years), heart failure was an indepen-
dent predictor of rapid kidney function decline.
483
Standard therapy for the management of systolic heart
failure includes an ACE-I or ARB and a beta-blocker licensed
for heart failure such as bisoprolol or carvedilol. Meta-
analysis of 25 trials of RAAS blockade (ACE-I or ARB) in
people with CKD or proteinuria, demonstrated a reduction
in the risk for heart failure and reduction in cardiovascular
outcomes.
484
Subgroup analysis from the Valsartan Heart
Failure Trial (Val-HeFT) demonstrated the beneficial effect of
valsartan in people with CKD and that valsartan reduced the
eGFR by the same amount in people with and without
CKD.
471
Candesartan was also clinically effective in the
CHARM study regardless of underlying kidney function
468
while ir besartan was associated with a 23% reduction in
hospital admissions related to heart failure in people with
nephropathy due to type 2 diabetes.
485
Analysis of data on
bisoprolol from the Cardiac Insufficiency Bisoprolol Study II
(CIBIS II)
486
identified 32% of subjects with eGFR o60 ml/
min/1.73 m
2
. These subjects were more likely to die or be
admitted to hospital than those with eGFRZ60 ml/min/1.73
m
2
. However, they showed a similar benefit from bisoprolol
treatment. A post hoc analysis of data on carvedilol from the
Carvedilol Post-Infarct Survival Control in Left Ventricular
Dysfunction (CAPRICORN) and Carvedilol Prospective
Randomized Cumulative Survival (COPERNICUS) trials
identified 60.8% of participants with eGFR o60 ml/min/
1.73 m
2
.
487
Carvedilol was well tolerated by subjects with
CKD, and decreased all-cause, cardiovascular, and heart
failure mortality. However, doubts were raised over the
benefits when eGFR o45 ml/min/1.73 m
2
.
Aldosterone antagonists are included in standard heart
failure management, usually in addition to an ACE-I or ARB,
leading to concerns relating to risk of hyperkalemia,
particularly in people with lower GFRs. In a study of
spironolactone therapy in severe heart failure, Pitt et al.
488
included subjects with SCr concentrations up to 2.5 mg/dl
(221 mmol/l). There was a 30% reduction in mortality with
spironolactone and the incidence of hyperkalemia was low.
However, a review of real-world practice demonstrated a
significant increase in hyperkalemia with the combination of
RAAS blockade and an aldosterone antagonist, highlighting
Kidney International Supplements (2013) 3, 91–111 95
chapter 4

the need for close monitoring with the introduction of
combination therapy.
489
In the APPROACH study on the specialist management
of heart failure, subjects with CAD and CKD, despite
being at high risk, were less likely to be taking ACE-Is,
beta-blockers, statin, and aspirin. This study demonstrated
that those subjects taking beta blockers had lower mortality,
however ACE-Is were only of benefit when eGFR was
460 ml/min/1.73 m
2
.
472
International Relevance
The treatment of heart failure is similar around the globe. The
ability to closely monitor kidney function or to offer
conventional therapies may differ, however. Irrespective, this
statement should hold true internationally. The National Heart
Care Project of community subjects admitted to hospital with
heart failure studied the differences between people of black
and white ethnic groups. People with worse renal function
were more likely to be black, older and female, and black
people had a greater prevalence of hypertension and diabetes
but less ischemic heart disease. Black people had a lower risk of
mortality at every level of creatinine, for every 0.5 mg/dl
(44.2 mmol/l) increase in creatinine, 1-year death risk increased
10% in black people and 15% in white people.
470
Implications for Clinical Practice and Public Policy
Cardiorenal syndrome, an impairment of kidney function in
the presence of heart disease, is a marker of worse prognosis.
In addition to GFR, the presence and severity of albuminuria
and anemia aid in prognostication and management.
490
Where the link between LVH and heart failure in CKD is
so strong and the outcome poor for people with LVH, efforts
should be made to optimize care particularly BP. There is a
lack of robust evidence for the optimal medical management
of heart failure specifically in people with CKD; however, the
prevalence of people with early CKD is so high in heart
failure trials that we can apply standard treatment to these
people. For those people with eGFR o45 ml/min/1.73 m
2
,
the situation is less clear.
487
Areas of Controversy, Confusion, or Non-consensus
There are difficulties in making the diagnosis of heart failure
in people with CKD, especially diastolic heart failure.
Biomarkers such as B-type natriuretic peptide (BNP) may
be abnormally elevated in CKD, but may or may not
be as responsive to treatment or diagnostically accurate in
people with CKD (see Chapter 4.2). There is little evidence
to guide the management of left ventricular systolic
dysfunction in people with CKD and evidence for the
management of diastolic heart failure is generally lacking.
However, there is evidence that medications to improve the
outcome of peop le with heart failure are underused in people
with CKD.
Appropriate management of the metabolic complications
of kidney disease which can exacerbate heart failure needs to
be clarified. Clinical studies are required on the role of
device therapy in people with CKD (e.g., pacemakers,
defibrillators, etc.).
4.2 CAVEATS WHEN INTERPRETING TESTS FOR CVD IN
PEOPLE WITH CKD
BNP/N-terminal-proBNP (NT-proBNP)
As GFR diminishes, the prevalence and severity of cardio-
vascular abnormalities increase and are accepted as major
causes of morbidity and mortality in people with CKD. The
presence of congestive heart failure (CHF) in people with
CKD conveys a worse prognosis than either condition alone.
Therefore, early diagnosis and aggressive management of
CHF are both highly desirable. A number of cardiac
biomarkers (i.e., BNPs and cardiac troponins) appear to
have both increasing clinical importance and increasing
facility for detection and stratification of CHF. The stimulus
for secretion of these biomarkers is the hemodynamic load
(i.e., myocardial stretch) and their secretion is associated with
the severity of CHF and the degree of left ventricular
dysfunction. They are thus useful markers for diagnosis,
management, and prognosis in people with normal renal
function. However, when the eGFR is less than 60 ml/min/
1.73 m
2
, the accuracy of plasma BNP and NT-proBNP levels
for detection and stratification of CHF becomes unreliable
and the degree of responsiveness to treatment is not known.
4.2.1: In people with GFR o60 ml/min/1.73 m
2
(GFR cate-
gories G3a-G5), we recommend that serum concen tra-
tions of BNP/NT - proBNP be interpreted with caution
and in relation to GFR with respect to diagnosis of
heart failure and assessment of volume status. (1B)
RATIONALE
This statement is intended to remind clinicians that although
the prevalence of fluid overload and heart failure increases
with lower GFR categories, BNP becomes less reliable as a
predictor of fluid overload and heart failure at lower GFRs.
While it is associated with worse outcomes, cutoff values
indicative of heart failure in general populations may or may
not be appropriate and changes in values with treatment may
or may not have the same meaning.
Evidence Base
Natriuretic peptides belong to a family of circulatory peptide
hormones from either myocardial cell orig in (atrial natriure-
tic peptide [ANP] and BNP) or endothelial cell origin
(C-ty pe).
491,492
They play an important role in regulating BP
and body fluid volume by their natriuretic and diuretic
actions, arterial dilatation, and inhibition of the RAAS.
493
Natriuretic peptides for the diagnosis of CHF have been a
diagnostic breakthrough in cardiology where determination
of their concentration in serum can help identify patients
with left ventricular systolic dysfu nction.
494,495
Concentrations of natriuretic peptides increase in
people with CHF and other CVDs as a consequence of
96 Kidney International Supplements (2013) 3, 91–111
chapter 4

pressure and volume overload.
496,497
Among people with
progressive LVH, concentrations of NT-proBNP are typically
elevated in proportion to the degree of left ventricular mass
increase.
Overall, BNP levels provide a better index of left
ventricular mass and load than do ANP levels and BNP has
emerged as a superior biomarker to ANP for clinical
applications involving CHF and left ventricular dysfunc-
tion.
491,498
NT-proBNP may have analytical advantages over
BNP because of greater stability due to a longer half-life.
The cause of an elevated BNP level is multifactorial in
origin and may reflect cardiac dysfunction, and/or changes in
renal function.
499
Since renal dysfunction intrinsically was
shown to affect BNP levels in some studies, the diagnosti c
value of BNP levels in the presence of CKD has been
questioned.
500,501
Earlier research hypothesized that measurement of
natriuretic peptides could be of use in monitoring
excess fluid volume and dry weight in people requiring
dialysis,
502,503
but this remains unproven and the significance
of plasma natriuretic peptides and their clinical role in
dialysis remains unclear.
491
Although several studies revealed higher BNP levels as
GFR declined, outcomes of other studies suggest that the
relationship between renal function and BNP levels may be
most strongly dependent on cardiac and volume-related
factors.
504–506
To test this hypothesis, Tagore et al. studied
BNP levels in a cohort of 143 clinically euvolemic subjects
with CKD in whom absence of heart disease was cli nically
validated and found that plasma BNP levels were indepen-
dent of GFR.
504
Suresh and Farrington
507
studied people on
dialysis and concluded that BNP levels were predictive of
presence of left ventricular dysfunction, cardiac events, and
survival in the presence of ESRD, suggesting that BNP levels
may be informative across the full range of renal function and
even in its absence. As previously estab lished by several
studies, the level of NT-proBNP is a strong prognostic
marker in both the general population
508
and in various
disease states, e.g., acute and CHF,
509,510
coronary heart
disease,
511,512
and hypertension.
513
International Relevance
The issue of how to differentially interpret BNP in the
presence or absence of CHF and within the context of
declining renal function has received considerable
attention but remains incompletely resolved from a clinical
perspective. Changes in either renal or cardiac function could
potentially impact levels of natriuretic peptides and incom-
plete knowledge of an individual’s clinical status relative
to the degree of cardiac pathology or renal dysfunction
could confound interpretation of BNP levels. Given the cost
of biomarkers assays and the uncertainties related to
interpretation of serum levels of BNP/NT-proBNP in people
with CKD, competing priorities for health-care resource may
dictate reliance on clinical evaluation and local practices
of care in some areas of the world. Until better data are
available, clinicians are asked to use this laboratory
measurement with caution as a diagnostic test for heart
failure in those with CKD.
Implications for Clinical Practice and Public Policy
Since people with ESRD with elevated levels of BNP have a
higher RR of death, this biomarker could be used as an
individual risk marker with the aim of identifying those at
high risk. Clinic al correlation is of utmost importance to
ensure accurate diagnosis and appropriate therapy.
Studies to understand the variability of BNP by categories
of eGFR and urine ACR, within and between individuals over
time, and the response to therapy should be undertaken so as
to better inform clinicians.
Areas of Controversy, Confusion, or Non-consensus
The relationship between renal function and BNP, the role of
natriuretic peptides as markers of fluid overload and as
predictors of mortality, and the utility of serial monitoring of
BNP/NT-proBNP levels in people with CKD, and respon-
siveness to therapy remains unknown.
Clarification of issues and key points
Natriuretic peptides may be surrogate markers for prediction
of mortality in people with CKD reflecting the association
of mortality with left ventricular dysfunction and LVH.
Before routine measurements of these biomarkers are
recommended, their utility in guiding or changin g clinical
practice should be assessed.
Pediatric Considerations
There is robust literature in both children and neonates
regarding both the normative values of BNP
514
and its value
in predicting outcomes of pediatric heart failure.
515
To date
no studies in children specifically address the issue of reduced
GFR in this population. However, application of similar
principles would likely apply.
Troponins
In recent years newer markers of myocardial injury have been
introduced into clinical practice.
516,517
Among these, cardiac
troponins have proven to be specific markers of myocardial
damage.
518,519
Cardiac troponins have been considered the
gold standard biochemical test for diagnosis of myocardial
damage because they have nearly absolute myocardial tissue
specificity, as well as high sensitivity, thereby reflecting even
microscopic zones of myocardial necrosis.
517,520
Further-
more, increasing evidence indicates that abnormal troponin
measurements identify a subgroup of patients who have an
increased risk of major cardiac events
519,521–525
and that
measurement of cardiac troponin T (cTnT) prior to
commencing renal replacement is a significant independent
predictor of sur vival.
526
However, these markers are renally
excreted and are often found to be elevated in people with
CKD irrespective of specific symptomatology. Thus, there is
uncertainty as to the clinical importance of their different
Kidney International Supplements (2013) 3, 91–111 97
chapter 4

magnitudes of elevation in CKD. There are no data on
‘expected’ values of these markers in the context of CKD, or
at different categories of GFR and albuminuria.
4.2.2: In people w ith GFR o60 ml/min/1.73 m
2
(GFR
categories G3a-G5), we recommend that serum
concentrations of troponin be interpreted with
caution with respect to diagnosis of acute coronary
syndrome. (1B)
RATIONALE
This statement is intended to remind clinicians that isolated
elevated serum concentrations of troponin (in the absence of
symptoms or electrocardiographic changes) may not necessa-
rily portend a diagnosis of ACS at lower GFR categories. While
there are data linking troponin elevations with poor outcomes,
the specific implications for an individual in clinical practice
are not clear. Hence, this statement simply cautions the
clinician with respect to interpretation but does not suggest
that the laboratory values are not of significance. In a patient
with clinical symptoms suggestive for ischemic heart disease,
an elevated cardiac troponin I (cTnI) is suggestive for ACS.
Evidence Base
cTnT and cTnI are low-molecular-weight proteins that form
part of the troponin complex and are integral components of
the myofibrillar contractile apparatus of the heart.
527
Loss of
integrity of cardiac myocyte membranes causes release of
cardiac troponins into the circulation, which can be detected
by highly sensitive assays developed for cTnT and cTnI to
diagnose ACS.
521
A potent stimulus for the release or
production of natriuretic peptides is mechanical stretch-
ing,
528
whereas plasma cTnT specifically and sensitively
reflects myocardial injury and is considered to be an index
of irreversible myocardial change.
521
In addition, cTnT levels
can predict multi-vessel CAD in people requiring dialysis.
529
However, the significance of screening people with CKD for
CAD using these cardiac biomarkers is questionable
530
since
increases in serum cardiac troponin concentration can occur
in the absence of an ACS.
531
Most studies
517,532–545
have focused on people with ESRD
where increases in serum cTnT concentrations have been
observed in 20%–90% of subjects
540
but generally much
lower when cTnI was measured. Information about cardiac
troponins and their relationship with comorbidity is sparse
in people with CKD who are not receiving dialysis
treatment.
526,527,546,547
Increased cardiac troponin concentra-
tions may occur early in CKD, including GFR categories
30-59ml/min/1.73m
2
, and more commonly as CKD advances.
Powerful prognostic data emerging from the dialysis
population demonstrate that increased concentrations of
cardiac troponins reflect either subclinical myocardial
damage caused by silent ischemia or myocardial remodeling
in the development of LVH.
539,542
Increased levels of cTnT measured with the highly
sensitive assay, well below the detection range of standard
assays, have been shown to be associated with cardiac
structural abnormalities including LVH (both left ventricular
wall thickening and dilation) and left ventricular systolic
dysfunction,
548
and to have a graded association with all-
cause and CVD mortality independent of traditional risk
factors, renal function, and levels of other biomarkers such as
high sensitivity CRP and NT-proBNP.
These findings suggest that chronic elevation of troponin
levels may be mediated to a greater extent by indices of heart
failure (such as higher left ventricular mass, left ventricular
dysfunction, or increased NT-proBNP levels) than indices of
atherosclerosis or ischemia.
548
International Relevance
The costs of these tests, the differing sensitivity of the assays
available, the paucity of data in people with CKD, and the
poor correlation of serum troponin levels with ACS in people
with CKD should be weighed against competing local health-
care priorities and resource before their introduction into
routine clinical practice. Emp hasis should be placed on the
clinical context and local standard practices of care.
Implications for Clinical Practice and Public Policy
Troponin elevation may result from repeated episodes of
clinically silent MI,
522,549
although CAD frequently underlies
troponin elev ation and increased cTnT is also frequently
encountered in patients at low risk for CAD. Troponin
elevations are as frequent in subjects without CAD as in those
with known CAD
517,550–552
and in people with CKD, serial
increasing cTn levels may be of greater utility for diagnosis of
ACS. In the context of symptoms and signs of ACS,
elevations of troponins should not be ignored nor incorrectly
attributed to simply ‘CKD.’ Thus the impor tance of ordering
the test for specific indications for those with CKD should be
emphasized.
Areas of Controversy, Confusion, or Non-consensus
Information about cardiac troponins and their relationship
with comorbidity is sparse in people with CKD who are not
receiving dialysis treatment. Although elevated values for
TnT are common in people with low GFR in the absence of
ACS or CHF, the prevelance of elevated TnI in the absence of
ACS or CHF in people with low GFR may be only 0.4-6%
depending on the cutoff value chosen.
532
Clarification of Issues and Key Points
As pointe d out by Roberts et al.,
553
although management
strategies in people without CKD based on these biomarkers
are quite clear, in people with CKD it is not the case despite
the strong data on the prognostic implications. These
biochemical markers can only be useful if they are interpreted
in the context of the clinical history and examination with
consideration given as to what management is appropriate
for each individual , and if a cardiovascular therapy that is
effective in this population is identified or developed.
553
The
importance of understanding and contextualizing elevations
98 Kidney International Supplements (2013) 3, 91–111
chapter 4

in troponin values in CKD populations cannot be overstated:
under-tr eatment in symptomatic individuals should be avoided,
and better understanding of treatment strategies in those
asymptomatic individuals is the focus of ongoing study .
Pediatric Considerations
While normative values exist for troponin in children,
554
its
utility as a general screening marker for myocardial ischemia
in pediatrics has recently been called into question,
555
and no
data exist relating reduced renal function and troponin levels
in children.
Non-invasive testing
Most studies of cardiac evaluation in people with CKD have
involved candidates for kidney transplantation in order to
identify existing cardiac conditions amenable to risk
modification and to exclude people with short expected
survival due to cardiac morbidity. Among the tests for
cardiovascular assessment, coronary angiography is consid-
ered invasive, expensive, and associated with risks such as
contrast-induced nephropathy and cholesterol embolism.
When trying to detect CAD or predict future cardiovascular
events in people with CKD, nuclear imaging modalities using
thallium tracers have shown conflicting results, especially in
the context of small vessel disease. Clinicians should be aware
of the limitations of radionuclear testing as a screening tool for
significant CAD given the limitations of the test. Exercise-
dependent techniques are limited by poor exercise tolerance of
many subjects and people with CKD frequently have cardiac
evaluation performed with pharmacological agents.
556,557
4.2.3: We recommend that people with CKD presenting with
chest pain should be investigated for underlying
cardiac disease and other disorders acc ording to the
same local practice for people without CKD (and sub-
sequent treatment should be initiated similarly). (1B)
4.2.4: We suggest that clinicians are familiar with the
limitations of non-invasive cardiac tests (e.g., exercise
electrocardiograph y [ECG], nuclear imaging, echo-
cardiography, etc.) in adults with CKD and interpret
the results accordingly . (2B)
RATIONALE
These statements are worded in this way firstly because
although people with CKD are at increased risk from
cardiovascular events, observational study has shown that
they are frequently discriminated against in terms of
investigation and management of such events. Secondly,
non-invasive cardiac tests have known additional limitations
in people with CKD and such limitations and their
consequences need to be clearly understood by health
professionals caring for people with CKD.
Evidence Base
Noninvasive stress testing appears to be the most common
first approach to cardiac evaluation of asymptomatic
patients, prompted by diabetes, age and risk-factor burden.
These tests include myocardial perfusion studies, stress
echocardiography, and most recently, cardiac computed
tomographic angiography. However, people with CKD are
underrepresented in studies evaluating the diagnostic sensi-
tivities and specificities of noninvasive tests. Exercise ECG is
limited by lack of specificity of the ST-segment response and
by inability of many people with CKD to exercise to a
diagnostic workload.
These tests have imperfect sensitivity and specificity in
people with CKD or, in the case of tomographic angiography,
evaluation in this population is as yet unpublished.
556
CAD
has been detected in a high proportion of people
on long–term dialysis therapy. Studies describing asso-
ciations of angiographic coronary stenosis with subseque nt
clinical events in people with ESRD, including those under-
going transplant evaluations, have reached inconsistent
conclusions.
558–562
Abnormalities on myocardial perfusion studies correlate
well w ith the presence of CAD in the general population,
with mean weighted sensitivity of 88% and mean weighted
specificity of 74%.
563
The performance of myocardial
perfusion studies in identifying CAD in people w ith CKD
is more variable, with reported sensitivities and specificities
in people with ESRD ranging from 37% to 90% and 40% to
90%, respectively.
564–567
Nonetheless, results from myocardial
perfusion studies have prognostic value for cardiac events
and mortality.
568,569
In a meta-analysis of 12 studies
involving thallium-201 scintigraphy and dobutamine stress
echocardiography, people with ESRD with inducible ischemia
had approximately 6 times the risk of MI and 4 times the risk
of cardiac death as those without inducible defects.
570
Moreover, subjects with fixed defects also had nearly 5 times
the risk of cardiac death.
The prognostic value of myocardial perfusion studies has
been shown with other perfusion tracers. For example, in a
study of 126 people with ESRD who underwent technetium-
99m myocardial perfusion studies as part of their pre-
transplant assessment, the presence of a reversible defect was
associated with 3 times the risk of post-transplant cardiac
events (HR 3.1; 95% CI 1.1-18.2) and nearly twice the risk of
death (HR 1.92; 95% CI 1.1-4.4) compared with normal test
results.
571
De Lima et al.
572
prospectively studied 126 renal transplant
candidates clinically classified as moderate (age 450 years) or
high (diabetes, extra cardiac vascular disease, or known CAD)
coronary risk with myocardial perfusion studies, dobutamine
stress echocardiography, and coronary angiography. Significant
CAD, defined as greater than 70% stenosis in 1 or more major
epicardial artery on angiography was found in 42% of the
sample. After a median follow-up of 46 months, clinical risk
stratification and coronary angiography predicted major
cardiac events, but myocardial perfusion studies and dobuta-
mine stress echocardiography did not.
Newer imaging techniques are under current evaluation
but a review here is beyond the scope of this guideline; these
Kidney International Supplements (2013) 3, 91–111 99
chapter 4

techniques include: stress myocardial perfusion single
photon emission computed tomography (SPECT);
557,573–576
18
F-BMS, a novel fluorine-based myocardial per-
fusion tracer;
557,577
and gated imaging using radio-labelled
ammonia (
13
NH
3
).
International Relevance
People with CKD who are being investigated prior to
transplantation should be assessed according to the national
and international cardiology guidelines and taking into
account local practice and availability of resource.
Implications for Clinical Practice and Public Policy
There is still a need for a safe, noninvasive, diagnostic
modality that will allow stratification of cardiovascular risk
among people with CKD. With the development of new
imaging markers, it becomes difficult for clinicians to choose
the imaging tests that can best aid clinical decisions for a
given patient. Imaging methods should provide accuracy and
precision at acceptable cost. Until advanced imaging
techniques (e.g., cardiac MRI, positron emission tomogra-
phy, cardiac computed tomography) are standardized,
clinicians should rely on the most reliable and familiar
techniques in their environment.
578
Areas of Controversy, Confusion, or Non-consensus
Clinical and cost-effectiveness could be improved; cardiac
evaluation in people w ith CKD should be based on data from
clinical trials.
Clarification of Issues and Key Points
If cardiac evaluation is at present predominantly used for
risk stratification and prognosis, in the future it may guide
optimal therapy and monitor clinical progress.
557
4.3 CKD AND PERIPHERAL ARTERIAL DISEASE
There is a strong link between CKD and peripheral arterial disease
(PAD). Symptoms of PAD may only be present in a minority
of people who have clinical evidence of P AD.
579
It is there-
fore important to measure ankle-brachial index and perform
regular systematic assessment of the lower limbs of people at
high risk of PAD to identify bruits, loss of pulses, cool pale
extremities, delay in venous filling, and skin ulceration.
580
4.3.1: We recommend that adults with CKD be regularly
examined for signs of peripheral arterial disease and
be considered for usual approaches to therapy. (1B)
4.3.2: We suggest that adults with CKD and diabetes are
offered regular podiatric assessment. (2A)
RATIONALE
These statements are worded in this way in order to highlight
the increased risk of PAD in people with CKD, particularly
those with CKD and diabetes.
Evidence Base
The prevalence of PAD is common in people with CKD and
it increases with lower levels of GFR (Table 31). PAD can be
Table 31 | Peripheral arterial disease and CKD
Study Population PAD definition Outcome of interest
O’Hare et al.
585
NHANES age 40+ ABI o0.9 24% prevalence in people with CKD and a CrCl of
o60 ml/min (o 1 ml/s) versus 3.7% in those with
normal kidney function
O’Hare et al.
587
HERS study, postmenopausal women
with known CHD
PAD event rates (amputation,
revascularization, or lumbar
sympathectomy)
Incident PAD event rates were 0.55%, 0.92%,
and 2.73% per year with CrCl 60, 30-59, and
30 ml/min/1.73 m
2
, respectively
O’Hare et al.
586
Cardiovascular Health Study, adults
age 65+
Lower-extremity PAD procedure
(bypass surgery, angioplasty, or
amputation)
HR for PAD procedure 2.5 (95% CI 1.2-5.1) for
highest quintile of cystatin C (Z1.28 mg/l) versus
lowest (r0.9 mg/l)
De Vinuesa et al.
582
102 adults in a CKD clinic, mean age
70
±
11 years, GFR 15-60 ml/min/
1.73 m
2
ABI o0.9 17% signs and symptoms of PAD, which had
passed unnoticed; 32% had ABI o0.9 (mean
0.64
±
0.25)
Liew et al.
584
6-year follow up of 1027 subjects (ABI
index recording and GFR measured
within 90 d)
ABI o0.9 6-year mortality rate for CKD and PAD 45% versus
28% CKD alone, 26% PAD alone, and 18% for
neither condition
Wattanakit et al.
588
6760 subjects, aged 45–84 years in the
Multi-Ethnic Study of Atherosclerosis
ABI o0.9 Albuminuria was associated with PAD in subjects
with diabetes (odds ratio 1.90, 95% CI 1.19–3.04)
but not in those without
Lash et al.
583
3612 subjects age 58.2
±
11.0 years
in the Chronic Renal Insufficiency
Cohort study
ABI o0.9 Overall, 16% prevalence of PAD in subjects with
GFRo60 ml/min/1.73 m
2
, increasing from 4% in
those with GFR460 to 22% in those with
GFRo30 ml/min/1.73 m
2
Bello et al.
581
920,985 subjects with GFR and
proteinuria assessment, median
follow-up 35 months (IQR 22-44)
Time to first hospitalization with
PAD
1891 of subjects (0.2%) hospitalized at least once
for PVD, adjusted rates increased with lower GFR
Abbreviations: ABI, ankle-brachial index; CHD, coronary heart disease; CI, confidence interval; CKD, chronic kidney disease; CrCl, creatinine clearance; d, day; GFR, glomerular
filtration rate; HERS, Heart and Estrogen/Progestin Replacement Study; HR, hazard ratio; IQR, interquartile range; NHANES, National Health and Nutrition Examination Survey;
PAD, peripheral arterial disease; PVD, peripheral vascular disease.
100 Kidney International Supplements (2013) 3, 91–111
chapter 4

attributed to the greater prevalence of traditional risk factors
such as diabetes, hypertension, dyslipidemia, advanced age,
and renal-specific factors.
581–588
Diabetes is the leading cause of CKD globally and the risk
of diabetic foot ulceration and lower extremity amputation in
subjects with diabetes increases dramatically with lower GFR.
In a cohort of 90,617 individuals with diabetes over a median
observation time of 2.4 years, the HR for amputation
increased from 2.08 (95% CI 1.68–2.58) for those with GFR
30-59 ml/min/1.73 m
2
to 7.71 (95% CI 5.29–11.26) for those
with GFR o30 ml/min/1.73 m
2
compared to reference
subjects with GFR 460 ml/min/1.73 m
2
.
589
Screening for PAD is recommended for adults in the
general population based on age and number of risk
factors.
590
Although an ankle-bra chial index of o0.9 is
generally considered evidence of PAD, the utility of this
measure is unproven in CKD because of the greater
prevalence of vessel calcifica tion. Evidence-based medical
therapies for PAD in people with CKD are similarly lacking.
Smoking cessation is mandatory. Aspirin may be beneficial
for prevention of cardiovascular events. Clopidogrel has not
been studied in people with CKD and PAD. No RCTs
evaluating percutaneous versus surgical revascularization
techniques have been conducted in people with CKD and
PAD but outcome studies all suggest that CKD confers an
increased risk of adverse outcome, regardless of the
revascularization technique employed.
591,592
These state-
ments do not apply to pediatric practice.
International Relevance
There is no evidence to suggest that the international
approach to identification and management of PAD in
people with CKD should differ.
Implications for Clinical Practice and Public Policy
People with CKD are more at risk of PAD and need regular
assessment and monitoring. The role of ankle-brachial index
versus other diagnostic techniques may have implications for
future practice. Prospective data on non-surgical therapies and
data regarding percutaneous versus surgical revascularization
are required to inform policy and recommended procedure.
Areas of Controversy, Confusion, or Non-consensus
There remains ongoing debate as to the timing and use of
specific diagnostic criteria for identification of PAD in people
with CKD and no studies have examined the utility of
conventional methods in early detection of PAD in CKD. The
use of specific medical and surgical management strategies in
people with CKD and PAD has not been evaluated. There is no
reason to believe that treatment strategies should differ, though
risks of diagnostic testing (such as angiography) remain real.
4.4 MEDICATION MANAGEMENT AND PATIENT SAFETY IN CKD
Many drugs and investigative pharmaceuticals are renally
excreted and their dosage may need to be reduced in patients
with CKD in order to avoid toxicity. As many people with
CKD are elderly, there should also be consideration of
chronological age.
It is beyond the scope of this guideline to list all the
agents that may need dose adjustment in CKD or which
should be avoided due to potential nephrotoxicity. This
information is widely available in documents which may exist
at local, regional, or national bodies (e.g., British National
Formulary: www.bnf.org), and other textbo oks of pharma-
cology.
593,594
However key classes of commonly prescribed
drugs in people with CKD will be mentioned together with
suggestions for dose adjustment. Much of this guidance is
based upon our understanding of pharmacology and
pharmacokinetics rather than randomized control trial
evidence.
The statements presented here are intended to inform
clinicians caring for those with CKD or at risk for CKD with
respect to common clinical situations or exposures which
may put people at risk for AKI or progression of CKD.
4.4.1: We recommend that prescriber s should take GFR
into account when drug dosing. (1A)
4.4.2: Where precision is required for dosing (due to
narrow therapeutic or toxic range) and/or estimates
may be unreliable (e.g., due to low muscle mass),
we recommend methods based upon cystatin C or
direct measurement of GFR. (1C)
4.4.3: We recommend temporary discontinuation of
potentially nephrotoxic and renally excreted drugs
in people with a GFR o60 ml/min/1.73 m
2
(GFR
categories G3a-G5) who have serious intercurrent
illness that increases the r isk of AKI. These agents
include, but are not limited to: RAAS blockers
(including ACE-Is, ARBs, aldosterone inhibitors,
direct renin inhibitors), diuretics, NSAIDs, metfor-
min, lithium, and digoxin. (1C)
4.4.4: We recommend that adults with CKD seek medical
or pharmacist advice before using over-the-counter
medicines or nutritional protein supplements. (1B)
4.4.5: We recommend not using herbal remedies in
people with CKD. (1B)
4.4.6: We recommend that metformin be continued in
people with GFRZ45 ml/min/1.73 m
2
(GFR cate-
gories G1-G3a); its use should be reviewed in those
with GFR 30–44 ml/min/1.73 m
2
(GFR category G3b);
anditshouldbediscontinuedinpeoplewithGFR
o30 ml/min/1.73 m
2
(GFR categories G4-G5). (1C)
4.4.7: We recommend that all people taki ng potentially
nephrotoxic agents such as lithium and calcineurin
inhibitors should have their GFR, electrolytes and
drug levels regularly monitored. (1A)
4.4.8: People with CKD should not be denied therapies
for other conditions such as cancer but there
should be appropriate dose adjustment of cytotoxic
drugs according to knowledge of GFR. (Not
Graded)
Kidney International Supplements (2013) 3, 91–111 101
chapter 4

RATIONALE
These statements are worded in this way in order to inform
appropriate prescribing and management of medical condi-
tions in people with CKD. Recommendation 4.4.1 is
specifically worded to ensure that clinicians remember that
in those circumstances w here accurate GFR is required
(narrow therapeutic or toxic w indow), a direct measurement,
not an estimate of GFR should be undertaken.
Evidence Base
Potential problems associated with use of medication in
people with CKD include:
a) Reduced ability to excrete drugs and/or their metabolites
b) Increased sensitivity to medications (e.g., those bound to
albumin in hypoalbuminemic states such as nephrotic
syndrome)
c) Diminished tolerance of side effects, particularly in the
elderly
d) Loss of efficacy
13
Wherever possible, people with CKD should receive the
same treatment as those with normal renal function. However,
the dosages may need adjustment according to GFR.
There are medicines whose toxicity is worsened in acute
illness particularly in a setting of dehydration such as
diarrhea and vomiting. General advice about appropriate
dosing and when to restart these agents should be given to
people taking these drugs during intercurrent illness, together
with a recommendation for consultation with a health-care
professional as soon as possible.
Use of herbal and over-the-counter medicines is very
common worldwide and some (such as those containing
aristolochic acid)
595,596
are known to be nephrotoxic. There is
no good qualit y safety or efficacy data for many of these
compounds.
Table 32 details specific advice for various classes of drugs
where CKD may be an issue. These include RAAS blocking
agents,
262
beta-blockers, analgesics, antibiotics, lithium,
597
hypoglycemic agents,
598–600
lipid-lowering agents,
449,601–604
certain chemotherapeutic agents, and anticoagulants.
605
International Relevance
This guidance is based upon knowledge of pharmacology that
has universal relevance. The main international implication is
centered on costs of so me newer therapies compared to the
older ones. Somewhat paradoxically, as the weight of
evidence resides mainly with agents that have been available
for longer, they have the advantage of being less costly, and
have the side effects that are well documented; thus many of
these older agents are preferred.
Pediatric Considerations
No specific recommendation for or against the use of a BSA
adjusted eGFR in relation to drug dosing can be made in
children with CKD as the majority of drug studies have been
performed in adult males and extrapolated to children
without consideration for BSA and/or pediatric renal
function or clearance.
The exception to this rule would be that for any drug
where pediatric pharmacokinetic or pharmacodynamic
studies exist, the method used in calculating the effect of
renal function should be used when estimating the need for
dose adjustments or modifications for the individual patient.
Numerous resources for dose adjustments for pediatric
CKD and ESRD patients exist, including those found in
numerous textbooks
606,607
and online resources.
608
An additional value of dedicated pediatric renal multi-
disciplinary teams, in terms of caring for children with CKD
and ESRD, is their ability to provide anticipatory guidance
for the patients and families. All such teams should at the
least provide w ritten or online information to their patients
and families directing them to seek advice in situations where
they may be prescribed medications from other providers or
may be seeking over-the-counter drugs or supplements. The
presence of trained pediatric pharmacists as part of such
teams can be invaluable in achieving such support and
education for both families as well as the CKD health-care
providers and community resources such as local
pharmacists.
LIMITATIONS
People with CKD are often excluded or not identified from
trials of medications for non-renal disease. Recommenda-
tions are partly based upon knowledge of pharmacology
rather than controlled trials in carefully defined populations.
RESEARCH RECOMMENDATIONS
National and international research groups, and those
with CKD-focus organizations (International Society of
Nephrology, International Federation of Kidney Foundations,
and other national bodies) should ensure adequate repre-
sentation of people with CKD in clinical trials, leading to
an improved understanding of pharmacodynamics of those
with CKD.
4.5: IMAGING STUDIES
4.5.1: Balance the risk of acute impairment in kidney
function due to contrast agent use against the
diagnostic value and therapeutic implications of
the investigation. (Not Graded)
RATIONALE
Use of iodinated radiocontrast media has been associated
with AKI with reported rates of 0-11% depending on the
population under study, the type of agent that is used, and
the definition of nephrotoxicity.
609
The following recom-
mendations are concordant with those from the American
College of Radiology (ACR),
610
the European Societ y of
Urogenital Radiology (ESUR)
611
and the KDIGO Clinical
Practice Guideine for Acute Kidney Injury.
7
102 Kidney International Supplements (2013) 3, 91–111
chapter 4

Table 32 | Cautionary notes for prescribing in people with CKD
Agents Cautionary notes
1. Antihypertensives/cardiac medications
RAAS antagonists (ACE-Is,
ARBs, aldosterone antagonists,
direct renin inhibitors)
K Avoid in people with suspected functional renal artery stenosis
K Start at lower dose in people with GFR o45 ml/min/1.73 m
2
K Assess GFR and measure serum potassium within 1 week of starting or following any dose escalation
K Temporarily suspend during intercurrent illness, planned IV radiocontrast administration,
bowel preparation prior to colonoscopy, or prior to major surgery
K Do not routinely discontinue in people with GFR o30 ml/min/1.73 m
2
as they remain nephroprotective
Beta-blockers K Reduce dose by 50% in people with GFR o30 ml/min/1.73 m
2
Digoxin K Reduce dose based on plasma concentrations
2. Analgesics
NSAIDS
K Avoid in people with GFR o30 ml/min/1.73 m
2
K Prolonged therapy is not recommended in people with GFR o60 ml/min/1.73 m
2
K Should not be used in people taking lithium
K Avoid in people taking RAAS blocking agents
Opioids
K Reduce dose when GFR o60 ml/min/1.73 m
2
K Use with caution in people with GFR o15 ml/min/1.73 m
2
3. Antimicrobials
Penicillin K Risk of crystalluria when GFR o15 ml/min/1.73 m
2
with high doses
K Neurotoxicity with benzylpenicillin when GFR o15 ml/min/1.73 m
2
with high doses (maximum 6 g/day)
Aminoglycosides
K Reduce dose and/or increase dosage interval when GFR o60 ml/min/1.73 m
2
K Monitor serum levels (trough and peak)
K Avoid concomitant ototoxic agents such as furosemide
Macrolides K Reduce dose by 50% when GFR o30 ml/min/1.73 m
2
Fluoroquinolones K Reduce dose by 50% when GFR o15 ml/min/1.73 m
2
Tetracyclines K Reduce dose when GFR o45 ml/min/1.73 m
2
; can exacerbate uremia
Antifungals
K Avoid amphotericin unless no alternative when GFR o60 ml/min/1.73 m
2
K Reduce maintenance dose of fluconazole by 50% when GFR o45 ml/min/1.73 m
2
K Reduce dose of flucytosine when GFR o60 ml/min/1.73 m
2
4. Hypoglycemics
Sulfonylureas
K Avoid agents that are mainly renally excreted (e.g., glyburide/ glibenclamide)
K Other agents that are mainly metabolized in the liver may need reduced dose when
GFR o30 ml/min/1.73 m
2
(e.g., gliclazide, gliquidone)
Insulin
K Partly renally excreted and may need reduced dose when GFR o30 ml/min/1.73 m
2
Metformin K Suggest avoid when GFR o30 ml/min/1.73 m
2
, but consider risk-benefit if GFR is stable
K Review use when GFR o45 ml/min/1.73 m
2
K Probably safe when GFR X45 ml/min/1.73 m
2
K Suspend in people who become acutely unwell
5. Lipid-lowering
Statins
K No increase in toxicity for simvastatin dosed at 20 mg per day or simvastatin 20 mg /ezetimide
10 mg combinations per day in people with GFR o30 ml/min/1.73 m
2
or on dialysis
449
K Other trials of statins in people with GFR o15 ml/min/1.73 m
2
or on dialysis also showed no excess toxicity
Fenofibrate K Increases SCr by approximately 0.13 mg/dl (12 mmol/l)
6. Chemotherapeutic
Cisplatin
K Reduce dose when GFR o60 ml/min/1.73 m
2
K Avoid when GFR o30 ml/min/1.73 m
2
Melphalan K Reduce dose when GFR o60 ml/min/1.73 m
2
Methotrexate K Reduce dose when GFR o60 ml/min/1.73 m
2
K Avoid if possible when GFR o15 ml/min/1.73 m
2
7. Anticoagulants
Low-molecular-weight heparins
K Halve the dose when GFR o30 ml/min/1.73 m
2
K Consider switch to conventional heparin or alternatively monitor plasma anti-factor Xa in those at high risk
for bleeding
Warfarin
K Increased risk of bleeding when GFR o30 ml/min/1.73 m
2
K Use lower doses and monitor closely when GFR o30 ml/min/1.73 m
2
8. Miscellaneous
Lithium
K Nephrotoxic and may cause renal tubular dysfunction with prolonged use even at therapeutic levels
K Monitor GFR, electrolytes, and lithium levels 6 monthly or more frequently if the dose changes
or the patient is acutely unwell
K Avoid using concomitant NSAIDs
K Maintain hydration during intercurrent illness
K Risk-benefit of drug in specific situation must be weighed
Abbreviations: ACE-I, angiotensin-converting enzyme inhibitor; ARB, angiotensin-receptor blocker; CKD, chronic kidney disease; GFR, glomerular filtration rate; NSAIDs, non-
steroidal anti-inflammatory drugs; RAAS, renin-angiotensin-aldosterone system; SCr, serum creatinine.
Kidney International Supplements (2013) 3, 91–111
103
chapter 4

Radiocontrast
4.5.2: We recommend that all people with GFR o60 ml/
min/1.73m
2
(GFR categories G3a-G5) undergoing
elective investigation involving the intravascular
administration of iodinated radiocontrast media
should be managed according to the KDIGO
Clinical Practice Guideline for AKI including:
K Avoidance of high osmolar agents (1B);
K Use of lowest possible radiocontrast dose (Not
Graded);
K Withdrawal of potentially nephrotoxic agents
before and after the procedure (1C);
K Adequate hydration with saline before, during,
and after the procedure (1A);
K Measurement of GFR 48–96 hours after the
procedure (1C).
RATIONALE
These statements have been worded in order to inform safe
radiological investigation of people with CKD and to avoid
potential nep hrotoxcity that can be associated with radi-
ological imaging .
Evidence Base
Radiocontrast media associated AKI is a largely preventable
cause of morbidity and mortality. There is no internationally
agreed definition but most studies use an increase in SCr of
40.5 mg/dl (44 mmol/l) and/or a 25% increase from baseline
SCr within 3 days of the procedure.
7,609–611
Epidemiological
studies and case series have identified the following risk
factors for AKI:
a) GFR o60 ml/min/1.73 m
2
(particularly if o30 ml/min/
1.73 m
2
)
b) Diabetes
c) Concurrent dehydration
d) CHF
e) 470 years of age
f) Concurrent use of known nephrotoxic agents such as
NSAIDs
g) Use of high osmolality agents (especially in those with
GFR o60 ml/min/1.73 m
2
)
h) Use of large doses of radiocontrast media
i) Intra-arterial injection
j) Gout (hyperuricemia)
Numerous studies of preventative strategies have been
performed with the following conclusions:
a) High osmolar agents pose a greater risk of AKI in people
with CKD.
609
b) Iso-osmolar agents compared to low-osmola r agents are
associated with lower rates of AKI in some but not all
studies. Wherever possible iso-osmolar agents should be
used in people with CKD at high risk for AKI (although
these tend to be more expensive).
612
c) Although risk for AKI increases with GFR o60 ml/min/
1.73 m
2
, the rates are particularly high (7.8% in one
study) when GFR is o30 ml/min/1.73 m
2
. Implementing
preventative strategies for all with a GFR o60 ml/min/
1.73 m
2
may not be practical but a graded risk assess-
ment taking into account all factors may be more
realistic. Some guidelines such as American College
Radiology
610
and ESUR
611
provide a checklist as a
means of identifying patients at risk of AKI befo re
investigation.
d) Extracellular volume expansion is w idely recommended
although there are few good quality trials on which to
base an ideal protocol.
609
0.9% saline given by contin-
uous infusion appears superior to 0.45% saline or bolus
injection and there is no demonstration of consistent
superiority of sodium bicarbonate over saline. Current
guidance suggests either infusion of 1 ml/kg body weight/
hour for 3-12 hours before and after the procedure or
100 ml/hr, beginning 6 to 12 hours before and continuing
4 to 12 hours after intravascular iodinated contrast
medium administration.
609,610
e) Use of N-acetylcysteine or ascorbic acid as preventative
measures has not been shown to be a consistent benefit.
International Relevance
This guidance has universal relevance although there are cost
implications as the iso-osmolar contrast media are more
expensive.
LIMITATIONS
Some of the guidance is based upon limited evidence and
there is a need for more research into simple preventative
measure such as pre-investigation rehydration (see below).
There has not previously been a universal definition for AKI
following administration of contrast media. However,
recommendations from the KDIGO AKI Guideline suggest
that the same general AKI definition and staging be used for
changes in kidney function, irrespective of etiology.
RESEARCH RECOMMENDATIONS
Prospective studies using direct measures of GFR before and
after administration of radiological contrast media are
required to help define the incidence of AKI. Such studies
would also be able to validate creatinine or other estimates of
GFR in people undergoing radiological investigation.
Prospective controlled trials of rehydration using different
fluids (saline, bicarbonate, Hartmann’s) and validated
estimates of GFR are urgently required.
Definitive studies of N-acetylcysteine and other anti-
oxidants would help determine their usefulness or otherwise.
Gadolinium-containing contrast media
Gadolinium is a rare earth element that is naturally highly
toxic. When bound to proprietary chelating agents, it is
essentially biologically inert in people with normal renal
function and provides excellent contrast during MRI. These
104 Kidney International Supplements (2013) 3, 91–111
chapter 4

chelates are excreted unchanged by the kidneys by glomerular
filtration and have much lower direct nephrotoxicity than
conventional radioiod ine contrast media.
613
However over
200 cases of a scleroderma-like condition now termed
nephrogenic systemic fibrosis (NSF) following gadolinium
use in patients with CKD have been reported.
613, 614
4.5.3: We recommend not using gadolinium-containing
contrast media in people with GFR o15 ml/min/
1.73 m
2
(GFR category G5) unless there is no
alternative appropriate test. (1B)
4.5.4: We suggest that people with a GFR o30 ml/min/
1.73 m
2
(GFR categories G4-G5) who require
gadolinium-containing contrast media are preferen-
tially offered a macrocyclic chelate preparation. (2B)
RATIONALE
These statements have been worded in order to enable safe
administration of gadolinium to people with CKD. As with
all tests requested in the CKD population, clinicians should
be aware of the risk-benefit ratio of the use of gadolinium in
people with GFR 15-29 ml/min/1.73 m
2
.
Evidence Base
NSF is an untreatable and sometimes fatal condition that
complicates the use of gadolinium-containing contrast media
in people with CKD. Prevention is therefore the best
approach with avoidance of gadolinium exposure unless
clinically indicated and to use the lowest risk agent at the
lowest dose.
613,615
Although the risk of AKI w ith iodinated contrast media is
much greater than that for NSF with gadolinium, the former
is treatable with dialysis whereas the latter is not. Thus,
consideration of more conventional imaging techniques
should be undertaken in all with a GFR o30 ml/min/
1.73 m
2
.
610,613–615
A recent meta-analysis and review have highlighted those
patients most at risk and quantified an OR for NSF of
between 20-50 for those with a GFR o15 ml/min/1.73 m
2
.
614
While there is general agreement that patients with a GFR
415 ml/min/1.73 m
2
are at increased risk, it is not possible
to derive a precise estimate. No patients with a GFR 430 ml/
min/1.73 m
2
have developed NSF without concomitant
liver failure.
614
There is some suggestion that the type of gadolinium
preparation plays a role:
614,615
Linear chelated preparations
such as gadodiamide may be more likely to cause NSF and
should be avoided when GFR is o30 ml/min/1.73 m
2
.
Gadoteridol, gadobutrol, or gadoterate should be considered
if MRI with contrast is clinically essential.
610,613,615
Moreover,
as gadolinium is freely dialysed,
616
most guidelines recom-
mend dialysis in patients with a GFR o15 ml/min/1.73 m
2
or for those already on dialysis immediately after (and
perhaps repeated 24 hours later) completion of the
procedure.
610,615
The role of dialysis in people with GFR
above 15 ml/min/1.73 m
2
is uncertain.
International Relevance
This guidance has universal relevance although there are cost
implications as the non-linear chelated preparations are more
expensive.
Pediatric Considerations
Regar ding both Recommendations 4.5.3 and 4.5.4, a number
of considerations specific to the use of gadolinium preparations
in young children and neonates must also be considered in
addition to the general admonishments against their use in
situations of GFR o30 ml/min/1.73 m
2
.Inparticular,theFDA
currently does not license any gadolinium-based contrast agent
(GBCA) product for use in children less than 2 years of age; and
likewise the European Medicines Agency cautions against the
use of an y GBCA in a child less than 1 year of age.
In addition, the ability to accurately estimate the relative
value of a neonate or young infant’s GFR leads to great
difficulty in terms of assigning risk of GBCA exposure if one
bases this on renal clearance. In a recent paper by Meng and
colleagues,
617
they surveyed a worldwide group of both
cardiologists and radiologists with respect to their use of
gadolinium in their pediatric MRI practice. While 93% of the
respondents did evaluate renal function in some or all of
their patients, only a slight majority (54%) used an
estimating formula, most often the Schwartz equation, with
the remainder relying on SCr alone (31%) or urine output
(6%). Perhaps of most concern was that renal function was
assessed in only 33% and 31%, respectively, of patients
classified as ‘all neonates’ or ‘all patients o1 week of age.’
Equally concerning was the fact that 13% of the respondents
would give gadolinium to so me of these children in the face
of a GFR o30 ml/min/1.73 m
2
.
In recognition of our inability to accurately measure GFR
in the neonate and, by extension, the clearance of compounds
such as gadolinium, all nephrologists and radiologists must
exercise caution in terms of use of GBCA in this potentially
high-risk population, and all other imaging modalities
should be considered prior to choosing one requiring
gadolinium exposure.
LIMITATIONS
The evidence is largely based upon case series ra ther than
prospective studies or RCTs.
RESEARCH RECOMMENDATIONS
As for iodinated contrast media, a prospective study of people
with CKD undergoing nuclear MRI with gadolinium contrast
would help define change in GFR and validate estimators.
Because NSF is such a serious condition, an RCT of dialysis in
people with GFR o30 ml/min/1.73 m
2
would help to
determine risk-benefit in these patients, though recruitment
may be difficult due to potential ethical conc erns.
Bowel preparation
The increasing use of colonoscopy as a screening tool for
bowel cancer has resulted in many people undergoing bowel
Kidney International Supplements (2013) 3, 91–111 105
chapter 4

preparation with oral sodium phosphate-containing prepara-
tions. Case reports of acute and late irreversible renal failure
with biopsy-proven phosp hate deposition have led to a new
disease entity termed acute phosphate nephropathy.
618,619
Less than 40 definite cases have been reported and these have
been extensively reviewed.
4.5.5: We recommend not to use oral phosphate-contain-
ing bowel preparations in people with a GFR o 60
ml/min/1.73 m
2
(GFR categories G3a-G5) or in
those known to be at risk of phosphate nephropathy.
(1A)
RATIONALE
This statement has been worded in order to enable safe bowel
preparation in people with CKD who need to undergo
investigation of bowel disease.
Evidence Base
Electrolyte disturbances that are sometimes severe and
include hyperphosphatemia, hypocalcemia, hypo- and
hypernatremia, and hypokalemia have been reported in
normal volunteers undergoing oral phosphate bowel prepa-
ration.
620
Renal inju ry has been reported in a small number
of people altho ugh the condition is likely to be under-
recorded. A recent study from Iceland estimated the
incidence to be around 1 per thousand doses
621
but others
would suggest incidence rates of between 1% and 4%.
618,619
Two broad patterns of renal inju ry have been des-
cribed. An early symptomatic response associated with severe
hyperphosphatemia and hypocalcemia and a later (days to
months) irreversible kidney injury associated with a specific
tubulointerstitial calcium phosphate deposition.
618,619
The following people are said to be at particular risk
although the link to kidney injury is associative in many cases
and firm evidence is lackin g:
a) GFR o60 ml/min/1.73 m
2
b) 460 years of age
c) Female
d) Hypertension
e) Diabetes
f) CHF
g) Dehydration
h) Active colitis
i) Concurrent use of RAAS blocking agents, diuretics,
lithium, NSAIDs
j) Large and/or repeat dosing of oral phosphate prepara-
tions
k) Hypoparathyroidism
Although the FDA has banned oral phosphate solutions,
there is no qualitative difference with tablet preparations.
Both should be avoided in people at risk and this is the
current recommendation of the American Society for
Gastrointestinal Endoscopy.
622
There is some debate as to whether the cause of the kidney
injur y is entirely due to dehydration rather than phosphate
use per se.
623
However, it is hard to unde rstand why calcium
phosphate deposits occur and cause late renal impairment if
dehydration was the only factor.
There is a single RCT of phosphate versus non- phosphate
preparations but assessment of renal function was limited to
change in SCr and GFR at baseline is not reported.
624
Notwithstanding these limitations, there were greater changes
in serum potassium, calcium, and phosphate in those given
sodium phosphate-containing preparations.
As there are non-phosphate-containing bowel prepara-
tions available, these should be used in all the above groups
(and arguably in all people given the biochemical abnorm-
alities observed in normal volunteers). As for radiocontrast
media, rehydration with saline may be required in the frail
and ill irrespective of the bowel preparation that is used.
International Relevance
This guidance has universal relevance but non-phosphate-
containing bowel preparations are more expensive, so the use
of these agents may differ around the world.
LIMITATIONS
The data are based upon case series with a limited number of
affected individuals and retrospective population studies.
RESEARCH RECOMMENDATIONS
Prospective study in people with normal renal function and
those with different severities of CKD are urgently required
in order to define the acute biochemical and metabolic effects
of phosphate-containing bowel preparations. There is also a
need for a study of all people undergoing bowel preparation
with whatever preparation in order to explore the effects on
GFR on the incidence of this complication. More definitive
exploration of rehydration therapy (type and volume) in
people with CKD undergoing bowel preparation is urgently
needed.
4.6 CKD AND RISKS FOR INFECTIONS, AKI,
HOSPITALIZATIONS, AND MORTALITY
The following section gives guidance for the care of people
with CKD given that they are at increased risk for a number
of events such as infections, AKI, CVD, hospitalizations, and
mortality. Appreciating that increased risk and implementing
some of the recommendations below may result in improved
outcomes for people. It will be important to develop policies
and robust research agendas to address areas which do not
have a substantial evidentiary base.
CKD and risk of infections
CKD is associated with significant major infectious compli-
cations, which occur at rates 3 to 4 times the general
population. Infection is an impor tant cause of morbidity and
mortality among patients with kidney failure and is the
106 Kidney International Supplements (2013) 3, 91–111
chapter 4

second leading cause of death following CVD. CKD may be a
risk-multiplier for acute infectious disease-associated mor-
tality, as it is for CVD. Despite a less effective response to
vaccination, there are data suggesting benefit from immuni-
zation among people with CKD as in the general population.
4.6.1: We recommend that all adults with CKD are
offered annual vaccination with influenza vaccine,
unless contraindicated. (1B)
4.6.2: We recommend that all adults with eGFR o30 ml/
min/1.73 m
2
(GFR categories G4-G5) and those at
high risk of pneumococcal infection (e.g., nephrotic
syndrome, diabetes, or those receiving immuno-
suppression) receive vaccination with polyvalent
pneumococcal vaccine unless contraindicated. (1B)
4.6.3: We recommend that all adults with CKD who have
received pneumococcal vaccination are offered
revaccination within 5 years. (1B)
4.6.4: We recommend that all adults who are at high risk
of progression of CKD and have GFR o30 ml/min/
1.73 m
2
(GFR categories G4-G5) be immunized
against hepatitis B and the response confirmed by
appropriate serological testing. (1B)
4.6.5: Consideration of live vaccine should include an
appreciation of the patient’s immune status and
should be in line with recommendations from
official or governmental bodies. (Not Graded)
4.6.6: Pediatric immunization schedules should be followed
according to official international and regional
recommedations for children with CKD . (Not Graded)
RATIONALE
CKD is associated with alterations in primary host defense
mechanisms and increases the risk of bacterial infections
(Table 33). Epidemiological study suggests that the 3 most
commonly seen infectious complications in the CKD
population are: urinary tract infection, pneumonia, and
sepsis. In the general population, there is strong evidence that
preventive measures are effective in adults and there are data
suggesting benefit from immunization in people with CKD.
Evidence Base
Previous investigations have firmly established ESRD as a
strong risk factor for infectious complications.
625–628
How-
ever, few epidemiologic reports have addressed the risk of
infections in people with CKD not treated with dialysis.
629,630
Data from the US Renal Data System (USRDS) suggest that
higher rates of hospital admission because of septicemia are
noted in people with CKD compared with those without
CKD.
631
Comprehensive studies of the absolute rates, risk
factors, clinical course, and outcomes of different ty pes of
clinically relevant infections across the spectrum of CKD are
not available.
Among people with CKD, very few studies have examined
the incidence or prevalence of infections and no published
data describe GFR category–specific infection rates. Among
Medicare beneficiaries aged more than 66 yr, people with
diagnosed CKD seem to have substantially higher rates of
hospitalization for diagnoses of pneumonia and sepsis
compared with people without diagnosed CKD.
628
In
addition to an increase d incidence of being hospitalized
with infections, people with CKD have longer lengths of
hospital stay during infection-related admissions compared
with people without CKD.
632
Among USRDS data admission rates for all causes, CVD
and infection, are 38–46% higher for those with CKD than
for those without.
633
Rates for pneumonia in CKD were
nearly three times higher than those for non-CKD.
Hospitalizations for bacteremia/septicemia, were nearly four
times higher for people with CKD compared to those
without. Hospitalization rates for urinary tract infections
were three-fold higher for people with CKD.
In people with ESRD, the function of polymorphonuclear
white blood cells, lymphocytes, and monocytes is altered,
resulting in an impaired host response to infection.
636–638
However, these issues have not been adequately investigated
in people with CKD.
There is a growing body of evidence in the general
population for reduction in infections, hospitalizations, and
mortality as a result of immuni zations. Current data suggest
that vaccination is an underused prevention strategy in the
CKD and ESRD populations.
639
Potential barriers to
immunization that are specific to the CKD populations have
not been systematically examined. Although lower vaccine
responsiveness has been widely recognized in the ESRD
population, to what extent moderate to advanced CKD
modifies vaccine responsiveness remains unclear.
640–642
Studies that have examined vaccination in the setting of
CKD or ESRD have been limited by small study size, variable
follow-up, and ascertainment of surrogates for vaccine
effectiveness such as antibody response and rate of antibody
decline after vaccination as opposed to vaccine efficacy for
preventing infection.
643
Influenza A and B vaccine. Despite potentially impaired
antibody responses, a 2-year analysis of US Medicare claims
data found that people vaccinated against influenza A and B
on dialysis had a substantially lower chance of any-cause
hospital admission and any-cause death than those patients
not vaccinated on dialysis.
644
This finding might indicate
clinical effectiveness of vaccinating this population but its
observational design mig ht also reflect differences in the
underlyin g clinical status among people vaccinated and not
Table 33 | Risk factors for infection in people with CKD
Advanced age
High burden of coexisting illnesses such as diabetes
Hypoalbuminemia
625
Immunosuppressive therapy
628
Nephrotic syndrome
634
Uremia
Anemia and malnutrition
635
High prevalence of functional disabilities
Abbreviation: CKD, chronic kidney disese
Kidney International Supplements (2013) 3, 91–111
107
chapter 4

vaccinated. No unique adverse events related to influenza
vaccine have been identified in people on dialysis.
Pneumococcal vaccine. People with kidney disease
vaccinated with the pneumococcal vaccine seem to
develop different serotype-specific titers, develop lower levels
of antibody titers, and have a more rapid loss of antibody
titers as compared with healthy control subjects.
639,645,646
Practitioners should be aware of the impact of
specific vaccines on responsiveness and duration of respon-
siveness. Revaccination practices will be dictated by that
knowledge.
Hepatitis B vaccine. Widespread hepatitis B virus (HBV)
vaccination at the onset of dialysis has led to a marked
reduction of HBV infections in people with ESRD,
although improved screening of blood products and
dissemination of recommendations for reducing the spread
of HBV infections in dialysis units have also likely
contributed.
647
Among people with moderate to advanced
CKD, hepatitis B vaccination responsiveness has been shown
to range from approximately 60 to 80% depending on the
dosage, number of administered vaccines, and study
population. Although findings have been inconsistent as to
whether the level of GFR affects vaccine responsiveness in
people with CKD
640,642
those with higher GFR are more
likely to respond with seroconversion, independent of other
factors.
640
S. aureus vaccine. StaphVAX has not been shown to be
efficacious in reducing the risk for Staphylococcus aureus
bacteremia in people on hemodialysis.
648,649
No data are
published in people with CKD.
Live vaccines. Due to the fact that people with CKD are
often immunocompromised, live vaccines should only be
used with caution on an individual basis.
In summ ary, although some vaccines (like influenza) in
usual doses provide protection, other vaccines (HBV and
pneumococcal) require more frequent dosing or larger doses
to achieve and maintain protective antibody titers. Frequency
and type of vaccination will vary according to local
circumstances and prevalence of disease.
International Relevance
The availability of different vaccinations may vary worldwide,
as does the prevalence of specific bacterial, viral, and other
infections. It is reasonable to offer individuals appropriate
immunization according to local practices.
Implications for Clinical Practice and Public Policy
Vaccines for influenza, hepatitis B, and pneumococcus are
currently recommended for people with CKD by the local,
regional, or national advisory committees on immunization
practices from most countries.
Current recommendations are to:
K Provide influenza vaccination annually to people with
CKD.
K Provide pneumococcal vaccine with a single booster dose
5 years after the initial dose.
K Provide HBV vaccine to people with CKD who are likely
to require RRT. Although the recommendation is to give
the HBV vaccine during more severe CKD (GFR o15 ml/
min/1.73 m
2
), it may be preferable to give this earlier to
maximize the chances of achieving immunity; there are
data to support this practice.
640
This would also ensure
that all patients are immunized against HBV before
receiving a transplant. As protective antibody levels may
fall, this should be checked (possibly annually) with
booster doses given if appropriate.
Areas of Controversy, Confusion, or Non-consensus
Much remains to be understood concerning impaired host
response to infection in patients with CKD.
K Studies should be undertaken to determine the absolute
rates, risk factors, and clinical course of different types of
clinically relevant infections across the spectrum of CKD,
by GFR and albuminuria category, and by cause.
K The outcomes across the range of acute infecti ons in
CKD population need to be ascertained.
K Studies should be undertaken to assess the rate of decline
of antibody titers post-vaccination and the efficacy of
immunization in people with CKD.
Pediatric Considerations
Current immunization schedules for children are regularly
updated by both the US Centers for Disease Control and
Prevention
650
and American Academy of Pediatrics.
651
Curr ent and comprehensive immunization recommendations
for children with CKD have been published by Neu in 2012.
652
The paper addresses key issues regarding the use of vaccines
in CKD pediatric populations who are receiving concomitant
immunosuppression and in those a waiting transplantation.
The need for , and interpretation of, protective antibody levels
for those vaccines where this is indicated is described.
An oversimplified summary of the recommendations
would be to provide all recommended childhood vaccines
to every child with CKD with the exception of any live viral
vaccine in a child receiving immunosuppressive medications.
Likewise children on dialysis should not receive the live
attenuated influenza vaccine although the inactivated version
can and should be given to all children with CKD on an
annual basis otherwise. Pneumococcal vaccination is par ti-
cularly important in children with nephrotic syndrome and
those with CKD, and current vaccination schedules and
products should be carefully reviewed to ensure proper
serotype coverage is being provided. Hepatitis B status and
vaccination are of extreme importance in all children who
may go onto dialysis – and specific recommendations for
ongoing monitoring and inter pretation of antibody levels
should be carefully reviewed.
CKD and risk of AKI
Due to the epidemiological association between CKD and
AKI and the number of observational studies reporting an
association between pre-existing CKD and AKI, CKD is
108 Kidney International Supplements (2013) 3, 91–111
chapter 4

considered the most consistent pre-existing condition
associated with a high risk of AKI. However the potential
linkage between patients with AKI, CKD, and ESRD has been
inadequately studied to date and remains ill defined. This
section describes AKI as a complication which needs to be
managed in those with CKD. Given its association with
progression, it is also described in that section.
4.6.7: We recommend that all people with CKD are
considered to be at increased risk of AKI. (1A)
4.6.7.1: In people with CKD, the recommendations
detailed in the KDIGO AKI Guideline should
be followed for management of those at risk
of AKI during intercurrent illness, or when
undergoing investigation and procedures that
are likely to increase the risk of AKI. (Not
Graded)
RATIONALE
Observational data suggest a strong association between pre-
existingCKDandAKI.TheappreciationthatCKDpatients
may be more susceptible to AKI is the purpose of the above set
of statements. However, methodological issues such as how
CKD and AKI are defined in clinical studies and the statistical
adjustments for non-uniformity of comorbidities among
various studies may affect the validity of observed associations.
Evidence Base
CKD is designated as a risk factor for AKI because of
the epidemiological association between the two.
263,264
A
number of studies in a variety of settings report an associ-
ation between pre-existing CKD and AKI.
265-271
CKD is a
potent predictor of acute decline in kidney function
following exposure to radiocontrast,
272
major surgery,
273
and other medical conditions.
274
Hsu et al.
14
compared the pre-hospitalization MDRD GFR
of 1764 adult members of the Kaiser Permanente Northern
California health-care system who developed dialysis-requir-
ing AKI during hospitalization with 600,820 individuals who
did not. Compared with a reference baseline GFR of Z60 ml/
min/1.73 m
2
, a baseline GFR of 45–59 ml/min/1.73 m
2
was
associated with an adjusted OR of in-hos pital AKI of 1.66
(95% CI 1.40–1.97). For GFR values of 15–29 ml/min/1.73 m
2
,
the adjusted OR for in-hospital AKI was 20.42 (95% CI
17.40–23.9 6). The presence of diabetes, hypertension, and
proteinuria increased the likelihood of developing in-hospital
AKI, with adjusted ORs of 1.99 (95% CI 1.78–2.23), 1.55
(95% CI 1.37–1.76) and 2.84 (95% CI 2.52–3.19), respectively.
The authors concluded that CKD is the main risk factor for
AKI during hospitalization. A contrasting approach by Singh
et al. defined AKI as dialysis-requiring acute renal failure.
275
Because the clinical decision to dialyze a patient is frequently
influenced by a higher overall SCr, presence of hemodialysis
access, or consideration of inevitable progression to ESRD,
this definition of AKI could bias toward capturing more AKI
cases in CKD patients. Moreover, in patients with advanced
CKD, the progression of CKD to ESRD may sometimes be
difficult to separate from acute-on-chronic renal failure. A
cohort study by Lafrance et al. followed a referred CKD
population in British Columbia for a median of 19.4 months
after achieving a GFR of r30 ml/min/1.73 m
2
. Forty-five
percent had at least one episode of AKI.
276
In another cohort
study of 920,985 adults in Alberta, Canada with at least one
outpatient measurement of SCr and proteinuria and not
requiring chronic dialysis, risk of admission with AKI
increased with heavier proteinuria and reduced GFR.
16
International Relevance
These guidelines about AKI have relevance around the world.
While the causes of AKI may differ by region, country, socio-
economic status, and age, the consequences remain the
similar. Where there are no facilitities to support AKI or
CKD, people will die.
Areas of Controversy, Confusion, or Non-consensus
Interpretation of published data examining the influence of
pre-existing CKD on the increased likelihood of AKI is
potentially confounded by a number of issues. These include
the comorbidities associated with CKD, influenced by
repeated exposure to various nephrotoxic insults or in-
hospital errors,
57,277
or primarily due to the altered
physiology in CKD. There are also methodological issues
such as how CKD and AKI are defined in clinical studies and
the varying statistical adjustments for comorbidities which
may affect the validity of observed associations.
A further important issue to clarify is whether pre-existing
CKD influences the outcome of AKI. Currently, there is no
single biomarker that can differentiate ‘acute’ from ‘chronic’
kidney disease and help to address this issue. Several large
observational and database studies report, surprisingly, lower
in-hospital mortality in patients with AKI superimposed on
CKD compared with controls.
278-283
Data from PICARD
reveal lower in-patient mortality and median length of stay in
ICU subjects with acute-on-chronic renal injury compared
with non-CKD subjects with AKI, though the post-discharge
dialysis rates were higher in subjects with pre-existing CKD.
284
Pediatric Considerations
The relative paucity of pediatric specific guidelines (due to
lack of high-quality studies) in the KDIGO AKI guideline
would suggest that the use of pediatric data and review
papers as well as relevant pediatric nephrology texts would be
of benefit to the practitioner interested in reviewing this topic
in greater detail and applying pediatric data to their
practice.
653–658
RESEARCH RECOMMENDATIONS
Prospectively designed clinical studies with a clear and
uniform definition of CKD and AKI and adjusted for
comorbidities are needed to determine:
K the frequency of AKI events in a CKD population
K the outcome of AKI in patients with CKD condition
Kidney International Supplements (2013) 3, 91–111 109
chapter 4

K the importance of proteinuria in addition to low GFR in
the risk of AKI
CKD and risk of hospitalization and mortality
Regardless of the method used to estimate GFR, hospitaliza-
tion and mortality rates are higher in people with CKD. Exact
rates var y with comorbidity and severity of CKD, and are not
well-defined. Selection of interventions that could reduce
hospitalizations, morbidit y, mortality, and costs in people
with CKD is not well-studied.
4.6.8: CKD disease management programs should be
developed in order to optimize the community
management of people with CKD and reduce the
risk of hospital admission. (Not Graded)
4.6.9: Interventions to reduce hospitalization and mor-
tality for people with CKD should pay close atten-
tion to the management of associated comorbid
conditions and cardiovascular disease in particular.
(Not Graded)
RATIONALE
There are observational and database studies reporting an
association between pre-existing CKD and hospitalizations
and mortality. A better understanding of the rates, causes,
and risk factors for hospitalization among people with CKD
would allow estimates of the economic burden of CKD and
identification of those at risk for increased resource
utilization. People with CKD are an ideal target for
interventions aimed at reduction of morbidity, hospitaliza-
tion, mortality, and costs. These statements suggest that a
coordinated approach to the identification and management
would result in better outcomes and are intended as ‘best
practices’ statements, recognizing the difficulty in developing
an evidence base while addressing issues related to resource
allocations (Table 34).
Evidence Base
Mortality rates remain high (16-22%) with the use of
dialysis, with more than half of all deaths related to
CVD. Less is known about mortality and CVD rates, and
resource use among persons with a reduced GFR who are
not yet receiving maintenance dialysis. Few studies have
investigated the association between CKD and the risk of
hospitalization.
Data from the USRDS reveal that hospitalization rates
vary with comorbidity and interact with degrees of CKD.
Adjusted rates are 38% higher in people with CKD and 19%
greater in people with CKD and GFR under 60 ml/min/
1.73 m
2
than in those with GFR 460 ml/min/1.73 m
2
, who
in turn are 20% higher than in people w ithout CKD-
illustrating the graded impact of advancing kidney disease.
Not surprisingly, rates of cardiovascular hospitalization are
greater for people with CKD, particularly those with
increasing severity of CKD.
659
In both the CKD and non-
CKD populations, adjusted rates of hospitalization increase
with greater comorbidity. In 2008, for example, the rate for
people with CKD with both diabetes and CHF was 726 per
1000 person-years at risk — 85% greater than the rate of 393
among people with neither diagnosis. Rates of admission for
CVD increase even more in the higher categories of GFR.
Among Medicare subjects, the rate of 141 admissions per 1000
person-years for those with GFR o60 ml/min/1.73 m
2
is 26%
higher than the rate of 112 reported for those with CKD and
GFR Z60 ml/min/1.73 m
2
. The admission rates of 101 and 90
reported for MarketScan and Ingenix i3 subjects with GFR
o60 ml/min/1.73 m
2
are 48 and 16% greater, respectively,
than those occurring in people with higher GFRs.
633
Adjusted
rates of mortality in USRDS 2008 increased with age, and
were highest in people with advanced categories of GFR:
31–72% higher, for example, in people with GFR o60 ml/
min/1.73 m
2
compared to those with no CKD. By gender,
rates in men with CKD were 91.8 per 1000 person-years at
risk compared to 85.6 in women. Rates for people with CKD
overall were similar in whites and African Americans, but in
people with GFR o60 ml/min/1.73 m
2
, rates for African
Americans were 18% higher than those for whites, at 95.0 and
80.5 per 1000 person-years, respectively.
633
Khan et al.
660
confirmed that hospital utilization among
people w ith CKD is high. During a median follow-up of 11.4
months, 47% of subjects had at least one hospitalization and
there were on average 0.96 hospitalizations, 6.6 hospital days,
and 4.0 outpatient nephrology visits per person-year at risk.
Cardiac disease/hypertension was the most common primar y
diagnosis of hospitalizations and progression of CKD/acute
kidney failure was the most common secondary cause of
hospitalization. The authors had previously shown that the
dialysis population at their institution had 2.2 hospit aliza-
tions and 14.8 hospital days per person-year at risk,
659
which
was similar to that among US hemodialysis patients between
1996 and 1998, who had 1.9 hospitalizations and 14 hospital
days per person-year at risk.
661
In the general population,
there were 0.31 hospitalizations and 1.9 hospital days per
person in 1998.
662
Go et al.
58
reported an independent, graded associatio n
between GFR and the risk of death, cardiovascular events,
and hospitalization in 1,120,295 adults w ithin a large,
integrated system of health-care delivery in whom SCr had
been measured between 1996 and 2000 and who had not
Table 34 | Components of community CKD management
programs
Disease monitoring
Integration with other chronic disease management programs including
diabetes, hypertension and heart failure
Medication management and dietary advice
Anemia management programs
Vaccination programs
Information and psychosocial support
Renal replacement therapy (dialysis and transplant) education
Advanced care planning and end-of-life care (where appropriate)
Abbreviation: CKD, chronic kidney disease.
110 Kidney International Supplements (2013) 3, 91–111
chapter 4

undergone dialysis or kidney transplantation. These risks
were evident at GFR o60 ml/min/1.73 m
2
and substantially
increased at GFR o45 ml/min/1.73 m
2
.
The higher hospital utilization among people with CKD
compared to the general population, and the similarity in the
comorbid conditions and the causes of hospitalization
between people with CKD and ESRD, confirm the hypothesis
that the complicati ons and comorbidity observed in ESRD
are manifest earlier in the disease process. Previous studies
have demonstrated an association between age, gender, race,
cardiac disease, peripheral vascular disease, serum albumin
and hematocrit levels, and resource utilization among people
on dialysis.
659,663,664
Holland et al.
665
identified baseline
predictors of first non-elective hospitalization among a
retrospective cohort of 362 predialysis subjects. Multivariate
analysis, adjusted for baseline creatinine concentration,
selected advanced age (RR 1.02; 95% CI 1.01–1.03), angina
(RR 1.9; CI 1.37–2.61), peripheral vascular disease (RR 1.55,
CI 1.05–2.27), and Hb concentration (RR 0.99, CI 0.94–0.98)
as independent predictors of hospitalization. These
comorbid conditions progressively worsen with advancing
kidney disease and result in a substantial proportion of
people having severe complications by the time they come
to RRT.
Interventions to reduce hospitalizations and mortality for
people with CKD should pay close attention to the manage-
ment of associated comorbid conditions and CVD.
666
Closer
attention to the management of heart disease in this
population could substantially improve outcomes.
667
The influence of correction of anemia on hospitalization is
controversial. Dru
¨
eke et al.
668
randomly assigned 603 subjects
with GFR 15-35 ml/min/1.73 m
2
and mild-to-moderate
anemia (Hb level: 11.0-12.5 g/dl [110-125 g/l]) to a target Hb
value in the normal range (13.0-15.0 g/dl [130-150 g/l]) or
the subnormal range (10.5-11.5 g/dl [105-115 g/l]). There
were no significant differences between the two groups in the
incidence of hospital admission (61% and 59%, respectively)
or mean duration of hospitalization for cardiovascular
reasons (33.0 and 28.2 days, respectively). Singh et al.
669
studied 1432 people with CKD, 715 of whom were randomly
assigned to receive a dose of epoetin alfa targeted to achieve
a Hb level of 13.5 g/dl (135 g/l) and 717 of whom
were assigned to receive a dose targeted to achieve a level
of 11.3 g/dl (113 g/l). The median study duration was
16 months. The primary end point was a composite of death,
MI, hospitalization for CHF (without RRT), and stroke. They
observed an increased risk of the primary composite end
point in the high Hb group as compared with the low Hb
group. Death and hospitalization for CHF accounted for
74.8% of the composite events. The Trial to Reduce
Cardiovascular Events with Aranesp Therapy (TREAT)
375
study involved 4038 people w ith diabetes, CKD, and anemia.
Subjects were randomly assigned to treatment with darbe-
poetin alfa to achieve a Hb level of approximately 13 g/dl
(130 g/l) or to placebo, with rescue darbepoetin alfa when the
Hb level was less than 9.0 g/dl (90 g/l). The primary end
points were the composite outcomes of death or a
cardiovascular event (nonfatal MI, CHF, stroke, or hospita-
lization for myocardial ischemia) and of death or ESRD.
Again there were no significant between-group differences in
the outcomes of interest.
International Relevance
It is of benefit for all jurisdictions to appreciate the increase
in resource utilization by CKD populations. Identification
of people at risk for increased resource utilization and
economic burden of CKD should result in strategies to
attenuate that risk or address the resource implications.
Selection of interventions that could reduce hospitalizations,
morbidity, mortality, and costs in these populations should
be evaluated.
DISCLAIMER
While every effort is made by the publishers, editorial board,
and ISN to see that no inaccurate or misleading data, opinion
or statement appears in this Journal, they wish to make it clear
that the data and opinions appearing in the articles and
advertisements herein are the responsibility of the contributor,
copyright holder, or advertiser concerned. Accordingly, the
publishers and the ISN, the editorial board and their respective
employers, office and agents accept no liability whatsoever for
the consequences of any such inaccurate or misleading data,
opinion or statement. While every effort is made to ensure that
drug doses and other quantities are presented accurately,
readers are advised that new methods and techniques
involving drug usage, and described within this Journal,
should only be followed in conjunction with the drug
manufacturer’s own published literature.
Kidney International Supplements (2013) 3, 91–111 111
chapter 4

Chapter 5: Referral to specialists and models of care
Kidney International Supplements (2013) 3, 112–119; doi:10.1038/kisup.2012.68
Early identification and referral of people with CKD has the
potential to reverse, delay, or prevent progression of disease
and is a key focus of international initiatives in the area of
kidney disease. The goals of early identifi cation and referral
are several-fold and include:
1. Provision of specific therapy based on diagnosis
2. Slowing/arresting CKD progression
3. Evaluation and management of comorbid conditions
4. Prevention and management of CVD
5. Identification, prevention, and management of CKD-
specific complications (e.g., malnutrition, anemia, bone
disease, acidosis)
6. Planning and preparation for RRT (e.g., choice of
modality, access-placement and care, preemptive trans-
plantation)
7. Psychosocial support and provision of conservative care
and palliative care options where required
5.1: REFERRAL TO SPECIALIST SERVICES
5.1.1: We recommend referral to specialist kidney care
services for people with CKD in the following
circumstances (1B):
K AKI or abrupt sustained fall in GFR;
K GFR o30 ml/min/1.73 m
2
(GFR categories
G4-G5)*;
K a consistent finding of significant albuminuria
(ACR Z300 mg/g [Z30 mg/mmol] or AER
Z300 mg/24 hours, approximately equivalent
to PCR Z500 mg/g [Z50 mg/mmol] or PER
Z500 mg/24 hours);
K progression of CKD (see Recommendation
2.1.3 for definition);
K urinary red cell casts, RBC 420 per high
power field sustained and not readily
explained;
K CKD and hypertension refractory to treatment
with 4 or more antihypertensive agents;
K persistent abnormalities of serum potassium;
K recurrent or extensive nephrolithiasis;
K hereditary kidney disease.
5.1.2: We recommend timely referral for planning renal
replacement therapy (RRT) in people with pro-
gressive CKD in whom the risk of kidney failure
within 1 year is 10-20% or higher
w
, as determined
by validated r isk prediction tools. (1B)
RATIONALE
This statement reminds the practitioner that there is a need
for timely referral for RRT planning in order to ensure good
decision making and outcomes. The use of the word ‘timely’
is vague as this is not yet determined, and is based on patient
and system factors. The actual amount of time required at a
minimum is at least 1 year to ensure appropriate education,
understanding and referrals to other practitioners (e.g.,
vascular access surgeons, transplant teams, etc). The second
part of the statement refers to the fact that those who are
progressing (versus those who are stable) are the ones who
will benefit from this referral. Hence, there is a need to apply
prediction tools to help identify the risk of progression. We
have not stated which prediction tool is preferred as these
may differ dependin g on information available in any
individual or local experience. Examples of prediction tools
can be found in recent publications.
257,260,261
The scope of nephrology practice includes a wide variety
of conditions including not only ESRD but also acute and
chronic primary and systemic diseases involving individual
elements of the kidney, resistant hypertension, and biochem-
ical derangements. There are thus more potential benefits of
nephrology referral than those widely recognized such as
identification of reversible causes of CKD, provision of
treatment that may slow progression of CKD, management of
the metabolic complications of advanced CKD, and prepara-
tion for dialysis and transplantation.
In certain people, such as those with diabetes, transition to a
severe reduction in GFR and kidney failure may progress rapidly.
In such individuals early nephrology referral is the watchw ord
but when an individual’ s kidney function is relatively stable (rate
of decline in GFR o5 ml/min/1.73 m
2
/year), we suggest using
the grid as a guide (Figure 21). Where refer is marked by an
asterisk, referring clinicians may wish to discuss with their
nephrology service depending on local arrangements.
Evidence Base
Although referral recommendations in the literature are
inconsistent, criteria for nephrology referral include SCr or
GFR, proteinuria, hematuria, BP, and electrolyte derange-
chapter 5 http://www.kidney-international.org
& 2013 KDIGO
*If this is a stable isolated finding, formal referral (i.e., formal consultation
and ongoing care management) may not be necessary and advice from
specialist services may be all that is required to facilitate best care for the
patients. This will be health-care system dependent.
w
The aim is to avoid late referral, defined here as referral to specialist services
less than 1 year before start of RRT.
112
Kidney International Supplements (2013) 3, 112–119
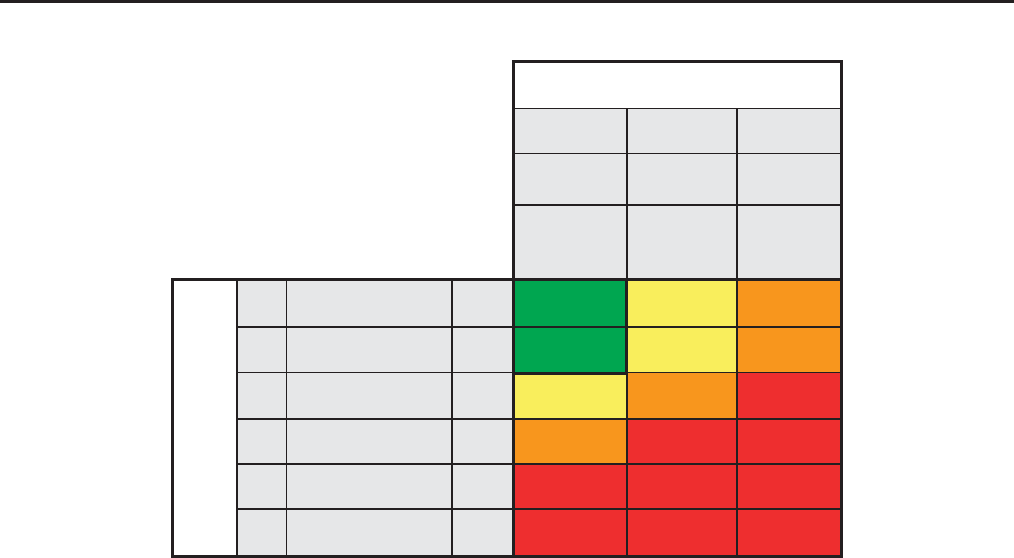
ment.
670
Data relating to referral for those with glomerular
disease, hypertension, AKI, and diabetes may be found in the
relevant guidelines.
7,8,10,262
In this section we will briefly consider summaries of the
evidence relating to timely referral for planning RRT in
people with progressive CKD. In this aspect the literature
concerning late referral in the last quarter of a century has
been remarkably consistent; both studies and narrative
reviews identifying a number of adverse consequences of
late referral and related benefits of early referral (Table 35).
Patients who are aged 475 years, female, non-Caucasian,
uninsured, of lower socioeconomic or educational status, or
have multiple comorbidities are most at risk for non-referral
for CKD care.
671,672
Patients with kidney disease have never
been randomized to early or late referral to nephrology
services and the definition of late referral in the published
studies varies; three months is probably less than the absolute
minimum amount of time required for assessment, education,
preparation for RRT and creation of access but is the most
frequently employed definition. Overall there are more than 50
studies in the published literature and a meta-analysis of 22 of
these studies from 10 different countries serves to underline
some of the key messages (Table 36), giving an indication of
the size of the differences in mortality and hospital length of
stay and also highlighting the significantly lower serum
albumin level in late referred patients.
673
A systematic review considered twenty-seven longitudinal
cohort studies providing data on 17,646 participants of
whom 11,734 were referred early and 5912 (33%) were
referred late.
674
OR for mortality reductions in patients
referred early were evident at 3 month s (OR 0.51; 95% CI
0.44-0.59; Po0.0001) and remained significant at 5 years
(OR 0.45; 95% CI 0.38-0.53; Po0.0001). Initial hospitaliza-
tion was 8.8 days shorter with early referral (95% CI -10.7 to
-7.0 days; Po0.0001). Differences in mortality and hospita-
lization data between the 2 groups were not explained by
differences in prevalence of diabetes mellitus, previous CAD,
BP control, serum phosphate, and serum albumin. Early
referral was associated with better preparation and earlier
placement of dialysis access and better uptake of peritoneal
dialysis.
Over a decade ago McLaughlin et al. evaluated the cost
implications of early versus late referral.
675
Outcomes of
interest were total cost of patient care, patient life-years,
patient life-years free of RRT and hospital length of stay.
Mean total costs per patient over five years were US$87,711
and US$110,056 for early and late referrals, respectively. The
mean patient life-years were 3.53 and 3.36 years, respectively,
and the patient life-years free of RRT were 2.18 and 1.76
years, respectively. Those patients referred early spent
significantly less time in hospital (length of stay 25 days
versus 41 days). Klebe et al. subsequently investigated the
annualized cost of implementation of referral guidelines for
CKD.
676
Although CKD guideline implementation resulted
in significant increases in nephrology referral and additional
investigation, they estimated that the associated costs could
Persistent albuminuria categories
Description and range
A1 A2 A3
Normal to
mildly
increased
Moderately
increased
Severely
increased
GFR categories (ml/min/ 1.73 m
2
)
Description and range
G1 Normal or high ≥90 Monitor Refer*
G2 Mildly decreased 60–89 Monitor Refer*
G3a 45–59 Monitor Monitor Refer
G3b 30–44 Monitor Monitor Refer
G4 Severely decreased 15–29 Refer* Refer* Refer
G5 Kidney failure <15 Refer Refer Refer
Mildly to moderately
decreased
Moderately to
severely decreased
<30 mg/g
<3 mg/mmol
30–300 mg/g
3–30 mg/mmol
>300 mg/g
>30 mg/mmol
Figure 21 | Referral decision making by GFR and albuminuria. *Referring clinicians may wish to discuss with their nephrology service
depending on local arrangements regarding monitoring or referring. GFR, glomerular filtration rate. Modified with permission from
Macmillan Publishers Ltd: Kidney International. Levey AS, de Jong PE, Coresh J, et al.
30
The definition, classification, and prognosis of chronic
kidney disease: a KDIGO controversies conference report. Kidney Int 2011; 80: 17-28; accessed http://www.nature.com/ki/journal/v80/n1/
full/ki2010483a.html
Kidney International Supplements (2013) 3, 112–119 113
chapter 5

be recouped by delaying dialysis requirement by 1 year in one
individual per 10,000 patients managed according to guide-
lines.
International Relevance
Local practice and resource will dictate local referral practice
but regardless of the health-care system, delay or prevention
of progression of both CKD and complications associated
with CKD will add value. Local organizations will determine
the best methods of communication and interaction between
patients, specialists, and primary care physicians.
Implications for Clinical Practice and Public Policy
Implementation of referral guidelines will inevitably lead to
an increased workload for specialist nephrology services.
However, introduction of local initiatives in conjunction with
primary care providers can improve the appropriateness and
quality of the referral. Loca l initiatives combined with
national policy and practice changes can lead to an
improvement in the outcomes of CKD patients regardless
of the level of resource available.
Pediatric Considerations
Current pediatric practice in most areas of the world would
suggest a higher level of kidney function for referral than that
for adults, though the principles remain the same. Much of
the pediatric nephrologist’s consultation occurs from infants,
or even antenatal sources, where the identification of the
child as being ‘at-risk’ may be apparent from radiographic
studies performed in utero. The relatively non-specific signs
and symptoms of most forms of renal disease in the young
child mandates a higher level of suspicion in the referring
physician, and a lower threshold of acceptance of the consult
in the subspecialty clinic accepting these referrals.
Attempts to develop universal guidelines for referral
of children to pediatric nephrology services would be
dependent on local resources (as is the case for adults) and
it is of value to consider in broad categories the types of
conditions for which referral to a pediatric nephrologist
would be expected to provide benefit to the referring
physician and patient/family.
In a recent review Barakat
677
attempts to address a
number of these issues by outlin ing the most common
presentations of a child with significant renal disorders.
Barakat and Chesney also suggest a number of specific areas
in terms of initial investigations, management, and follow-up
where the primary care physician can legitimately play a role
in the care of the child with renal disease and provides a list
of suggested referral triggers.
678
While there will be variation in referral triggers, referral
would be recommended for the following: acute or chronic
reduction in renal function, poorly treated or severe
hypertension, severe electrolyte abnormalities, the finding
of significant abnormalities in urina ry tract structure, or the
presence of systemic diseases likely to produce renal effects.
Similarly, the need for education in progressive conditions,
performance of and interpretation of renal biopsies, and
allaying parental/patient anxiety would also be acceptable
reasons for referral.
There is no ‘minimum acceptable value’ of renal
dysfunction below which one can be certain to see significant
abnormalities in clearance, electrolyte, or other side effects
associated with progressive renal disease in children such as
growth failure or neurocognitive issues. However, the
Table 35 | Early versus late referral: consequences and benefits
Consequences of late referral Benefits of early referral
Anemia and bone disease Delay need to initiate RRT
Severe hypertension and fluid overload Increased proportion with permanent access
Low prevalence of permanent access Greater choice of treatment options
Delayed referral for transplant Reduced need for urgent dialysis
Higher initial hospitalization rate Reduced hospital length of stay and costs
Higher 1-year mortality rate Improved nutritional status
Less patient choice of RRT modality Better management of CVD and comorbid conditions
Worse psychosocial adjustment Improved patient survival
Abbreviations: CVD, cardiovascular disease; RRT, renal replacement therapy.
Table 36 | Outcomes of early versus late referral
Variable Early referral mean (SD) Late referral mean (SD) P value
Overall mortality, % 11 (3) 23 (4) o0.0001
1-year mortality, % 13 (4) 29 (5) 0.028
Hospital length of stay, days 13.5 (2.2) 25.3 (3.8) 0.0007
Serum albumin at RRT start, g/dl [g/l] 3.62 (0.05) [36.2 (0.5)] 3.40 (0.03) [34.0 (0.3)] 0.001
Hematocrit at RRT start, % 30.54 (0.18) 29.71 (0.10) 0.013
Abbreviation: RRT, renal replacement therapy.
Adapted from Am J Med, Chan MR, Dall AT, Fletcher KE, et al.
673
Outcomes in patients with chronic kidney disease referred late to nephrologists: a meta-analysis. 120:
1063-1070, 2007, with permission from Elsevier; accessed http://download.journals.elsevierhealth.com/pdfs/journals/0002-9343/PIIS000293430700664X.pdf
114 Kidney International Supplements (2013) 3, 112–119
chapter 5

KDOQI CKD Guidelines
1
stated that while a child with a
eGFR o30 ml/min/1.73 m
2
warranted referral to a pediatric
nephrologist in all cases, that in fact it was also reasonable to
consider any child with evidence of CKD– and in particular
those with eGFR o60 ml/min/1.73 m
2
– for referral to a
pediatric nephrologist regarding evaluation and manage-
ment.
43
It is reasonable to presume that, as in adults, the concept
of referral ‘early’ or ‘above’ some minimum level of function
should allow for numerous benefits to the patient and family,
but again there are few data for this in children. The most
informative information on both of these issues can be
derived from two papers examining the issue of late referrals
of children who eventually required ESRD care. Kennedy
et al.
679
demonstrated that at the time of referral to their
center, the children 4 1 year of age had a median Schwartz
eGFR of only 27 (IQR 9-52) ml/min/1.73 m
2
, and in fact 55%
of these children over the age of 1 year were referred with an
eGFR o30 ml/min/1.73 m
2
, meeting one of two definitions
of a late referral. When they considered their second
definition of initiation of RRT within 90 days of referral,
commonly used in the literature for this par ticular topic,
30% of the eligible cohort required RRT within that 90-day
window and hence were considered late referrals on this
basis. The potential effect on the child’s health could be
ascribed to such a delay in referral: those children had
lower mean Hb (8.7
±
0.6 g/dl [87
±
6 g/l]) versus (12.8
±
0.6 g/dl [128
±
6 g/l]) and higher median urea (34 (IQR
5-14) mmol/l [203 (IQR 30-84) mg/dl] versus 6 (IQR 5-14)
mmol/l [36 (IQR 30-84) mg/dl], Po0.001) than those who
presented with eGFR 430 ml/min/1.73 m
2
. Secondary
analysis confirmed this was also true in the groups if one
looked at those above or below an eGFR of 60 ml/min/
1.73 m
2
. The second paper by Boehm and colleagues
680
was a
retrospective single pediatric center report that demonstrated
that over B30 years of referrals (N ¼ 111), 24% of them
could be considered as late referrals, as defined by need for
RRT within 90 days of referral. In this late referral group, the
eGFR (Schwartz) was significantly lower than those present-
ing later, (14.9 versus 34.2 ml/min/1.73 m
2
,Po0.001) as was
the Hb at presentation (8.0 g/dl [80 g/l] versus 10.5
±
2.3 g/
dl [105
±
23 g/l]; Po0.001). Importantly the Hb deficit
persisted at the time of RRT initiation, with the Hb in the late
referral group being 8.5 g/dl [85 g/l] versus 9.8
±
1.9 g/dl [98
±
19 g/l] in the earlier referred patients, Po0.01. The other
metric of interest chosen by these authors to evaluate the
possible detrimental effect of late referral was the likelihood
of a given child having a pre-emptive transplant, i.e., prior to
dialysis initiation. While this association was not confirmed
in Kennedy’s paper, Boehm and his group demonstrated that
in their patient population children who were referred late
only 11% were preemptively transplanted as compared to
40% of children who presented to care more than 90 days
prior to the need for some form of RRT. Of further interest,
although somewhat counterintuitive, the proportion of
patients in the Boehm study who initiated hemodialysis
was not statistically different between those in the late versus
early referral groups, 62% versus 67%, Po0.05 respectively.
The underlying concept of referral to a pediatric
nephrologist in the face of rapid progression of renal failure
is of course applicable in full. However, no current validated
risk of progression tool exists in pediatric nephrology. As to
what level of functional decline might be considered
‘concerning,’ the best evidence is derived from the long-
itudinal iohexol GFR data as accrued in the CKiD trial. Data
from that study related to the annualized rate of decline in
renal function for children with glomerular conditions is
10.5% as compared to those with a non-glomerular causes
in whom the annualized rate of change is only 3.9%.
71
Any
child whose rate of decline exceeded these two values would at
least warrant much closer follow-up and/or investigation for
modifiable factors to slow progression. Note this is not to say
these rates of decline should be considered to be ‘normal’ for
either category. All efforts to slow decline in renal function
would still be of vital importance in every individual.
5.2: CARE OF THE PATIENT WITH PROGRE SSIVE CKD
The following section describes recommended structures and
key milestones for people with progressive CKD. The
recommendations are intended to model best practices but
it is appreciated that different health-care systems, geogra-
phical issues, and economic considerations will have variable
abilities to implemen t these recommendations.
Key aspects of caring for people with progressive CKD, as
they approach end of life or RRToptions are addressed in this
section.
5.2.1: We suggest that people with progressive CKD should
be managed in a multidisciplinary care setting. (2B)
5.2.2: The multidisciplinary team should include or have
access to dietary counseling, education and counsel-
ing about different RRT modalities, transplant
options, vascular access surgery, and ethical, psy-
chological, and social care. (Not Graded)
RATIONALE
Optimal care is that care which leads to the best outcomes for
the individual, the population, and society. The model of care
varies according to CKD severity, which will determine the
target population and goals. These statements are worded to
predominantly encompass those people likely to progress to
ESRD. CKD models of care follow the same principles
embodied in the chronic disease model of care (Figure 22).
The specific components for CKD models of care include:
protocols for laboratory and clinic v isits; attention to
cardiovascular comorbidities and CKD-associated comorbid-
ities such as anemia; a vaccination program (see Recommen-
dation 4.6.1-4.6.6); an education program which includes
both general CKD and RRT education (including conserva-
tive management where appropriate); self-management;
lifestyle modification including diet, exercise, and smoking
Kidney International Supplements (2013) 3, 112–119 115
chapter 5
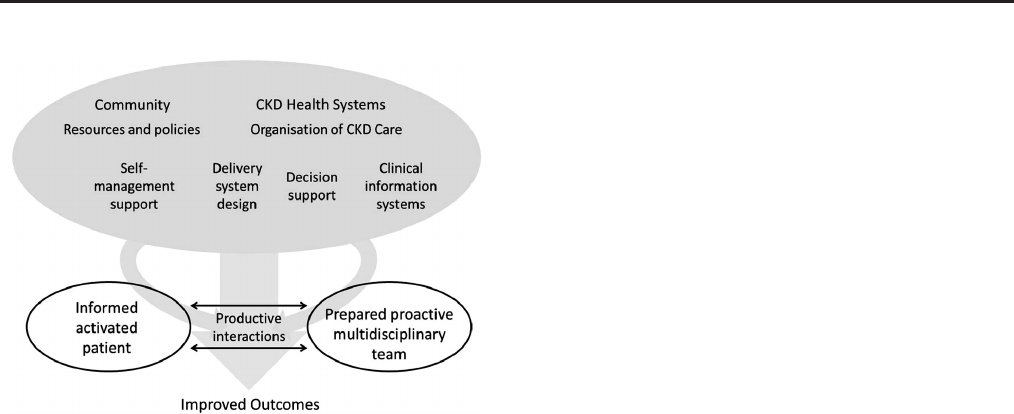
cessation; and counseling and support for factors such as
social bereavement, depression, and anxiety.
International Relevance
Standardized and culturally appropriate protocols should be
consider ed. While it is recognized that resources may vary across
and within jurisdictions, recommendations here are based on
principles of care, which should be relevant across the globe.
Implications for Clinical Practice and Public Policy
CKD is a complex condition and co-exists with many other
conditions. Therefore models of care should be developed
that integrate the complexity of the clinical conditions
involved, patient-centered philosophies, and the health-care
environment. The principles of care are universal but
implementation may be customized to specific circum-
stances.
Pediatric Considerations
Papers addressing the need for and effectiveness in the utility
of multidisciplinary clinics for management of pediatric CKD
patients have been published. In 2012, Ajarmeh et al.
682
des-
cribed a retrospective comparison of two cohorts of children
aged 0-18 years of age and followed at a single Canadian
tertiary care referral center over a 1-year time period either
before or followin g the full implementation of a multi-
disciplinary CKD clinic which included the services of
dedicated pediatric nephrologists, renal nursing, pharmacy,
dietitians, social workers, and a renal database manager. In
2009, Menon et al.
683
published data from a large American
pediatric referral center and compared the outcomes of
children from one of two 5-year cohorts in the period
immediately preceding or following the initiation of a specific
Chronic Renal Insuffi ciency clinic which was staffed by
pediatric nephrologists, nurse clinicians, transplant coordi-
nators, dietitians, social workers, and a psychologist.
Ajarmeh et al.
682
compared the two cohorts in a number of
areas during their year of follow-up whereas Menon et al.
683
chose to look at each individual patient in the 12 months
immediately prior to initiating RRT. While the studies had
different variables of interest and used slightly different
approaches for comparison of results between their groups,
both demonstrated a reduction in the rate of decline in
estimated renal function, improved Hb levels, and improve-
ment in at least some aspect of bone mineral metabolism
control in their respective cohorts cared for in the multi-
disciplinary era. In addition, both studies also demonstrated a
decrease in resource utilization by patients in the multi-
disciplinary clinic as measured by median length of stay
682
and
number of unplanned admissions per patient per year.
683
Also
of note, Menon et al. demonstrated that in the patients who
initiated dialysis (which was how the cohort was chosen), more
patients who started hemodialysis did so with a functioning
arteriovenous fistula or arteriovenous graft at the time of
initiation compared to those from the general nephrology clinic
era (85.7 versus 20%, P ¼ 0.02).
5.3: TIMING THE INITIATION OF RRT
5.3.1: We suggest that dialysis be initiated when one or
more of the following are present: symptoms or
signs attributable to kidney failure (serositis, acid-
base or electrolyte abnormalities, pruritus); inability
to control volume status or blood pressure; a
progressive deterioration in nutritiona l status re-
fractory to dietary intervention; or cognitive im-
pairment. This often but not invariably occurs in the
GFR range between 5 and 10 ml/min/1.73 m
2
.(2B)
5.3.2: Living donor preemptive renal transplantation in
adults should be considered when the GFR is
o20 ml/min/1.73 m
2
, and there is evidence of
progressive and irreversible CKD over the pre-
ceding 6-12 months. (Not Graded)
RATIONALE
The statement is worded very precisely to highlight the need
for RRT to address symptoms and to avoid the institution of
dialysis therapy at an arbitrary number representing the
degree of residual renal function. Given the risks and benefits
of RRT, as well as the potential imprecision of measurements,
patients need to be treated according to symptoms and signs,
not simply based on a laboratory value.
Data from national registries has shown a consistent rise
in GFR at initiation of RRT. This rise has been driven partly
by a desire to improve nutritional status and also by earlier
observational data suggesting adverse outcomes associated
with RRT initiation at lower GFR. These early data were
subject to much confounding. RCT data, supported by large
registry-based studies corrected for confounding, show no
survival advantage to early start dialysis (Initiating Dialysis
Figure 22 | The CKD chronic care model. CKD, chronic kidney
disease. Adapted by permission from BMJ Publishing Group
Limited. Improving the quality of health-care for chronic
conditions. Epping-Jordan JE, Pruitt SD, Bengoa R, et al.
681
Qual Saf Health Care. 13: 299-305, 2004; accessed
http://qualitysafety.bmj.com/content/13/4/299.full.pdf+html
116 Kidney International Supplements (2013) 3, 112–119
chapter 5

Early and Late [IDEAL] study, see Evidence Base Below).
Thus, the statement as written should help the clinician to
balance symptoms with laboratory values in decision making.
The statement regarding living donor transplantation is
intended to ensure that practitioners think about this option
in the context of the totality of RRT options. It is worded to
be concordant with local living donation transplant policies.
It aims to reflect the need for risk-benefit assessment of
preemptive renal transplantation in all individuals. Note that
the statement asks clinicians to consider this option, but does
not state at which level of GFR the preemptive transplant
would occur as this is a matter of local practice and patient-
specific considerations.
Evidence Base
The perceived survival advantage of early start of dialysis was
questioned by a study from the Netherlands Cooperative
Study on the Adequacy of Dialysis Study Group (NECO-
SAD).
684
Of 253 patients with new ESRD, 94 (37%)
started dialysis late (GFR 4.9
±
1.7 ml/min/1.73 m
2
) and 157
started in a timely fashion (GFR 7.1
±
2.4 ml/min/1.73 m
2
).
Although there was a small gain in survival time over 3 years
after start of dialysis for the timely start group (2.5 months)
there was no significant difference in survival between the
2 groups and the gain in survival time was offset by a n
estimated lead-time bias of between 4.1 and 8.3 months. In a
study eliminating the effect of lead-time bias Traynor et al.
found no benefit in survival in those initiating dialysis
early.
685
Subjects starting dialysis early (N ¼ 119) had a
median IQR CrCl of 10.4 (9.1-11.9) ml/min versus 6.7 (5.6-
7.5) ml/min in 116 subjects starting dialysis late. After
correction for multiple confounders, they found an 11%
greater hazard for mortality with each 1 ml/min increase in
CrCl at start of dialysis (HR 1.11; 95% CI 1.01-1.21;
P ¼ 0.024). Subsequent observational studies, including large
dialysis registry studies from around the globe, have all
suggested increased mortality when dialysis is started
early.
686–696
These studies encountered multiple difficulties
in drawing conclusions from this observational approach
including lead-time bias, problems w ith estimating GFR from
SCr in people with low muscle mass and/or fluid overload,
the fact that people with symptoms and/or increased
comorbidity are more likely to start dialysis early, and finally
survivor bias in studies including people only when they start
dialysis (and therefore excluding those who die prior to
starting diaysis). These confounders were largely addressed by
a multicenter controlled trial in which 828 adults with
progressive CKD and CrCl 10-15 ml/min/1.73 m
2
were
randomly assigned to early (CrCl 10-14 ml/min [0.17-
0.23 ml/s]) or late (CrCl 5-7 ml/m in [0.08-0.12 ml/s]) initia-
tion of dialysis.
697
The study protocol allowed an earlier start
where deemed clinically necessary by investigators and
although the difference in GFR at dialysis initiation between
the 2 groups was significant (early star t CrCl 12.0 versus late
start 9.8 ml/min, MDRD GFR 9.0 versus 7.2 ml/min/
1.73 m
2
), it was less than planned because 19% of early
starters started late and 76% of late starters started early. There
was no difference in mortality between the early and late start
groups and no difference in the study’s secondary outcomes
(cardiovascular events, infectious events and complications of
dialysis) between the 2 groups. Finally, a further observational
study sought to examine the effect of early initiation of dialysis
on survival in a ‘healthy’ group of 81,176 subjects with ESRD
aged 20-64 years, without diabetes, and with no comorbidity
other than hypertension.
698
The unadjusted 1-year mortality
by MDRD GFR at dialysis initiation ranged from 6.8% in the
reference group (GFR o5.0 ml/min/1.73 m
2
)to20.1%in
the highest GFR group (Z15.0 ml/min/1.73 m
2
). In an even
healthier subset of 35,665 subjects with serum albumin
concentrations of 3.5 g/dl or higher prior to hemodialysis
initiation, the 1-year mortality was 4.7%. In this group the
adjusted HR for mortality was 1.27 for GFR 5.0-9.9 ml/min/
1.73 m
2
, 1.53 for GFR 10.0-14.9 ml/min/1.73 m
2
,and2.18for
GFR Z15.0 ml/min/1.73 m
2
compared with the reference
group of GFR o5.0 ml/min/1.73 m
2
.
International Relevance
The availability of resources for formal multidisciplinary
teams, educational materials, and access to specialized
counseling for diet, advance directives, access planning, and
pre-emptive transplantation varies around the world. These
statements are proposed so that ‘best practices’ can be
documented or aspired to. The need for education, planning,
and appropriate expertise for the management of this patient
group is internationally relevant. The me thods, frequency,
and tools with which this can be accomplished will be region
specific.
Implications for Clinical Practice and Public Policy
There is a need to focus on regular symptom assessment as
part of CKD revie w in those with lower eGFR values.
Individual assessment and availability of resources will
dictate specific timin g of therapies.
Clinicians should be aware of the impact of early dialysis
start on QOL
699
before recommending this strategy to
patients.
Pediatric Consideration
Timing of initiation of dialysis.
As mig ht be expected,
information as to the proper timing for initiation of dialysis
does not exist for children. Thus, it would seem reasonable to
follow the guidelines as set out for the adults. A review by
Greenbaum and Schaefer
700
provides the reader with
generally agreed upon absolute and relative indications for
initiation of dialysis in the child. The absolute indications
listed are those of neurologic consequences attributable to
uremia; hypertension that fails to respond to antihyperten-
sive therapy; pulmonary edema unresponsive to diuretics;
pericarditis; bleeding tendency; and refractory nausea or
vomiting. However, the authors clearly state that there is no
current consensus as to the validity of the relative indications
and specifically the level at which each engenders the need for
Kidney International Supplements (2013) 3, 112–119 117
chapter 5

dialysis. Relative indications to commence dialysis incl ude
less severe uremic symptoms, hyperkalemia, hyperph os-
phatemia, malnutrition, and growth failure. The authors
point out as well that the answer to ‘early’ versus ‘late’
dialysis initiation cannot be addressed in any meaningful
way in children as there is currently lack of data; and the
unique factors to consider in a child such as growth,
psychosocial development, likely renal transplant, and need
for extended time on both dialysis and in transplant make the
probability of a clin ical trial to address this issue nearly
impossible.
Timing of living donor transplant. There is no direct
evidence to guide the pediatric nephrologist as to the ‘best’
timing for a living dono r transplant in children. In each
individual case the relative likelihood of improvement in
presumed uremic symptoms and burden of disease as related
to management of CKD will need to be balanced against the
risks of transplant surgery itself and the attendant medication
risks with prolonged immunosuppression. While there is
little doubt as to the benefit in overall lifespan accrued by a
child who is transplanted as opposed to one maintained on
standard dialysis therapies during their childhood (average
life expectancy of 63 versus 38 years respectively),
701
the issue
as to what level of residual function is sufficient to justify
delay or conversely proceeding to transplant is simply not
readily available.
It should also be noted that reliance on GFR as the sole
marker of disease intensit y is unlikely to be sufficient for
making such a decision, and patient symptoms and/or
unique family factors may play a significant role in the
decision as to timing of a living donor transplant (e.g., a
young grandparent available and healthy to donate at age 60
years may not be eligible at age 63 years and so an earlier
transplant would be favored).
5.4: STRUCTURE AND PROCESS OF COMPREHENSIVE
CONSERVATIVE MANAGEMENT
5.4.1: Conservative management should be an option in
people who choose not to pursue RRT and this
should be supported by a comprehensive manage-
ment program. (Not Graded)
5.4.2: All CKD programs and care providers should be
able to deliver advance care planning for people
w ith a recognized need for end-of-life care,
including those people undergoing conservative
kidney care. (Not Graded)
5.4.3: Coordinated end-of-life care should be available
to people and families through either primary
care or specialist care as local circumstances
dictate. (Not Graded)
5.4.4: The comprehensive conservative management
program should include protocols for symptom
and pain management, psychological care, spiri-
tual care, and culturally sensitive care for the
dying patient and their family (whether at home,
in a hospice or a hospital setting), followed by the
provision of culturally appropriate bereavement
suppor t. (Not Graded)
RATIONALE
These statements are intended to highlight the need for
comprehensiv e conservative care processes and resources in the
care of this complex patient group . There is increasing
recognition that provision of organized care to those who are
dying or choose to not pursue dialysis and transplant care is of
value to the patients and their families. Clinicians involved in
caring for these patients should be alerted to this need. These
statements are positioned so as to overtly state this need.
In different societies or cultural areas, the form
and structure of this care may vary tremendously and
families or religious organizations may be able to deliver
expert and sensitive care. The details here are listed not to be
prescriptive but rather to articulate the ‘best practices’ in
communities where resources may be available and to serve a
construct to review in those locations where resources are
more limited.
Evidence Base
The evidence base for these statements is limited to
documents describing the burden of illness and unm et needs
in pati ents with CKD as they decide either not to pursue
dialysis or to withdraw from RRT. As the readers will
appreciate, there are neither RCTs nor even large cohort
studies to inform these statements within the CKD popula-
tions. Analogies with oncology have been drawn and
literature from there has been used to justify the concepts
above.
Patients with advanced CKD have extensive palliative care
needs for years before death. Throughout their illness
trajectory, symptom burden is high.
702–707
The number and
severity of physical and emotional symptoms are similar to
those of cancer patients hospitalized in palliative care
settings.
703,708
Advanced CKD patients managed without
dialysis are equally symptomatic.
706
An increasing number of
dialysis patients die after withdrawal of dialysis (10-15% in
1990, 20% in 2004), primarily due to poor QOL, representing
the second leading cause of death after CVD.
Conservati ve care focuses on slowing the decline in
renal function, actively managing symptoms, advance care
planning, and the provision of appropriate palliative care.
In oncology, receiving early palliative care is asso ciated with
better QOL, fewer emergency department visits and
hospitalizations, less aggressive care at the end of life, and
surprisingly, longer survival.
709
These observations
suggest that development of clinical models that integrate
appropriate palliative care, including the creation of
conservative care pathways, are likely to be hugely beneficial
and would help avoid harmful dialysis to those
patients unlikely to realize benefit. Such conservative care
programs are slowly being developed, primarily in the UK
and Canada.
118 Kidney International Supplements (2013) 3, 112–119
chapter 5

The literature describing experiences of specific programs
and symptom burden in CKD, as well as advocating for better
care, has only become more prevalent since 2005.
709–716
Poor quality, hospitalized deaths began to improve for
patients dying with cancer as a result of an increasing
emphasis on palliative care teams providing attention to
symptom control, psychosocial needs, and options for
location of care. The literature suggests that current models
of palliative care for cancer patients that deinstitutionalized
health-care services have not only improved patient out-
comes and quality of care
717–720
but have been cost-
neutral.
721
International Relevance
All countries have people with CKD who withdraw either
voluntarily or involuntarily from dialysis services. Best care
for those patients will obviously need to respect cultural and
religious values, but would necessarily be based on the same
philosophical grounds of maintaining dignity of the in-
dividual. Appreciating the need for and articulating con-
servative care pathways overtly would be internationally
applicable.
Implications for Clinical Practice and Public Policy
There is a need to ensure appropriate access to services
and education surrounding quality care during terminal
stages of a chronic condition. There is increasing attention
to this in many societies but not in all. Appreciating the
variability in the resources required and their availability
to different groups of patients is important for implementa-
tion. Furthermore, involvement of religious and cultural
leaders in recognition of these care practices can be enhanced
if they are overtly recognized as part of the care continuum.
There is a need for robust assessment of best practices in
CKD and other chronic conditions so that we may provide
best care throughout the continuum of life. Researchers
around the world are actively pursuing this so that we may
have better tools, programs, and ultimately, be tter outcomes
for our patients.
Pediatric Consideration
Despite the lack of published evidence, there is no reason
to believe that children and families faced with the need
to consider conservative care or a palliative approach to
their impending ESRD would not benefit from all of the
above recommendations. It would seem reasonable for
pediatric nephrology centers to liaise with their pediatric
oncology or palliative care teams, ethicists, and pastoral care
providers to assist them in developing, defining, and/or
identifying locally available resources for situations where a
child is not deemed a candidate for RRT prior to their
occurrence.
DISCLAIMER
While every effort is made by the publishers, editorial board,
and ISN to see that no inaccurate or misleading data, opinion
or statement appears in this Journal, they wish to make it
clear that the data and opin ions appearing in the articles and
advertisements herein are the responsibility of the contri-
butor, copyright holder, or advertiser concerned. Accord-
ingly, the publishers and the ISN, the editorial board and
their respective employers, office and agents accept no
liability whatsoever for the consequences of any such
inaccurate or misleading data, opinion or statement. While
every effort is made to ensure that drug doses and other
quantities are presented accurately, readers are advised that
new methods and techniques involving drug usage, and
described within this Journal, should only be followed in
conjunction with the drug manufacturer’s own published
literature.
Kidney International Supplements (2013) 3, 112–119 119
chapter 5

Methods for guideline development
Kidney International Supplements (2013) 3, 120–127; doi:10.1038/kisup.2012.69
AIM
The overall aim of this project was to develop an evidence-
based clinical practice guideline for evaluation and manage-
ment of CKD. The guideline consists of recommendation
statements, rationales, and a summary of systematically
generated evidence on relevant pre-defined clinical topics. To
a large extent the guideline builds on the output of the
KDIGO Controversies Conference in 2009,
30
which generated
epidemiological data to support a revision of the classifica-
tion and staging system. The vision for this KDIGO guideline
is that it would endorse the current CKD definition as an
imperfect convention for describing a state of function, revise
classification based on risk, revise risk states, and rev ise and
update action plans in view of the revised classifications.
Additional systematic evidence review focused on specific
topics.
OVERVIEW PROCESS
The guideline development process included the following steps:
K Appointing Work Group members and the ERT
K Discussing process, methods, and results
K Developing and refining topics
K Identifying populations, interventions or predictors, and
outcomes of interest
K Selecting topics for systematic evidence review
K Standardizing quality assessment method ology
K Developing and implementing literature-search strategies
K Screening abstracts and retrieving full text articles on the
basis of predefined eligibility criteria
K Creating data extraction forms
K Extracting data and performing critical appraisal of the
literature
K Grading the methodology and outcomes in individual
studies
K Tabu lating data from individual studies into summary
tables
K Grading the strength of recommendations on the basis of
the quality of evidence and other considerations
K Finalizing guideline recommendations and supporting
rationales
K Sending the guideline draft for peer review to the KDIGO
Board of Directors in January 201 2 and for public review
in May 2012
K Publishing the final version of the guideline
Collaboration Among Participants
The KDIGO Co-Chairs appointed the Work Group
Co-Chairs, who then assembled the Work Group of domain
experts, including individuals with expertise in internal
medicine, adult and pediatric nephrology, diabetology/
endocrinology, clinical chemistry, and epidemiology. The
Tufts Center for Kidney Disease Guideline Development and
Implementation at Tufts Medical Center in Boston, Massa-
chusetts, USA, was contracted to conduct systematic evidence
review and provide expertise in guideline development
methodolog y. The ERT consisted of physician–methodolo-
gists with expertise in nephrology, a project coordinator, a
research assistant, and a medical writer–editor. The ERT
instructed and advised Work Group members in all steps of
literature review, critical literature appraisal, and guideline
development. The Work Group and the ERT collaborated
closely throughout the project.
The Work Group and its Chairs, KDIGO Co-chairs, ERT,
and KDIGO support staff met for three 2-day meetings for
training in the guideline development process, topic discus-
sion, and consensus development.
Throughout the project, the ERT offered suggestions for
guideline development and led discussions on systematic
review, literature searches, data extraction, assessment of
quality and applicability of articles, evidence synthesis,
grading of evidence and guideline recommendations, and
consensus development. The Work Group took the primary
role of writing the recommendation statements and ratio-
nales and retained final responsibility for their content.
Defining Scope and Topics
This KDIGO CKD guideline was set out to update the
KDOQI Clinical Practice Guidelines for CKD: Evaluation,
Classification, and Stratification
1
in 2002, which spans many
topics related to the diagnosis, classification, stratification,
and management of CKD.
1
The Work Group Co-Chairs prepared the first draft of the
scope of work document as a series of open-ended questions
to be considered by Work Group members. At their first 2-
day meeting, members added further questions until the
initial working document included all topics of interest to the
Work Group. The inclusive, combined set of questions
formed the basis for the deliberation and discussion that
followed. The Work Group strove to ensure that all topics
deemed clinically relevant and worthy of review were
identified and addressed.
Updating the topics of definitions and classification was
based on the output from the KDIGO Controversies
Conference and the CKD Prognosis Consortium.
4,30
Additional topics that relate to explicit selection of
diagnostic tests or interventions were chosen to undergo
systematic review of the best available evidence. Systematic
methods for guideline development http://www.kidney-international.org
& 2013 KDIGO
120
Kidney International Supplements (2013) 3, 120–127

evidence review entails a priori question formulation,
specification of important outcomes for the review, systema-
tic searches, data extraction, tabulation, analysis, and
synthesis of evidence and is described in detail for each of
the specific questions. The process followed for each evidence
review topic (a total of four non-treatment topics and four
treatment topics) is detailed below.
The eight topics for which the ERT conducted searches
and evidence review are shown in Table 37. For the systematic
review topics, the Work Group and ERT further developed
and refined each topic and specified screening criteria,
literature search strategies, and data extraction forms.
Many other topics were not suitable to be addressed by in-
depth evidence review. When the anticipated outcome of an
extensive literature search was unlikely to yield evidence that
directly informs practice choices, the approach chosen was
that of a narrative review.
APPROACH TO EVIDENCE REVIEW TOPICS
Formulating Questions of Interest
Questions of interest were formulated according to the
PICODD (Population, Intervention or Predictor, Compara-
tor, Outcome, study Design, and Duration of follow-up)
criteria. Details of the PICODD criteria are presented in
Table 37.
Literature Searches and Article Selection for Evidence Review
Topics
Search strategies were developed by the ERT, with input from
the Work Group, for each topic of interest (whether
treatment or non-treatment topics). The ERT performed
literature searches and conducted abstract and article
screening. The ERT also coordinated the methodological
and analytic processes, data extraction, and summarizing of
the evidence. Before initiating our own de novo systematic
review, we searched for existing systematic reviews that could
be used. The searches and search terms are provided in
Supplemental Table 1 and the search dates and yields for all
topics are presented in Table 38.
Selection of Outcomes of Interest
The Work Group selected outcomes of interest on the basis of
their importance for informing clinical decision making.
Importance of mortality and ESRD was considered to be
critical; the importance of progression of CKD and
categorical or continuous measures of kidney function was
considered to be high; and the importance of QOL, BP, gout
attacks, and proteinuria was considered to be moderate.
Data Extraction
Text articles were extracted by the ERT onto forms
customized to capture data on design, methodology, baseline
characteristics, interventions or predictors, comparators,
outcomes, results, and limitations of individual studies.
Study methodology and risk of bias were also systematically
graded for each outcome and recorded.
Summary Tables
Pertinent information for systematic review topics was
tabulated in summary tables. Summary tables list out-
comes of interest as well as relevant population character-
istics, descriptions of inter ventions and comparators,
results, and quality grades for each outcome. Categorical
and continuous outcomes were summarized separately.
Work Group members reviewed all summary table data
and quality grades.
Evidence Profiles
Evidence profiles are usually constructed as a means to assess
the quality and record quality grades and descriptions of
effect for each outcome across studies, as well as the quality
grades and description of net benefits or harms of the
intervention or comparator across studies. These profiles aim
to make the evidence synthesis process transparent. However,
since no treatment or non-treatment topic had more than
one study in a summary table for which the quality was
graded, no evidence profiles were generated, and the
information in the summary table shows the highest level
of synthesis.
Grading of Quality of Evidence for Outcomes of Individual
Studies
Methodological quality.
Methodological quality (internal
validity) refers to the design, c onduct, and reporting of
outcomes of a clinical study. A previously devised three-level
classification system for quality assessment was used to grade
the overall study quality and quality of all relevant outcomes in
the study (Table 39). Variations of this system have been used
in most KDOQI and all KDIGO guidelines and have been
recommended for the US Agency for Healthcare Research and
Quality Evidence-based Practice Center program.
722
Each study was given an overall quality grade on the basis
of its design, methodology (randomization, allocation,
blinding, definition of outcomes, appropriate use of statis-
tical metho ds, etc.), conduct (drop-out percentage, outcome
assessment methodologies, etc.), and reporting (internal
consistency, clarity, thoroughness, and precision, etc.). Each
reported outcome was then evaluated and given an individual
grade depending on the quality of reporting and methodo-
logical issues specific to that outcome. However, the quality
grade of an individual outcome could not exceed the quality
grade for the overall study.
Grading the Quality of Evidence and the Strength of
Guideline Recommendations
A structured approach, based on the GRADE approach,
723–725
was used to grade the quality of the overall evidence and the
strength of recommendations for each topic. This grading
scheme-with two levels for the strength of a recommendation
together with four levels of grading for the quality of the
evidence, as well as the option of an ungraded statement
for general guidance-was adopted by the KDIGO Board in
December 2008.
Kidney International Supplements (2013) 3, 120–127 121
methods for guideline development

Table 37 | Topics of interest for the management of CKD guideline
Topic Question Population
Treatment,
predictor or
index test
Comparator or
alternative test
or gold standard
Outcome (ranked
by importance) Study design
Inclusion
criteria
Methodological
approach
No. of reports
or articles
yielded
Non-Treatment
Prediction
equations for
GFR
How do SCr- or
SCysC-based prediction
equations perform
compared to gold
standard measurement
of GFR?
Patients in steady state
with or without CKD
and other special
populations (DM, HTN,
KTR or donors, various
age groups, races, or
ethnic groups)
GFR prediction based
on SCr or SCysC must
be directly traceable to
(not just indirectly
calibrated to) IDMS
standards
Gold standard measure
of GFR (e.g.,
iothalamate, EDTA,
iohexal, inulin clearance)
Not 24-hr urine
collection
Bias precision,
accuracy expressed
as P
30
w
Cross-sectional or
diagnostic test
study
Overall N
4100 N
450 for
subgroups
SR
Equation must be
validated in an external
dataset
15
Prediction
equations for risk of
kidney failure
What prediction
equations exist to
predict kidney failure?
GFR o60 ml/min/
1.73 m
2
and/or
kidney transplant
Candidate predictors
had to include at least
age, sex, SCr, and DM
status
NA ESRD,
mortality
Longtudinal cohort
study
Equation
must be
validated in
an external
dataset
N 4100
Studies identified by
WG
2
Gadolinium
exposure and
nephrogenic
fibrosing
dermopathy
What is the incidence
of NFD after exposure
to gadolinium?
GFR o60 ml/min/
1.73 m
2
and/or
kidney transplant
Gadolinium exposure No gadolinium
exposure
Nephrogenic
fibrosing
dermopathy
SR of case–control
and cohort studies
None Search for SRs 1
AKI and phosphate-
containing bowel
preparations
What is the incidence
of AKI after taking a
phosphate-containing
bowel preparation?
Any GFR category;
individuals without
CKD
Exposure to phosphate-
containing bowel
preparation
No exposure to
phosphate-containing
bowel preparation
Categorical kidney
outcomes
Narrative review of
case–control
studies;
observational
cohort studies
None Search for recent
guidelines, SRs, RCTs,
observational studies, or
narrative reviews
2
Treatment
Treatment with
bicarbonate
Does treatment with
bicarbonate in CKD
improve clinical
outcomes?
GFR o60 ml/min/
1.73 m
2
and/or kidney
transplant
Bicarbonate or citrate No bicarbonate or
citrate
ESRD, categorical or
continuous kidney
function; QOL and
BP control as
adverse events
RCT N 4100 Review of Cochrane SRs
plus systematic search
for RCTs published after
search dates in most
recent Cochrane SR
1SR+1RCT
Treatment with
allopurinol
Does treatment with
allopurinol in CKD
improve clinical
outcomes?
GFR o60 ml/min/
1.73 m
2
and/or kidney
transplant with or
without hyperuricemia
Allopurinol No allopurinol Mortality,
cardiovascular
events, ESRD, CKD
progression, gout
attacks; QOL and BP
control as adverse
events; proteinuria
RCT N 4100 SR 1
Timing of initiation
of RRT in CKD
When should dialysis be
started: early or late?
GFR o30 ml/min/
1.73 m
2
Early start of dialysis Late start of dialysis Mortality, ESRD RCT N 4100 Review of studies
identified by WG
1
Protein restriction Should patients with
CKD be on a protein-
restricted diet?
GFR o60 ml/min/
1.73 m
2
and/or
kidney transplant
Reduced protein intake Usual protein intake ESRD, mortality,
categorical or
continuous kidney
function
RCT N 4100 Review of Cochrane SRs
plus systematic search
for RCTs published after
search dates in most
recent Cochrane SR
3 SR+ 1 RCT
Abbreviations: AKI, acute kidney injury; BP, blood pressure; CKD, chronic kidney disease; cys C, cystatin C; DM, diabetes mellitus; EDTA, ethylenediamine tetraacetic acid; ESRD, end-stage renal disease; GFR, glomerular filtration rate;
HTN, hypertension; IDMS, isotope dilution mass spectrometry; KTR, kidney-transplant recipient; NA, not applicable; NFD, nephrogenic fibrosing dermopathy; QOL, quality of life; RCT, randomized controlled trial; RRT renal-
replacement therapy; SCr, serum creatinine; SCysC, serum cystatin C; SR, systematic review; WG, Work Group.
w
Accuracy specifically measured as the percentage of GFR estimates within 30% (P
30
) of the measured GFR. Higher P
30
values reflect greater accuracy; values are decreased by both greater bias and worse precision.
122 Kidney International Supplements (2013) 3, 120–127
methods for guideline development

The strength of a recommendation indicates the extent to
which one can be confident that adherence to the
recommendation will do more good than harm. The quality
of a body of evidence refers to the extent to which our
confidence in an estimate of effect is sufficient to support a
particular recommendation.
724
The process of transparently
grading ev idence and recommendations for treatment topics
is described below in further detail. However, the approach
had to be adapted for the main topics of the KDIGO CKD
guideline because they were not treatment-related topics.
Grading the Quality of Evidence for Each Outcome Across
Studies
Following the GRADE approach, the quality of a body of
evidence pertaining to a particular outcome of interest was
initially categorized on the basis of study design (Table 40).
For questions of interventions, the initi al quality grade was
high if the body of evidence consisted of RCTs, low if it
consisted of observational studies, and very low if it consisted
of studies of other designs. For questions of interventions, the
Work Group decided to use only RCTs. The grade for the
quality of evidence for each intervention–outcome pair was
then lowered if there were serious limitations to the
methodological quality of the aggregate of studies, if there
was thought to be a high likelihood of bias, if there were
important inconsistencies in the results across studies, if there
was uncertainty about the directness of evidence (including
limited applicability of the findings to the population of
interest), if the data were sparse (for example if there was
only one study or if the results include just a few events or
observations and were uninformative) or imprecise (for
example the CI spans a range greater than 1 or confidence
limits are o0.5 to 42.0). The final grade for the quality of
the evidence for an intervention–outcome pair was then
assigned as hig h, moderate, low, or very low (Table 40).
Grading the Overall Quality of Evidence
The quality of the overall body of evidence was then
determined on the basis of the quality grades for all
Table 38 | Literature yield of primary articles for all topics
Topic
Systematic
review
performed Search dates Search terms
Articles
retrieved
Articles
with full
text
screened
Articles
used in
tables
Non-treatment topics
Prediction equations for eGFR Y MEDLINE search from 2006 until
2011, with an update in June 2011
‘‘kidney,’’ and ‘‘prediction,’’
‘‘equation,’’ ‘‘formula,’’ or
‘‘regression analysis’’
2921 86 15
Prediction equations for risk of
kidney failure or death
N nd nd 10* 10 2
Risk from gadolinium for
nephrogenic fibrosing
dermopathy
Y MEDLINE search from 1996 until
2011 was run in March 2011
‘‘kidney,’’ and ‘‘gadolinium,’’
‘‘nephrogenic fibrosing
dermopathy,’’ or ‘‘nephrogenic
systemic fibrosis,’’ and systematic
review
1
w
00
Risk for nephropathy from
phosphate-containing bowel
preparations
N MEDLINE search for recent (2008-
2011) narrative reviews was run in
March 2011
‘‘Phosphate nephropathy’’ 2 2 2
Treatment topics
Treatment with bicarbonate Y MEDLINE search from 2005 until
2011, with an update in May 2011
‘‘randomized controlled trial,’’ and
‘‘kidney,’’ or ‘‘metabolic acidosis,’’
and ‘‘bicarbonate’’
3981 6 1
Treatment with allopurinol Y MEDLINE search from 1996 until
2011, with an update in May 2011
‘‘randomized controlled trial,’’ and
‘‘kidney,’’ and ‘‘allopurinol’’
393 4 1
Earlier versus later initiation of
RRT in CKD
Nnd nd 8
z
81
Treatment with protein
restriction
Y Three Cochrane systematic reviews
were identified and used instead of
a de novo systematic review.
MEDLINE search from 1996-2011,
with an update in March 2011
‘‘randomized controlled trial,’’ and
‘‘kidney,’’ and ‘‘diet, protein-
restricted’’
561 1 1
Abbreviations: CKD, chronic kidney disease; eGFR, estimated GFR; nd, not done; RRT, renal replacement therapy.
*References were provided by Work Group members.
w
Systematic review provided to Work Group members.
z
References were provided by Work Group members.
Table 39 | Classification of study quality
Good
quality
Low risk of bias and no obvious reporting errors, complete
reporting of data. Must be prospective. If study of
intervention, must be randomized controlled study.
Fair
quality
Moderate risk of bias, but problems with study or paper are
unlikely to cause major bias. If study of intervention, must be
prospective.
Poor
quality
High risk of bias or cannot exclude possible significant biases.
Poor methods, incomplete data, reporting errors. Prospective
or retrospective.
Kidney International Supplements (2013) 3, 120–127 123
methods for guideline development

outcomes of interest, taking into account explicit judgments
about the relative importance of each outcome. The resulting
four final categories for the quality of overall evidence were
A, B, C, and D (Table 41).
Assessment of the Net Health Benefit Across All Important
Clinical Outcomes
The net health benefit was determined on the basis of the
anticipated balance of benefits and harms across all clinically
important outcomes (Table 42). The assessment of net
benefit also involved the judgment of the Work Group and
the ERT.
Grading the Strength of the Recommendations
The strength of a recommendation is graded as level 1 or
level 2. Table 43 shows the KDIGO nomenclature for grading
the strength of a recommendation and the implications of
each level for patients, clinicians, and policy makers. Recom-
mendations can be for or against doing something. Table 44
shows that the strength of a recommendation is determined
not only by the quality of the evidence, but also by other,
often complex judgments regarding the size of the net
medical benefit, values and preferences, and costs. Formal
decision analyses including cost analysis were not conducted.
Ungraded Statements
This category was designed to allow the Work Group to issue
general advice. Typically an ungraded statement meets the
following criteria: it provides guidance that is based on
common sense, it provides reminders of the obvious, and it is
not sufficiently specific to allow for application of evidence to
the issue and therefore it is not based on systematic evidence
review. The GRADE system is best suited to evaluate evidence
on comparative effectiveness. Some of our most important
guideline topics involve diagnosis and staging within CKD ,
about which the Work Group chose to provide ungraded
statements. These statements are indirectly supported by
evidence on risk relationships and are the consensus of the
Work Group. Thus, we believe that ungraded statements
should not be viewed as weaker than graded recommendations.
Table 40 | GRADE system for grading quality of evidence
Step 1: Starting grade for
quality of evidence based
on study design Step 2: Reduce grade Step 3: Raise grade Final grade for quality of evidence and definition
Randomized trials = High Study quality
-1 level if serious limitations
-2 levels if very serious limitations
Consistency
-1 level if important inconsistency
Directness
-1 level if some uncertainty
-2 levels if major uncertainty
Other:
-1 level if sparse or imprecise data
c
-1 level if high probability of
reporting bias
Strength of association
+1 level is strong
a
, no plausible
confounders
+2 levels if very strong
b
, no major
threats to validity
Other
+1 level if evidence of a
dose-response gradient
+1 level if all residual plausible
confounders would have
reduced the observed effect
High = Further research is unlikely to change confidence
in the estimate of the effect
Observational study = Low
Moderate = Further research is likely to have an
important impact on confidence in the estimate of effect,
and may change the estimate
Low = Further research is very likely to have an important
impact on confidence in the estimate and may change
the estimate
Any other evidence = Very
low
Very low = Any estimate of effect is very uncertain
Abbreviation: GRADE, Grading of Recommendations Assessment, Development, and Evaluation.
a
Strong evidence of association is defined as ‘significant relative risk of 42(o0.5)’ based on consistent evidence from two or more observational studies, with no plausible
confounders.
b
Very strong evidence of association is defined as ‘significant relative risk of 45(o0.2)’ based on direct evidence with no major threats to validity.
c
Sparse if there was only one study or if the results include just a few events or observations and were uninformative. Imprecise if the confidence interval spans a range
greater than 1 or confidence limits are o0.5 to 42.0
Adapted by permission from Macmillan Publishers Ltd: Kidney International. Uhlig K, Macleod A, Craig J et al.
725
Grading evidence and recommendations for clinical practice
guidelines in nephrology. A position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 2006; 70: 2058–2065; accessed http://www.nature.com/
ki/journal/v70/n12/pdf/5001875a.pdf
Table 41 | Final grade for overall quality of evidence
Grade
Quality of
evidence Meaning
A High We are confident that the true effect lies close to that of
the estimate of the effect.
B Moderate The true effect is likely to be close to the estimate of the
effect, but there is a possibility that it is substantially
different.
C Low The true effect may be substantially different from the
estimate of the effect.
D Very low The estimate of effect is very uncertain, and often will be
far from the truth.
Table 42 | Balance of benefits and harm
When there was evidence to determine the balance of medical benefits
and harm of an intervention to a patient, conclusions were categorized as
follows:
K For statistically significant benefit/harm, report as ‘benefit [or harm]
of drug X’.
K For non-statistically significant benefit/harm, report as ‘possible
benefit [or harm] of drug X’.
K In instances where studies are inconsistent, report as ‘possible
benefit [or harm] of drug X’.
K ‘No difference’ can only be reported if a study is not imprecise.
K ‘Insufficient evidence’ is reported if imprecision is a factor.
124
Kidney International Supplements (2013) 3, 120–127
methods for guideline development

Table 43 | KDIGO nomenclature and description for grading recommendations
Implications
Grade* Patients Clinicians Policy
Level 1
‘We recommend’
Most people in your situation would
want the recommended course of action
and only a small proportion would not.
Most patients should receive the
recommended course of action.
The recommendation can be evaluated as
a candidate for developing a policy or a
performance measure.
Level 2
‘We suggest’
The majority of people in your situation
would want the recommended course of
action, but many would not.
Different choices will be appropriate for
different patients. Each patient needs help
to arrive at a management decision
consistent with her or his values and
preferences.
The recommendation is likely to require
substantial debate and involvement of
stakeholders before policy can be
determined.
Abbreviation: KDIGO, Kidney Disease: Improving Global Outcomes.
*The additional category ‘‘Not Graded’’ was used, typically, to provide guidance based on common sense or where the topic does not allow adequate application of evidence.
The most common examples include recommendations regarding monitoring intervals, counseling, and referral to other clinical specialists. The ungraded recommendations
are generally written as declarative statements, but are not meant to be interpreted as being stronger recommendations than Level 1 or 2 recommendations.
Table 44 | Determinants of strength of recommendation
Factor Comment
Balance between desirable and
undesirable effects
The larger the difference between the desirable and undesirable effects, the more likely a strong recommendation
is warranted. The narrower the gradient, the more likely a weak recommendation is warranted.
Quality of the evidence The higher the quality of evidence, the more likely a strong recommendation is warranted.
Values and preferences The more variability in values and preferences, or more uncertainty in values and preferences, the more likely a
weak recommendation is warranted.
Costs (resource allocation) The higher the costs of an intervention—that is, the more resources consumed—the less likely a strong
recommendation is warranted.
Reproduced from Going from evidence to recommendations. Guyatt GH, Oxman AD, Kunz R, et al.
724
BMJ 336: 1049-1051, 2008 with permission from BMJ Publishing Group
Ltd.; accessed http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2376019/pdf/bmj-336-7652-analysis-01049.pdf
Table 45 | The Conference on Guideline Standardization
727
checklist for reporting clinical practice guidelines
Topic Description Discussed in KDIGO CKD Guideline
1. Overview material Provide a structured abstract that includes the
guideline’s release date, status (original, revised,
updated), and print and electronic sources.
Abstract and Methods for Guideline Development.
2. Focus Describe the primary disease/condition and
intervention/service/technology that the
guideline addresses. Indicate any alternative
preventative, diagnostic, or therapeutic
interventions that were considered during
development.
Evaluation and management of adults and children with CKD.
3. Goal Describe the goal that following the guideline is
expected to achieve, including the rationale for
development of a guideline on this topic.
This clinical practice guideline is intended to assist the practitioner
caring for patients with CKD and to prevent deaths, cardiovascular
disease events and progression to kidney failure while optimizing
patients’ quality of life.
4. User/setting Describe the intended users of the guideline (e.g.,
provider types, patients) and the settings in which
the guideline is intended to be used.
Providers: Nephrologists (adult and pediatric), Dialysis providers
(including nurses), Internists, and Pediatricians.
Patients: Adult and pediatric individuals at risk for or with CKD.
Policy Makers: Those in related health fields.
5. Target population Describe the patient population eligible for
guideline recommendations and list any exclusion
criteria.
Individuals at risk for or with CKD.
6. Developer Identify the organization(s) responsible for
guideline development and the names/
credentials/potential conflicts of interest of
individuals involved in the guideline’s
development.
Organization: KDIGO.
Refer to Biographic and Disclosure Information section.
7. Funding
source/sponsor
Identify the funding source/sponsor and describe
its role in developing and/or reporting the
guideline. Disclose potential conflict of interest.
KDIGO is supported by the following consortium of sponsors:
Abbott, Amgen, Bayer Schering Pharma, Belo Foundation, Bristol-
Myers Squibb, Chugai Pharmaceutical, Coca-Cola Company, Dole
Food Company, Fresenius Medical Care, Genzyme, Hoffmann-
LaRoche, JC Penney, Kyowa Hakko Kirin, NATCO—The Organization
for Transplant Professionals, NKF-Board of Directors, Novartis,
Pharmacosmos, PUMC Pharmaceutical, Robert and Jane Cizik
Kidney International Supplements (2013) 3, 120–127 125
methods for guideline development

Table 45 | Continued
Topic Description Discussed in KDIGO CKD Guideline
Foundation, Shire, Takeda Pharmaceutical, Transwestern
Commercial Services, Vifor Pharma, and Wyeth.
No funding is accepted for the development or reporting of specific
guidelines.
All stakeholders could participate in open review.
8. Evidence collection Describe the methods used to search the scientific
literature, including the range of dates and
databases searched, and criteria applied to filter
the retrieved evidence.
Screening criteria are outlined in the methods chapter. The search
was updated through June 2011 and supplemented by articles
identified by Work Group members through November 2012.
We also searched for pertinent existing guidelines and systematic
reviews.
9. Recommendation
grading criteria
Describe the criteria used to rate the quality of
evidence that supports the recommendations and
the system for describing the strength of the
recommendations. Recommendation strength
communicates the importance of adherence to a
recommendation and is based on both the quality
of the evidence and the magnitude of anticipated
benefits and harms.
Quality of individual studies was graded in a three-tiered grading
system (see Table 39). Quality of evidence (Table 40) was graded
following the GRADE approach. Strength of the recommendation
was graded in a two-level grading system which was adapted from
GRADE for KDIGO with the quality of overall evidence graded on a
four-tiered system (Tables 41 and 43).
The Work Group could provide general guidance in ungraded
statements.
10. Method for
synthesizing
evidence
Describe how evidence was used to create
recommendations, e.g., evidence tables, meta-
analysis, decision analysis.
For systematic review topics, summary tables and evidence profiles
were generated.
For recommendations on treatment interventions, the steps
outlined by GRADE were followed.
11. Prerelease review Describe how the guideline developer reviewed
and/or tested the guidelines prior to release.
The guideline had undergone internal review by the KDIGO Board of
Directors in January 2012 and external review in May 2012. Public
review comments were compiled and fed back to the Work Group,
which considered comments in its revision of the guideline.
12. Update plan State whether or not there is a plan to update the
guideline and, if applicable, expiration date for
this version of the guideline.
There is no date set for updating this entire guideline. The need for
updating of the guideline will depend on the publication of new
evidence that would change the quality of the evidence or the
estimates for the benefits and harms. Results from registered
ongoing studies and other publications will be reviewed periodically
to evaluate their potential to impact on the recommendations in
this guideline. Specific sections may be updated separately from the
entire guideline within the next 3–5 years depending on the
evidence base.
13. Definitions Define unfamiliar terms and those critical to
correct application of the guideline that might be
subject to misinterpretation.
Acronyms and Abbreviations.
14. Recommendations
and rationale
State the recommended action precisely and the
specific circumstances under which to perform it.
Justify each recommendation by describing the
linkage between the recommendation and its
supporting evidence. Indicate the quality of
evidence and the recommendation strength,
based on the criteria described in Topic 9.
Each guideline chapter contains recommendations for evaluation
and management of CKD patients. Each recommendation builds on
a supporting rationale with evidence tables if available. The strength
of the recommendation and the quality of evidence are provided in
parenthesis within each recommendation.
15. Potential benefits
and harm
Describe anticipated benefits and potential risks
associated with implementation of guideline
recommendations.
The benefits and harm for each comparison of interventions is
provided in summary tables and summarized in evidence profiles.
The estimated balance between potential benefits and harm was
considered when formulating the recommendations.
16. Patient preferences Describe the role of patient preferences when a
recommendation involves a substantial element
of personal choice or values.
Many recommendations are ungraded which indicates a greater
need to help each patient arrive at a management decision
consistent with her or his values and preferences.
17. Algorithm Provide (when appropriate) a graphical
description of the stages and decisions in clinical
care described by the guideline.
Algorithm for proteinuria/albuminuria testing in Chapter 1.
18. Implementation
considerations
Describe anticipated barriers to application of the
recommendations. Provide reference to any
auxiliary documents for providers or patients that
are intended to facilitate implementation. Suggest
review criteria for measuring changes in care
when the guideline is implemented.
These recommendations are global and the Work Group
acknowledges the importance of local application. Review criteria
were not suggested because implementation with prioritization and
development of review criteria must proceed locally. Furthermore,
most recommendations are discretionary, requiring substantial
discussion among stakeholders before they can be considered for
adoptions as review criteria.
Suggestions were provided for future research.
Abbreviations: CKD, chronic kidney disease; COGS, Conference on Guideline Standardization; GRADE, Grading of Recommendations Assessment, Development, and
Evaluation; KDIGO, Kidney Disease: Improving Global Outcomes; NKF, National Kidney Foundation.
Adapted with permission from Shiffman RN, Shekelle P, Overhage JM, et al.
727
Standardized reporting of clinical practice guide lines: a proposal from the Conference on
Guideline Standardization. Ann Intern Med 2003; 139(6): 493-498 ; accessed http://annals.org/data/Journals/AIM/20049/0000605-200309160-00013.pdf
126 Kidney International Supplements (2013) 3, 120–127
methods for guideline development

The Work Group took on the primary role of writing the
recommendations and rationale statements and retained final
responsibility for the content of the guideline statements and
the accompanying narrative. The ERT reviewed draft
recommendations and grades for consistency with the
conclusions of the evidence review.
Format for Guideline Recommendations
Each chapter contains one or more specific recommen-
dations. Within each recommendation, the strength of
recommendation is indicated as level 1 or level 2 and the
quality of the supporting evidence is shown as A, B, C or D.
The recommendation statements and grades are followed by
the rationale and clarification of the wording of the
statement, a brief background with relevant definitions of
terms, and then a chain of logic which summarizes the key
points of the ev idence base and the judgments supporting the
recommendation. Some sections also contain research
recommendations in variable degrees of detail, suggesting
future research to resolve current uncertainties.
Limitations of Approach
Although the literature searches were intended to be
comprehensive, they were not exhaustive. MEDLINE was
the only database searched. Hand searches of journals were
not performed, and review articles and textbook chapters
were not systematically searched. However, important studies
known to domai n experts that were missed by the electronic
literature searches were added to the retrieved articles and
reviewed by the Work Group.
Summary of the Review Process
Several tools and checklists have been developed to assess the
quality of the methodological process for systematic r eview and
guideline development. These include the Appraisal of Guidelines
for R esearch and Evaluation (AGREE) criteria,
726
the Conference
on Guideline Standardization (COGS) checklist,
727
and the
Institut e of Medicine ’s rec ent Standards for Systematic Reviews
728
and Clinical Practice Guidelines W e Can Trust.
729
Table 45 and
Appendix 1 demonstrate the level of concurrence to the COGS
criteria and the Institute of Medicine standards, respectively.
SUPPLEMENTARY MATERIAL
Supplemental Table 1: Search strategy.
Appendix 1: Concurrence with Institute of Medicine standards for
systematic reviews and for guidelines.
Supplementary material is linked to the online version of the paper at
http://www.kdigo.org/clinical_practice_guidelines/ckd.php
Kidney International Supplements (2013) 3, 120–127 127
methods for guideline development

Biographic and Disclosure Information
Kidney International Supplements (2013) 3, 128–133; doi:10.1038/kisup.2012.70
Adeera Levin, MD, FRCPC (Work Group Co-Chair),is
Professor of Medicine, Division Head of Nephrology at the
University of British Columbia, and Consultant Nephrologist
at Providence Health Care/St Paul’s Hospital, in Vancouver
Canada.
Dr Levin is the Executive Director of the BC Renal
Agency, which oversees the care, planning, and budgets for
kidney services in the province of British Columbia. In this
capacity, she has leveraged her epidemiological training,
clinical knowledge, and health outcomes research to develop
an evidence-based transparent system which enhances the
care of patients across the continuum of care (from
identification of CKD to dialysis, transplant, and death).
She is active in international activities across the spectrum
of kidney activities including her appointment as Secretary
General of the International Society of Nephrology (ISN),
founding executive committee member of KDIGO, and as co-
Chair of the Declaration of Istanbul Custodian group
(DICG). Dr Levin has a long-standing clinical and research
interest in CKD and CVD, and is Founding Director of the
Kidney Function Clinic at St Paul’s Hospital.
As an active researcher and mentor, she is involved in a
national curriculum development for clinician scientists
interested in kidney disease (KRESCENT), and mentors
students, residents, and nephrology fellows in Vancouver as
well. She has over 210 peer-reviewed publications, numerous
book chapters, and is co-editor of a textbook Chronic Kidney
Disease: A Practical Guide to Understanding and Management.
Her major research areas of interest include non-traditional
risk factors for CVD in CKD patients, with particular focus
on anemia, phosphate and vitamin D, and progression of
CKD variability.
Advisor/Consultant: Merck; Otsuka
Grant/Research Support: Abbott; Amgen; Canadian
Institutes of Health Research; Genzyme, Kidney Foundation
of Canada; Merck; Ortho
Paul E Stevens, MB, FRCP (Work Group Co-Chair), is
Consultant Nephrologist and Associate Medical Director,
East Kent Hospitals University NHS Foundation Trust, Kent
and Canterbury Hospital in the UK. He is a member of the
Department of Health’s Renal Advisory Group and Renal
Pathology Group and immediate past-President of the British
Renal Society. Dr Stevens established the Kent Kidney Care
Centre, one of the largest general hospital renal units in the
UK and served as a clinical advisor on NICE publications
Anaemia Management in People with Chronic Kidney Disease
and Chronic Kidney Disease: National Clinical Guideline for
Early Identification and Management in Adults in Primary and
Secondary Care. He is also Chair of NICE Quality Standards
in Chronic Kidney Disease.
His main interests include the epidemiology of CKD and
AKI, prospective evaluation of a new method of assessing
adequacy in peritoneal dialysis, evaluation of anemia in
unreferred patients with CKD and in patients with diabetes,
collaborative research with the elderly CKD care team, and
the development of information technology-based solutions
in the referral and follow-up of patients with kidney disease.
Over the last several years, his program has also developed a
clinical decision support system to aid management of
CKD in primary care and this system is currently running live
in a population of over 300,000 primary care patients.
Dr Stevens has authored over 90 publications in peer-
reviewed journals and textbooks, and has given over 100
presentations and 65 guest lectures in national and
international society meetings.
Dr Stevens reported no relevant financial relationships
Rudy W Bilous, MD, is Professor of Clinical Medicine at
Newcastle University and Honorary Consultant Physician
at the James Cook University Hospital, Middlesbrough, UK.
He studied medicine at Guy’s Hospital and completed his
clinical training at various hospitals in London. He was an
National Institutes of Health (NIH) Fellow at the Unit for
Metabolic Medicine at Guy’s from 1980-1982 working for
Professor Harry Keen and with Drs John Pickup and
Giancarlo Viberti examining the effects of metabolic control
on microvascular complications. From 1985-1987 he was
Juvenile Diabetes Research Foundation fellow at the Uni-
versity of Minnesota working for Drs Mike Steffes and Mike
Mauer. He was awarded his MD from the University of
London in 1987 and elected Fellow of the Royal College of
Physicians of London in 1991. He served as Secretary and
Chair of the Specialty Advisory Committee for Endocrinol-
ogy and Diabetes and Chairman of the Professional section of
Diabetes UK. He is currently Chairman of the workgroup
reviewing the NICE guideline for Diabetes in Pregnancy
and President of the European Diabetic Nephropathy
Study Group of the European Association for the Study of
Diabetes.
Advisor/Consultant: Abbott; Boehringer Ingelheim; Novartis;
Roche
Speaker: Boehringer Ingelheim; Novo Nordisk; Roche
biographic and disclosure information http://www.kidney-international.org
& 2013 KDIGO
128
Kidney International Supplements (2013) 3, 128–133

Josef Coresh, MD, PhD, MHS, is Professor, Department of
Epidemiology, Medicine, and Biostatistics at Johns Hopkins
Bloomberg School of Public Health and School of Medicine
where he also received his medical degree and PhD in
epidemiology, and a master’s in biostatistics. He is also
presently the Director of the George W Comstock Center for
Public Health Research and Prevention, and the Cardio-
vascular Epidemiology Program and Training Grant at
Johns Hopkins Bloomberg School of Public Health.
Dr Coresh’s primary research interest has been the
investigation of risk factors for CVD in CKD and their role
in mediating the excess risk among patients with various
degrees of CKD severity. With the use of innovative
epidemiologic methods and statistical analyses, his recent
work uncovered several novel genetic susceptibility loci for
gout as well as a susceptibility locus (MYH9/APOL1) which
accounts for much of the excess risk of non-diabetic kidney
disease among African-Americans. Since 2000, he has focused
his attention on enhancing CKD classification, management
and prognostication by improving the estimation of kidney
function. In 2002, he served as Co-Chair on the KDOQI
Clinical Practice Guidelines for CKD which produced the
CKD definition and classification system that have been
adopted widely today. More recently, his research led to the
development of the 2009 CKD-EPI equation for estimating
kidney function which is rapidly become the new standard
for GFR estimation globally. In 2010, he became Steering
Committee Chair of Chronic Kidney Disease Prognosis
Consortium (CKD-PC) which analyzes data from two
million study participants in order to provide better
estimation on CKD prognosis. Dr Coresh has authored close
to 300 publications and has mentored over 60 students. In
recognition of his contributions, he was awarded the NKF’s
Garabed Eknoyan Award (2010), American Heart Associa-
tion’s 2010 Epidemiology and Prevention Mentoring Award
and served as plenary speaker at the American Association
for Clinical Chemistry 2011 Annual Meeting.
Advisor/Consultant: Merck
Grant/Research Support: Amgen; National Institutes of
Health; National Kidney Foundation
Speaker: Abbott; Amgen
Angel LM de Francisco, MD, PhD, is Head of Dialysis Unit
and Nephrology Section, Hospital Universitario Valdecilla,
Santander (Spain), and Full Professor of Nephrology at
Faculty of Medicine in Cantabria University. He completed
his medical residency and nephrology fellowship at Hospital
Puerta de Hierro in Madrid, and received his Doctor of
Medicine degree from University of Cantabria (Spain). Prof
de Francisco is an active member of several professional
societies, an Executive member of the KDIGO Board, and
was President of the Spanish Society of Nephrology from
2002 to 2008. He has been a frequently invited lecturer,
speaking extensively on advancements in treatment of
chronic kidney diseases and comorbid conditions such as
metabolic bone disease, anemia, dialysis, and kidney
transplantation. Prof de Francisco has authored or coau-
thored over 150 publications, including original peer-
reviewed articles and book chapters. His primary focus has
been on improving outcomes in patients receiving hemodia-
lysis, and on issues surrounding organ transplantation and
the impact of treatment guidelines on clinical practice.
Advisor/Consultant: Amgen
Speaker: Abbott; Fresenius
Paul E de Jong, MD, PhD, was Professor of Nephrology and
Head, Division of Nephrology of the Groningen University
Medical Center in Groningen, the Netherlands from 1992
to 2011. His main research interest is prevention of CKD.
From 1980 to 2000, he focused most of his attention on the
mechanisms of urinary protein loss in CKD patients and
options to lower proteinuria with ACE-I. His studies had
indeed shown that such treatment prevented ESRD progres-
sion. In 1996, he initiated the PREVEND study which
consisted of a large cohort of 8592 subjects followed for the
development of kidney disease and CVD. This prospective
longitudinal study demonstrated the impact of albuminuria,
even in the ranges of microalbuminuria and below, in
predicting future cardiovascular and renal events. As former
Director of the Groningen Institute of Kidney Diseases, his
research program encompasses the studying of underlying
susceptibility to kidney damage in health subjects and the
identification of common pathophysiological pathways
between CKD and CVD. Currently, he serves on the board
of KDIGO, CKD-PC, and European Kidney Health Alliance.
In 2010, he was the recipient of International Distinguished
Medal from the US National Kidney Foundation and was
awarded the Officer in the Order of Orange-Nassau for his
lifetime achievement by Her Majesty Queen Beatrix of the
Netherlands in 2011.
Dr de Jong reported no relevant finan cial relationships
Kathryn E Griffith, BM, BS, MSc, MRCP, MRCGP,isa
general practitioner affiliated with The University Health
Centre, York University and York Hospital Foundation Trust.
Dr Griffith has a long-standing interest in the relationship
between CVD and renal disease from a primary care
physician’s perspective. In this vein, she has served as a panel
member for the Royal College of Physicians (RCP)
Edinburgh Consensus Conference on CKD and was involved
in the development of the 18-week Commissioning Pathway
to CKD with the Department of Health. She is presently a
member of the Renal Advisory Group at the Department of
Health and a member of both UK Renal Association and
British Renal Society. Dr Griffith has also been involved in
the education of general practitioners and practice nurses,
and most recently she has lectured extensively on various
topics such as atrial fibrillation, heart failure, coronary heart
Kidney International Supplements (2013) 3, 128–133 129
biographic and disclosure information

disease, hypertension, and stroke. For the past two years, she
has worked with Yorkshire Cardiac and Stroke Networks
on a guide to stroke prevention in atrial fibrillation, and
contributed to the RCP Edinburgh Consensus on atrial
fibrillation management and European Society of Cardio-
logy’s clinical practice guideline on management of
dyslipidemias.
Advisor/Consultant: Boehringer Ingelheim
Honoraria: Pfizer
Travel Stipend: Merck Sharp & Dohme
Grant/Research Support: Roche
Speaker: Boehringer Ingelheim
Brenda R Hemmelgarn, MD, PhD, FRCP(C), is Associate
Professor in the Departments of Medicine and Community
Health Sciences, University of Calgary, Calgary, Alberta. She
is a clinician scientist with clinical training in nephrology
as well as a PhD in Epidemiology and Biostatistics.
Dr Hemmelgarn’s research interests are in the study of
chronic medical conditions including CKD, diabetes, hyper-
tension and CVD, as well as health services research aimed
at improving management of these chronic conditions.
Dr Hemmelgarn holds provincial and national salary awards
and operating grants, and is the recipient of the Roy and
Vi Baay Chair in Kidney Research. She is also actively
involved in Clinical Practice Guideline development both
nationally through the Canadian Society of Nephrology
and internationally through KDIGO, and has over 180
peer-reviewed publications.
Grant/Research Support: Roche
Kunitoshi Iseki, MD, is currently affiliated with
University Hospital of the Ryukyus in Okinawa, Japan.
Dr Iseki received his medical training at Kyushu University
in Fukuoka, Japan and completed his fellowship at University
of Southern California under Professors Shaul Massry
and Vito Campese. He was Senior Assistant Professor of
Medicine at Ryukyu University Hospital and later became
its Director of Dialysis Unit. Dr Iseki is a member of
numerous professional organizations including the Japanese
Society of Nephrology and Japanese Society for Dialysis
Therapy. He is also an organizing member for Asian Forum
of Chronic Kidney Disease Initiatives (AFCKDI) since 2007
and an ISN-COMGAN Educational Ambassador since
2009. Dr. Iseki has also participated and contributed data
to the CKD-PC whose collaborative meta-analyses were
conducted to study CKD prognosis. He has previously served
as Editorial board member for American Journal Kidney
Disease and is presently an editorial board member of
Nephrology, Nephron Clinical Practice and Nephrology Dialysis
Transplantation.
Dr Iseki reported no relevant financial relationships
Edmund J Lamb, PhD, FRCPath, is a Consultant Clinical
Scientist and Head of Clinical Biochemistry at East Kent
Hospitals University NHS Foundation Trust, Canterbury,
Kent, UK. He has a special interest in kidney disease and
undertook his PhD in the Renal Research Laboratory at
St Bartholomew’s Hospital, London. He has 30 years
experience as a clinical biochemist and his research
interests relate to the use of biochemical markers to diag-
nose and monitor kidney disease, including the assess-
ment of kidney function using eGFR, the evaluation of
renal bone disease, the use and role of cystatin C, and the
significance and use of cardiac biomarkers in kidney
disease. Dr Lamb has authored or co-authored more than
60 full peer-reviewed papers in this area. He is a member
of the UK Kidney Research Consortium Clinical Study
Group on CKD-MBD and he is the lead investigator on two
National Institute for Health Research-Research for Patient
Benefit funded studies investigating aspects of kidney
function testing. He has been involved in national and
international committees and guidelines (e.g., Royal College
of Physicians UK CKD Guidelines Group, NICE CKD
Guideline Development Group, KDIGO Chronic Kidney
Disease Guideline Work Group) and was recently involved
in the Department of Health initiative to roll-out eGFR
across England. He is Editor-in-Chief of Annals of Clinical
Biochemistry.
Grant/Research Support: Siemens
Andrew S Levey, MD, is Dr Gerald J and Dorothy
R Friedman Professor of Medicine at Tufts University School
of Medicine, Chief of the William B Schwartz, MD Division
of Nephrology at Tufts Medical Center, Senior Scientist at the
US Department of Agriculture Human Nutrition Research
Center on Aging at Tufts University, and Professor, Clinical
Research at the Sackler School of Graduate Biomedical
Sciences at Tufts University. His research is mainly in the
areas of epidemiology of CKD and CVD in CKD, controlled
trials to slow the progression of CKD, clinical assessment of
kidney function, assessment and improvement of outcomes
in dialysis and transplantation, and clinical practice guideline
development and implementation. Dr Levey is currently
Program Director for an NIDDK-funded clinical research
training program, ‘‘Clinical Trials, Epidemiology and Out-
comes Research in Nephrology.’’ He is also the Director of the
Tufts Center for Guideline Development and Implementation
and Editor of American Journal of Kidney Disease. Dr Levey
is past Chair of the NKF’s Task Force on Cardiovascular
Disease in Chronic Renal Disease and served on KDOQI
Work Group on Chronic Kidney Disease: Evaluation,
Classification and Stratification, KDOQI Work Group on
Hypertension and Antihypertensive Agents in Chronic
Kidney Disease, and KDIGO Work Group on Acute Kidney
Injury Guideline.
Dr Le vey reported no rele vant financial relationships
130 Kidney International Supplements (2013) 3, 128–133
biographic and disclosure information

Miguel C Riella, MD, PhD, FACP, is a graduate of Federal
University of Parana in Curitiba, Brazil and has undertaken
his post-graduate training in the United States, first in
Internal Medicine Residency at Mount Sinai Hospital in
New York City and then Research Renal Fellowship at the
University of Washington in Seattle, USA under the leader-
ship of Prof Belding H Scribner, pioneer of chronic
hemodialysis. Dr Riella’s main interest has been on dialysis,
particularly peritoneal dialysis and nutrition in uremia. More
recently his research work has concentrated on the relation-
ship of inflammation, uremia, nutrition and cardiovascular
complications, and interventional nephrology. As part of
his new Renal, Diabetes and Hypertension Research Center,
he has recently established the Islet Cell Laboratory at Catholic
University of Parana in Curitiba, Brazil for cell transplant in
diabetic patients. Dr Riella has published several books in
Portuguese and Spanish language in the area of nephrology
and nutrition. His book Principles of Nephrology and Electrolyte
Disturbances (Guanabara Koogan Group Gen, 2010) is on its
5
th
Edition. He has more than 150 publications and has given
more than 600 lectures locally and internationally. He is a past
Board member and executive committee member of the ISN
and is currently the Chairman of the ISN-Interventional
Nephrology Committee. Dr Riella is a member of the editorial
board of several medical journals, including as Associate Editor
of Hemodialysis International and International Editor of
Seminar in Dialysis for Interventional Nephrology. He is also
President and founder of Pro-Renal Foundation of Brazil
(www.pro-renal.org.br), a non-for-profit organization dedi-
cated to patient assistance, CKD prevention and education,
and supporting research in kidney and metabolic diseases. The
Foundation provides free medical care to more than 1800 renal
patients through a multidisciplinary team of physicians, nurses,
psychologists, social workers, and dietitians. The Foundation is
maintained by donations of the community and receives no
public funds. Dr Riella is the past-President of the Interna-
tional Federation of Kidney Foundations (IFKF) 2009-2011.
He is also the co-chairman of World Kidney Day, a joint
project of IFKF and and ISN. He is a member of the Executive
Committee of KDIGO, National Academy of Medicine in
Brazil, and the Academy of Medicine of Parana, Brazil. He has
received many awards such as the International Distinguished
Medal from the US NKF and Honorary Citizenship of the city
of Curitiba and of the State of Parana in Brazil.
Advisor/Consultant: Abbott
Michael G Shlipak, MD, MPH, is Professor of Medicine,
Epidemiology, and Biostatistics at University of California,
San Francisco. Dr Shlipak obtained his MD degree from
Harvard Medical School and his MPH from Harvard School
of Public Health. His research program has been dedicated to
understanding the causes and consequences of age-related
kidney disease and to utilize novel biomarkers for the early
detection of kidney damage and reduced kidney function.
Along with his colleagues from the Cardiovascular Health
Study and Health Aging and Body Composition study,
Professor Shlipak has conducted the pioneering work to
demonstrate with cystatin C the remarkable prognostic
importance of impaired kidney function in elders. He coined
the entity of ‘‘preclinical kidney disease’’ to describe the large
population of elders with elevated cystatin C levels, but
normal creatinine-based estimates of kidney function, who
are at high risk for death and CVD.
The recent work from his research group has been
dedicated to finding early markers of kidney injury in the
urine that will precede any detectable loss in kidney function.
They will study these urine biomarkers in several popula-
tions, including the ambulatory elderly, HIV-infected men
and women, and in population-based studies. The group
believes that these injury markers hold promise both for
understanding how various exposures impact the kidney, and
also to forecast future declines in kidney failure.
Dr Shlipak reported no relevant financial relationships
Haiyan Wang, MD, is Professor of Medicine at Peking
University First Hospital. She is the current President of
Peking University Institute of Nephrology and the Vice
Director, Academic Committee, Peking University Health
Science Center and Member, Academic Degree Committee,
Peking University Health Science Center.
Professor Wang is very active in the academic area of
national and international nephrology and internal medicine.
She was the Vice President of Chinese Medical Association
(1999-2009) and the President of Chinese Society of Internal
Medicine (2000-2008), as well as the President of Chinese
Society of Nephrology (1992-1998). She has also served
as the ISN Councilor member (2004-2008), Executive
Councilor of ISN (2006-2008) and is now Chair, East Asia
committee of ISN GO (2004-present).
Dr Wang reported no relevant financial relationships
Colin T White, MD, FRCPC, completed his medical degree at
the University of Ottawa, Canada and Pediatric Residency at
the University of Western Ontario, Canada. His pediatric
nephrology training took place at the University of Western
Ontario and University of British Columbia. He is a Clinical
Associate Professor in Pediatrics/Nephrology at the University
of British Columbia and has a clinical appointment at BC
Children’s Hospital where in addition to an interest and partici-
pation in the CKD clinic, he is the Director of the Dialysis
Program and is the Fellowship Program Director. Dr White was
a work group member for the 2006 and 2007 Updated KDOQI
Anemia Guidelines, and for the Canadian Society of Nephro-
logy’s Clinical Practice Guidelines for Anemia Management in
2007-2008; he is currently on the 2011-2012 CSN Workgroup
revisiting these guidelines. He is the site Principal Investigator
for the North American multicenter NIH-sponsored CKiDs
trial and has particular research interests in hemodialysis and
peritoneal dialysis as well as CKD management. His particular
Kidney International Supplements (2013) 3, 128–133 131
biographic and disclosure information

areas of research interest include estimation of clearance,
anemia protocol development, and translation of evidence
into functional guidelines. He is active at the undergraduate
medical school level and has coordinated the UBC Renal and
Genitourinary Course for 1st year medical students for nearly a
decade. He is currently the President of the Western Society of
Pediatric Research and an invited member of the Renal Work
Group for the World Health Organization’s ICD-11 Code
Revision.
Advisor/Consultant: Baxter Healthcare
Christopher G Winearls, MB, DPhil, FRCP, graduated in
1973 from University of Cape Town and as a Rhodes Scholar
completed a PhD from University of Oxford in transplant
immunology. Dr Winearls later pursued training in nephro-
logy at the Hammersmith Hospital where he was part of the
team that investigated the first clinical use of erythropoietin.
Dr Winearls was appointed consultant in Oxford in 1988 and
was Clinical Director 1995 to 2009. He has been Secretary
and later Clinical Vice President of The Renal Association
and an Editor of the Oxford Textbook of Clinical Nephrology.
His research interests include myeloma kidney and auto-
somal dominant polycystic kidney disease, and has recently
taken an interest in the new classification of CKD.
Dr Winearls reported no relevant financial relationships
KDIGO CHAIRS
Bertram L Kasiske, MD, is Professor of Medicine at the
University of Minnesota, USA. He received his medical
degree from the University of Iowa and completed his
Internal Medicine residency and fellowship training in
Nephrology at Hennepin County Medical Center where he
is currently Director of Nephrology.
Dr Kasiske is former Deputy Director of the United States
Renal Data System and former Editor-in-Chief of American
Journal of Kidney Diseases. He has served as Secretary/
Treasurer and on the Board of Directors of the American
Society of Transplantation, and on the Organ Procurement
and Transplantation Network/United Network of Organ
Sharing Board of Directors, and the Scientific Advisory
Board of the US NKF. He is currently serving on the Board of
Councilors of the ISN and he is the Principal Investigator for
a National Institutes of Health-sponsored, multicenter study
of long term outcomes after kidney donation. Dr Kasiske is
the Director of the Scientific Registry of Transplant
Recipients and has over 160 scientific publications in major
peer-reviewed journals, and 230 review articles, editorials and
textbook chapters. Dr Kasiske is also a recipient of the US
NKF’s Garabed Eknoyan Award in 2003.
Advisor/Consultant: Litholink
Grant/Research Support: Bristol-Myers Squibb; Merck
(Schering Pl ough)
David C Wheeler, MD, FRCP, holds an academic position in
Nephrology (Reader) at University College London, UK and
is an Honorary Consultant Nephrologist at the Royal Free
Hospital. His research is focused on the cardiovascular
complications of CKD and the role of vascular risk factors in
progression of kidney damage. Dr Wheeler is a member of
the International Steering Committee of the Study of
Heart and Renal Protection (SHARP) and was UK National
Coordinator for the trial. He is involved in several other
randomized trials and observational studies involving
patients with CKD.
Dr Wheeler is Co-Chair of KDIGO, having served
previously on its Executive Committee and Board. He
received an International Distinguished Medal from the
US NKF in recognition of his contribution to guideline
development. In the UK, he has been elected President of
the Renal Association for the term 2012-2014.
Dr Wheeler has served on the editorial boards of American
Journal of Kidney Diseases and Journal of the American Society
of Nephrology and is presently Co-Editor for Nephrology
Dialysis Transplantation.
Advisor/Consultant: Amgen
Honoraria: Abbott; Amgen; Fresenius; Otsuka; Shire
Travel Stipend: Amgen; Merck Sharp & Dohme; Shire
Grant/Research Support: Abbott; Genzyme
EVIDENCE REVIEW TEAM
Katrin Uhlig, MD, MS, is the Director, Guideline Develop-
ment at the Tufts Center for Kidney Disease Guideline
Development and Implementation, Boston, MA, USA,
Associate Professor of Medicine at Tufts University School
of Medicine, and a staff nephrologist at Tufts Medical Center.
Dr Uhlig completed her training in internal medicine,
nephrology, and rheumatology in Germany (Aachen Uni-
versity Hospital and Munich University Hospital) and the
USA (Georgetown University Medical Center and Tufts
Medical Center). Since 2001, she has been participating in or
directing the evidence review for KDOQI and KDIGO
guidelines. As Director of Guideline Development, Dr Uhlig
has a substantial role in coordinating the ERT. She
orchestrates and supervises the guideline development
process and didactic curriculum that provides Work Group
members with formal instruction on topics related to guide-
line development. As project director on individual guide-
lines, she directs and supervises the collection, evaluation,
grading, and synthesis of evidence and the drafting and
revisions of the final evidence report. She provides metho-
dological guidance and training to Work Group members at
meetings regarding topic refinement, key question formula-
tion, data extraction, study assessment, evidence grading,
and recommendation formulation. She provides nephrology
expertise in the interpretation and review of guide-
line recommendations and evidence reports. In this capacity,
132 Kidney International Supplements (2013) 3, 128–133
biographic and disclosure information

Dr Uhlig possesses unique knowledge as a methods expert in
evidence synthesis and critical literature appraisal in the
domain of nephrology.
In 2005, she co-chaired the KDIGO Evidence Rating
Group to develop a consensus on grading of KDIGO
guidelines and also co-chaired the KDIGO Consensus
Conference on Guideline Methodology in October
2007. From 2006 to 2007, she served as Co-Editor of
American Journal of Kidney Diseases . Her teaching and
research focus includes evidence-based medicine, systematic
review, clinical practice guideline development, and critical
literature appraisal. Dr Uhlig lectures on guideline topics and
is a co-instructor of an annual course on meta-analysis in the
Master of Science Program at the Sackler School of Graduate
Biomedical Sciences at Tufts University.
Dr Uhlig reported no relevant financial relationships
Dana C Miskulin, MD, MS, is Assistant Professor of
Medicine at Tufts University School of Medicine, Boston,
MA, USA. She completed a fellowship in Clinical Care
Research and participated in the conduct of systematic
reviews and critical literature appraisals for this guideline.
Her primary research interests are in comparative effective-
ness research in dialysis patients, BP treatment in dialysis
patients, and autosomal dominant polycystic kidney disease.
Dr Miskulin reported no relevant financial relationships
Amy Earley, BS, is a project coordinator at the Tufts Center
for Kidney Disease Guideline Development and Implementa-
tion in Boston, MA, USA. She is key in coordinating the
guideline development activities within the ERT, especially in
the development of the evidence reports for all guidelines.
Ms Earley also heads the actual evidence review, which
includes running searches, screening, data extraction,
drafting of tables and methods sections, proofing of guideline
drafts, and critical literature appraisals. She participates in
the conduct of research projects at the Center and actively
collaborates with other members of the Center on indepen-
dent research topics and manuscript submissions.
Ms. Earley reported no relevant financial relationships
Shana Haynes, MS, DHSc, is a research assistant at the Tufts
Center for Kidney Disease Guideline Development and
Implementation in Boston, MA, USA. She participates in
all aspects of evidence review and guideline development. She
screens abstracts and articles, extracts data, and assists in the
drafting and editing of evidence tables. Dr Haynes also assists
in the development of clinical practice guidelines and
conducts systematic reviews and critical literature appraisals.
Dr Haynes reported no relevant financ ial relationships
Jenny Lamont, MS, is a project manager and medical writer
at the Tufts Center for Kidney Disease Guideline Develop-
ment and Implementation in Boston, MA, USA. She
participates in all aspects of evidence review and guideline
development, assists in the preparation of talks and manu-
scripts, and edits KDIGO draft guidelines currently in
progress.
Ms. Lamont reported no relevant financial relationships
Kidney International Supplements (2013) 3, 128–133 133
biographic and disclosure information

Acknowledgments
Kidney International Supplements (2013) 3, 134–135; doi:10.1038/kisup.2012.71
A special debt of gratitude is owed to the KDIGO Co-Chairs
Kai-Uwe Eckardt, Bertram Kasiske, David Wheeler, and the
KDIGO Board for their invaluable guidance throughout
the development of this guideline. In particular, we thank the
ERT members: Katrin Uhlig, Dana Miskulin, Amy Earley,
Shana Haynes, and Jenny Lamont for their substanti al
contribution to the rigorous assessment of the available
evidence. We are also especially grateful to the Work
Group members for their exper tise throughout the
entire process of literature review, data extraction, meeting
participation, the critical writing and editing of the
statements and rationale, which made the publication of
this guideline possible. The generous gift of their time and
dedication is greatly appreciated. The entire Work Group and
its Co-Chairs would also like to extend a special debt of
gratitude to Michael Cheung, who has committed time and
energy above and beyond, to the formatting and proof
reading of the completed manuscript. Without his help, we
would have been hard pushed to complete this task. Finally,
and on behalf of the Work Group, we gratefully acknowledge
the careful assessme nt of the draft guideline by external
reviewers. The Work Group considered all of the valuable
comments made and where appropriate, suggested changes
were incorporated into the final publication. The following
individuals provided feedback during the public review of the
draft guideline:
Mario Abbud-Filho; Hugo Abensur; Patricia Abreu;
Matias Abuchanab; Nasrulla Abutaleb; Anil K Agarwal;
Ahmed Akl; Mona Al-Rukhaimi; Fernando A Almeida; Altun
Bulent; Maria Almerinda Ribeiro Alves; Pablo Amair;
Richard Amerling; Alessandro Amore; Ryoichi Ando; Rajeev
Annigeri; Ramadan Arafa; Mustafa Arici; Mariano Arriola;
Ferruh Artunc; Suheir Assady; Marcelo Rodrigues Bacci;
Ashraf AM Bakr; Breda Pee
`
ovnik Balon; Rashad S Barsoum;
Don Batisky; Dawlat Belal; Mohammed Benyahia; Murray
Berall; Anatole Besarab; Premila Bhat; Mar Biechy; Patrick
Biggar; Brenda Brewer; Inga Arune Bumblyte; Jian-Fang Cai;
Rafael Burgos Calderon; Bernard Canaud; Jorge B Cannata-
Andı
´
a; Antonio Carlos Duarte Cardoso; Laurence Carroll;
Sue Cary; Luis Castillo; Arlene Chabanuk; Philippe Chau-
veau; Jiang -Hua Chen; Kathleen Claes; Peter Clausen; Jean
Colaneri; Mario Cozzolino; Andrew Crannage ; Michael
Cunningham; Jane S Davis; Kimberly Davis; Rodrigo Daza;
Julia
´
n Segura de la Morena; Luca De Nicola; Rodrigo Bueno
de Oliveira; Rogerio Baumgratz de Paula; Goffredo Del
Rosso; David DeMicco; Sheila Deziel; Bruno Didier;
Guillermo J Rosa Diez; Ian Dittmer; Walter Guillermo
Douthat; Reham Eid Elsayed; Montserrat Mercedes Dı
´
az
Encarnacio
´
n; Tracy Evans-Walker; Joyce Ezaki-Yamaguchi;
Jarraya Faical; Bonita Falkner; Sandro Feriozzi; Manuel
Ferreira; Charles Ferro; Joseph T Flynn; Patricia Folk; Lui G
Forni; Costas Fourtounas; Roberto Jorge da Silva Franco;
Ping Fu; Naohiko Fujii; Susan L Furth; Suzanne Gagne; Sishir
Gang; Colin C Geddes; Rozina Ghazalli; Karla Giles; Rodney
Gilkey; Cher yl Gilmar tin; Matthias Girndt; Richard J
Glassock; Carlo Francisco S Gochuico; Eliezer Golan; David
J Goldsmith; Fatima Ramirez Gonzalez; Suzanne Gore; Sı
´
lvia
Gra
`
cia-Garcia; Martin Haas; Lisa Hall; Karen Hamacher;
Kathy Schiro Harvey; Niwrutti Hase; Domingo Hernandez;
Elisabeth M Hodson; Ronald J Hogg; Hallvard Holdaas;
Michael F Holick; Noriyuki Homma; Lai Seong Hooi; Mark
Houser; Christian Hugo; Salwa Ibrahim; Enyu Imai ; Jula
Inrig; Sanjay Jain; Matthew James; Alan G Jardine; Andrzej
Jaroszynski; Simon Jenkins; Chandra Mauli Jha; Kazibek
Joldoshov; Graham Jones; Kamyar Kalantar-Zadeh; Nada
Kanaan; Gu
¨
lc¸in Kantarci; Frederick J Kaskel; Cecelia Kasnick;
Hasan Kayabasi; James S Kennedy; Johannes Kessel; Bryce A
Kiberd; Vera Koch; Daisuke Koya; Peter Krisper; Batya
Kristal; Stefan Krivoshiev; Dirk R J Kuypers; Francisco
Lacordelle; Craig B Langman; Roberta Lattes; Rakesh
Lattupalli; Keith Lau; Edgar V Lerma; Visnja Lezaic; Hao
Li; Yuan Liang; Robert Liebl; Jimmy A Light; Petrica Ligia;
Tuta Liliana; Annabelle Sy Lim; Jelka Lindic; Zhangsuo Liu;
Attilio Losito; Richard Lund; Kelvin Lynn; Linlin Ma; Janette
Mansour; Abeera Mansur; Judith Marin; Alberto Martinez-
Castelao; Ken Massey; Timothy Mathew; Michael Mauer;
Sandro Mazzaferro; Peter A McCullough; Amanda Medland;
Gong Mengchun; Enisa Mesic; Sergio A Mezzano; Marius
Miglinas; Tanuja Mishra; Takehiko Miyaji; Rosario
Montan
˜
e
´
s-Bermu
´
dez; Assad Monzer; Jose
´
M Morales;
Eugen Mota; Ricardo Mouzo; Cesar Loza Munarriz; Mohsen
Nafar; Judit Nagy; Ramesh Naik; Takahiro Nakayama;
Lavinia Negrea; Armando Luis Negri; Robert G Nelson;
Marcus Vinı
´
cius de Pa
´
dua Netto; Alicia M Neu; Eezsafryna
Azalin Nordin; Maurizio Nordio; Diego Novo; Thomas
Oates; Yun Kyu Oh; Raymond Oliva; Patricia de Sequera
Ortiz; Messaoud Ouziala; Antonino Paglialunga; Sonia
Pasquali; Ioan Mihai Patiu; Saime Paydas; Glenda Payne;
Erling Bjerregaard Pedersen; Adriana Pen
˜
alba; Graciela
Pennacchiotti; Harry Brian Peppiatt; Gerson Marques Pereira
Junior; Virginia Pernas; Todd E Pesavento; Frida Liane
Plavnik; Claudio Ponticelli; Roberto Pontremoli; Amilee
Poucher; Kearkiat Praditpornsilpa; Anastasia Ptinopoulou;
Wajeh Qunibi; Rashida Rahman; Venkataraman Rama-
nathan; Rober to Ramirez; Harun ur Rashid; Hugh Rayner;
Leonardo Marin Restrepo; Enrique Andre
´
s Ribes; Peter Ricci;
acknowledgments http://www.kidney-international.org
& 2013 KDIGO
134
Kidney International Supplements (2013) 3, 134–135

Troels Ring; A Adibul Rizvi; Nicolas Roberto Robles;
Karen Rochelle; Hector J Rodriguez; Sara Rotaru; Jacques
Rottembourg; Luis M Ruilope; Domenico Russo; Karen E
Ryals; Jordi Bover Sanjuan; Francisco Santa-Cruz; Rafael
Santamaria; Bento C Santos; Sergio Santos; Kenneth R Say;
Sharon Schatz; Francesco Paolo Schena; Adalbert Schiller;
Bernhard Schmekal; Rebecca Schmidt; Ammar Serawan;
Mehmet Sever; Deepak Sharma; Rober t Shay; Mitesh B
Sheth; Alexander Shutov; Aleksandar Sikole; Fernando Simal;
Dhavee Sirivongs; Itzchak Slotki; Yves Smets; Kimberly
Smith; Laura Sola; Yalcin Solak; David Spiegel; Maria
Stasios; Pavai Sthaneshwar; Gere Sunder-Plassmann; Charles
Swanepoel; Miha
´
ly Tapolyai; Natalia Tomilina; Rezan
Topaloglu; Robert D Toto; Giorgio Triolo; Kriang Tungsanga;
Katherine R Tuttle; Anitha Vijayan; Rowan Walker; Marc R
Weber; Clare Weekes; Talia Weinstein; Matthew R Weir;
Kerstin Westman; Jack F Wetzels; Sara Wolfson; Mai-Szu Wu;
Yanhua Wu; Li Xuemei; Jerry Yee; Elena Zakharova;
Kostyantyn Zakon; Weiming Zhang; Carmine Zoccali;
Alessandro Zuccala
`
.
Participation in the review does not necessarily constitute
endorsement of the content of this report by the above
individuals, or the organization or institution they represent.
Adeera Levin, MD, FRCPC, Work Group Co-Chair
Paul E Stevens, MB, FRCP, Work Group Co-Chair
Kidney International Supplements (2013) 3, 134–135 135
acknowledgments

References
Kidney International Supplements (2013) 3, 136–150; doi:10.1038/kisup.2012.72
1. National Kidney Foundation. K/DOQI clinical practice guidelines for
chronic kidney disease: evaluation, classification, and stratification. Am J
Kidney Dis 2002; 39: S1–266.
2. Astor BC, Matsushita K, Gansevoort RT et al. Lower estimated glomerular
filtration rate and higher albuminuria are associated with mortality and
end-stage renal disease. A collaborative meta-analysis of kidney disease
population cohorts. Kidney Int 2011; 79: 1331–1340.
3. Gansevoort RT, Matsushita K, van der Velde M et al. Lower estimated GFR
and higher albuminuria are associated with adverse kidney outcomes. A
collaborative meta-analysis of general and high-risk population cohorts.
Kidney Int 2011; 80: 93–104.
4. Matsushita K, van der Velde M, Astor BC et al. Association of estimated
glomerular filtration rate and albuminuria with all-cause and cardiovas-
cular mortality in general population cohorts: a collaborative meta-
analysis. Lancet 2010; 375: 2073–2081.
5. van der Velde M, Matsushita K, Coresh J et al. Lower estimated
glomerular filtration rate and higher albuminuria are associated with all-
cause and cardiovascular mortality. A collaborative meta-analysis of
high-risk population cohorts. Kidney Int 2011; 79: 1341–1352.
6. Levey AS, Stevens LA, Coresh J. Conceptual model of CKD: applications
and implications. Am J Kidney Dis 2009; 53: S4–16.
7. KDIGO AKI Work Group. KDIGO clinical practice guideline for acute
kidney injury. Kidney inter., Suppl. 2012; 2: 1–138.
8. KDIGO GN Work Group. KDIGO clinical practice guideline for glomer-
ulonephritis. Kidney inter., Suppl. 2012; 2: 139–274.
9. KDIGO CKD-MBD Work Group. KDIGO clinical practice guideli ne for the
diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-
Mineral and Bone Disorder (CKD-MBD). Kidney Int Suppl 2009; 76(Suppl 113):
S1–130.
10. KDIGO BP Work Group. KDIGO clinical practice guideline for the
management of blood pressure in chronic kidney disease. Kidney inter.,
Suppl. 2012; 2: 337–414.
11. KDIGO Anemia Work Group. KDIGO clinical practice guideline for anemia
in chronic kidney disease. Kidney inter., Suppl. 2012; 2: 279–335.
12. Herzog CA, Asinger RW, Berger AK et al. Cardiovascular disease in
chronic kidney disease. A clinical update from Kidney Disease: Improving
Global Outcomes (KDIGO). Kidney Int 2011; 80: 572–586.
13. Matzke GR, Aronoff GR, Atkinson AJ, Jr. et al. Drug dosing consideration in
patients with acute and chronic kidney disease-a clinical update from Kidney
Disease: Improving Global Outcomes (KDIGO). Kidney Int 2011; 80: 1122–1137.
14. Hsu CY, Ordonez JD, Chertow GM et al. The risk of acute renal failure in
patients with chronic kidney disease. Kidney Int 2008;
74: 101–107.
15. Hailpern SM, Melamed ML, Cohen HW et al. Moderate chronic kidney
disease and cognitive function in adults 20 to 59 years of age: Third
National Health and Nutrition Examination Survey (NHANES III). J Am Soc
Nephrol 2007; 18: 2205–2213.
16. James MT, Hemmelgarn BR, Wiebe N et al. Glomerular filtration rate,
proteinuria, and the incidence and consequences of acute kidney injury:
a cohort study. Lancet 2010; 376: 2096–2103.
17. James MT, Quan H, Tonelli M et al. CKD and risk of hospital ization and
death with pneumonia. Am J Kidney Dis 2009; 54: 24–32.
18. Wilhelm-Leen ER, Hall YN, M KT et al. Frailty and chronic kidney disease:
the Third National Health and Nutrition Evaluation Survey. Am J Med
2009; 122: 664–671 e662.
19. Levey AS, Coresh J. Chronic kidney disease. Lancet 2012; 379: 165–180.
20. Wesson L. Physiology of the human kidney. Grune & Stratton: New York, 1969.
21. Rowe JW, Andres R, Tobin JD. Letter: Age-adjusted standards for
creatinine clearance. Ann Intern Med 1976; 84: 567–569.
22. Poggio ED, Rule AD, Tanchanco R et al. Demographic and clinical
characteristics associated with glomerular filtration rates in living kidney
donors. Kidney Int 2009; 75: 1079–1087.
23. Rule AD, Amer H, Cornell LD et al. The association between age and
nephrosclerosis on renal biopsy among healthy adults. Ann Intern Med
2010; 152: 561–567.
24. Barai S, Gambhir S, Prasad N et al. Levels of GFR and protein-induced
hyperfiltration in kidney donors: a single-center experience in India. Am J
Kidney Dis 2008; 51: 407–414.
25. Eastwood JB, Kerry SM, Plange-Rhule J et al. Assessment of GFR by four
methods in adults in Ashanti, Ghana: the need for an eGFR equation for
lean African populations. Nephrol Dial Transplant 2010; 25: 2178–2187.
26. Jafar TH, Islam M, Jessani S et al. Level and determinants of kidney
function in a South Asian population in Pakistan. Am J Kidney Dis 2011;
58: 764–772.
27. Stevens LA, Coresh J, Greene T et al. Assessing kidney function–
measured and estimated glomerular filtration rate. N Engl J Med 2006;
354: 2473–2483.
28. Remuzzi G, Benigni A, Remuzzi A. Mechanisms of progression and
regression of renal lesions of chronic nephropathies and diabetes. J Clin
Invest 2006; 116:
288–296.
29. KDIGO Transplant Work Group. KDIGO clinical practice guideline for
the care of kidney transplant recipients. Am J Transplant 2009;
9 (Suppl 3): S1–155.
30. Levey AS, de Jong PE, Coresh J et al. The definition, classification, and
prognosis of chronic kidney disease: a KDIGO Controversies Conference
report. Kidney Int 2011; 80: 17–28.
31. Levey AS, Eckardt KU, Tsukamoto Y et al. Definition and classification of
chronic kidney disease: a position statement from Kidney Disease:
Improving Global Outcomes (KDIGO). Kidney Int 2005; 67: 2089–2100.
32. Eckardt KU, Berns JS, Rocco MV et al. Definition and classification of CKD:
the debate should be about patient prognosis–a position statement
from KDOQI and KDIGO. Am J Kidney Dis 2009; 53: 915–920.
33. Eknoyan G. Chronic kidney disease definition and classification: no need
for a rush to judgment. Kidney Int 2009; 75: 1015–1018.
34. El Nahas M. Cardio-Kidney-Damage: a unifying concept. Kidney Int 2010;
78: 14–18.
35. Levey AS, Astor BC, Stevens LA et al. Chronic kidney disease, diabetes,
and hypertension: what’s in a name? Kidney Int 2010; 78: 19–22.
36. Winearls CG, Glassock RJ. Dissecting and refining the staging of chronic
kidney disease. Kidney Int 2009; 75: 1009–1014.
37. Silva FG. The aging kidney: a review – part I. Int Urol Nephrol 2005; 37:
185–205.
38. Silva FG. The aging kidney: a review–part II. Int Urol Nephrol 2005; 37:
419–432.
39. Weinstein JR, Anderson S. The aging kidney: physiological changes.
Adv Chronic Kidney Dis 2010; 17: 302–307.
40. King AJ, Levey AS. Dietary protein and renal function. J Am Soc Nephrol
1993; 3: 1723–1737.
41. Vehaskari VM. Orthostatic proteinuria. Arch Dis Child 1982; 57: 729–730.
42. Seikaly MG, Ho PL, Emmett L et al. Chronic renal insufficiency in
children: the 2001 Annual Report of the NAPRTCS. Pediatr Nephrol 2003;
18: 796–804.
43. Hogg RJ, Furth S, Lemley KV et al. National Kidney Foundation’s Kidney
Disease Outcomes Quality Initiative clinical practice guidelines for
chronic kidney disease in children and adolescents: evaluation,
classification, and stratification. Pediatrics 2003; 111: 1416–1421.
44. Schwartz GJ, Brion LP, Spitzer A. The use of plasma creatinine
concentration for estimating glomerular filtration rate in infants,
children, and adolescents. Pediatr Clin North Am 1987; 34: 571–590.
45. Aperia A, Broberger O, Elinder G et al. Postnatal development of renal
function in pre-term and full-term infants. Acta Paediatr Scand 1981; 70:
183–187.
46. Bueva A, Guignard JP. Renal function in preterm neonates. Pediatr Res
1994; 36: 572–577.
47. Fetterman GH, Shuplock NA, Philipp FJ et al. The Growth and Maturation
of Human Glomeruli and Proximal Convolutions from Term to
Adulthood: Studies by Microdissection. Pediatrics 1965; 35: 601–619.
48. Guignard JP, Torrado A, Da Cunha O et al. Glomerular filtration rate in
the first three weeks of life. J Pediatr 1975; 87: 268–272.
49. Haycock GB. Development of glomerular filtration and tubular
sodium reabsorption in the human fetus and newborn. BrJUrol1998;
81 (Suppl 2): 33–38.
50. Gallini F, Maggio L, Romagnoli C et al. Progression of renal function
in preterm neonates with gestational age o or ¼ 32 weeks. Pediatr
Nephrol 2000; 15: 119–124.
references http://www.kidney-international.org
& 2013 KDIGO
136
Kidney International Supplements (2013) 3, 136–150

51. Vieux R, Hascoet JM, Merdariu D et al. Glomerular filtration rate reference
values in very preterm infants. Pediatrics 2010; 125: e1186–1192.
52. Schwartz GJ, Furth SL. Glomerular filtration rate measurement
and estimation in chronic kidney disease. Pediatr Nephrol 2007; 22:
1839–1848.
53. Waters AM. Chapter 6, Part 2: Functional development of the nephron.
In: Geary DF, Schaefer F (eds). Comprehensive Pediatric Nephrology,
Mosby Elsevier: Philadelphia, PA, 2008, pp 111–129.
54. Langlois V. Chapter 2: Laboratory evaluation at different ages. In: Geary
DF, Schaefer F (eds) Comprehensive Pediatric Nephrology, Mosby Elsevier:
Philadelphia, PA, 2008, pp 39–54.
55. Furth SL, Cole SR, Moxey-Mims M et al. Design and methods of
the Chronic Kidney Disease in Children (CKiD) prospective cohort study.
Clin J Am Soc Nephrol 2006; 1: 1006–1015.
56. Copelovitch L, Warady BA, Furth SL. Insights from the Chronic
Kidney Disease in Children (CKiD) study. Clin J Am Soc Nephrol 2011;
6: 2047–2053.
57. Seliger SL, Zhan M, Hsu VD et al. Chronic kidney disease adversely
influences patient safety. J Am Soc Nephrol 2008; 19: 2414–2419.
58. Go AS, Chertow GM, Fan D et al. Chronic kidney disease and the risks of
death, cardiovascular events, and hospitalization. N Engl J Med 2004;
351: 1296–1305.
59. Coresh J, Astor BC, Greene T et al. Prevalence of chronic kidney disease
and decreased kidney function in the adult US population: Third
National Health and Nutrition Examination Survey. Am J Kidney Dis 2003;
41: 1–12.
60. Burgert TS, Dziura J, Yeckel C et al. Microalbuminuria in pediatric obesity:
prevalence and relation to other cardiovascular risk factors.
Int J Obes (Lond) 2006; 30: 273–280.
61. Csernus K, Lanyi E, Erhardt E et al. Effect of childhood obesity and
obesity-related cardiovascular risk factors on glomerular and tubular
protein excretion. Eur J Pediatr 2005; 164: 44–49.
62. Houser MT, Jahn MF, Kobayashi A et al. Assessment of urinary protein
excretion in the adolescent: effect of body position and exercise.
J Pediatr 1986; 109: 556–561.
63. Trachtenberg F, Barregard L. The effect of age, sex, and race on urinary
markers of kidney damage in children. Am J Kidney Dis 2007; 50:
938–945.
64. Brem AS. Neonatal hematuria and proteinuria. Clin Perinatol 1981; 8:
321–332.
65. Hogg RJ, Portman RJ, Milliner D et al. Evaluation and management of
proteinuria and nephrotic syndrome in children: recommendations from
a pediatric nephrology panel established at the National Kidney
Foundation conference on proteinuria, albuminuria, risk, assessment,
detection, and elimination (PARADE). Pediatrics 2000; 105: 1242–1249.
66. Jones CA, Francis ME, Eberhardt MS et al. Microalbuminuria in the
US population: third National Health and Nutrition Examination Survey.
Am J Kidney Dis 2002; 39: 445–459.
67. Levey AS, Coresh J. Should the K/DOQI definition of chronic kidney
disease be changed? Am J Kidney Dis 2003; 42: 626–630.
68. Uhlig K, Levey AS. Developing guidelines for chronic kidney disease: we
should include all of the outcomes. Ann Intern Med 2012; 156: 599–601.
69. North American Pediatric Renal Trials and Collaborative Studies.
NAPRTCS 2008 Annual Report. (https://web.emmes.com/study/ped/
annlrept/Annual%20Report%20-2008.pdf). Accessed September 7, 2012.
70. Ardissino G, Dacco V, Testa S et al. Epidemiology of chronic renal failure
in children: data from the ItalKid project. Pediatrics 2003; 111: e382–387.
71. Pierce CB, Cox C, Saland JM et al. Methods for characterizing differences
in longitudinal glomerular filtration rate changes between children with
glomerular chronic kidney disease and those with nonglomerular
chronic kidney disease. Am J Epidemiol 2011; 174: 604–612.
72. Furth SL, Abraham AG, Jerry-Fluker J et al. Metabo lic abnormalities,
cardiovascular disease risk factors, and GFR decline in children with
chronic kidney disease. Clin J Am Soc Nephrol 2011; 6: 2132–2140.
73. Wingen AM, Fabian-Bach C, Schaefer F et al. Randomised multicentre
study of a low-protein diet on the progression of chronic renal failure in
children. European Study Group of Nutritional Treatment of Chronic
Renal Failure in Childhood. Lancet 1997; 349: 1117–1123.
74. Staples AO, Greenbaum LA, Smith JM et al. Association between clinical
risk factor s and progression of chronic kidney disease in children.
Clin J Am Soc Nephrol 2010; 5: 2172–2179.
75. Ardissino G, Testa S, Dacco V et al. Proteinuria as a predictor of disease
progression in children with hypodysplastic nephropathy. Data from the
Ital Kid Project. Pediatr Nephrol 2004; 19: 172–177.
76. Wong CS, Pierce CB, Cole SR et al. Association of proteinuria with race,
cause of chronic kidney disease, and glomerular filtration rate in the
chronic kidney disease in children study. Clin J Am Soc Nephrol 2009; 4:
812–819.
77. Ardissino G, Testa S, Dacco V et al. Puberty is associated with increased
deterioration of renal function in patients with CKD: data from the ItalKid
Project. Arch Dis Child 2012; 97: 885–888.
78. Querfeld U, Anarat A, Bayazit AK et al. The Cardiovascular Comorbidity in
Children with Chronic Kidney Disease (4C) study: objectives, design, and
methodology. Clin J Am Soc Nephrol
2010; 5: 1642–1648.
79. Stevens LA, Levey AS. Measured GFR as a confirmatory test for estimated
GFR. J Am Soc Nephrol 2009; 20: 2305–2313.
80. Schwartz GJ, Munoz A, Schneider MF et al. New equations to estimate
GFR in children with CKD. J Am Soc Nephrol 2009; 20: 629–637.
81. Myers GL, Miller WG, Coresh J et al. Recommendations for improving
serum creatinine measurement: a report from the Laboratory Working
Group of the National Kidney Disease Education Program. Clin Chem
2006; 52: 5–18.
82. Miller WG. Estimating glomerular filtration rate. Clin Chem Lab Med 2009;
47: 1017–1019.
83. Kilpatrick ES, Verrill H. A national audit of estimated glomerular filtration
rate and proteinuria reporting in the UK. Ann Clin Biochem 2011; 48:
558–561.
84. McIntosh JF, Moller E, Van Slyke DD. Studies of urea excretion. III: The
influence of body size on urea output. J Clin Invest 1928; 6: 467–483.
85. Earley A, Miskulin D, Lamb EJ et al. Estimating equations for glomerular
filtration rate in the era of creatinine standardization: a systematic
review. Ann Intern Med 2012; 156: 785–795.
86. Levey AS, Coresh J, Greene T et al. Using standardized serum creatinine
values in the modification of diet in renal disease study equation for
estimating glomerular filtration rate. Ann Intern Me d 2006; 145: 247–254.
87. Levey AS, Stevens LA, Schmid CH et al. A new equation to estimate
glomerular filtration rate. Ann Intern Med 2009; 150: 604–612.
88. Horio M, Imai E, Yasuda Y et al. Modification of the CKD epidemiology
collaboration (CKD-EPI) equation for Japanese: accuracy and use for
population estimates. Am J Kidney Dis 2010; 56: 32–38.
89. Imai E, Horio M, Nitta K et al. Estimation of glomerular filtration rate by
the MDRD study equation modified for Japanese patients with chronic
kidney disease. Clin Exp Nephrol 2007; 11: 41–50.
90. Praditpornsilpa K, Townamchai N, Chaiwatanarat T et al. The need for
robust validation for MDRD-based glomerular filtration rate estimation in
various CKD populations. Nephrol Dial Transplant 2011; 26: 2780–2785.
91. Matsuo S, Imai E, Horio M et al. Revised equations for estimated GFR
from serum creatinine in Japan. Am J Kidney Dis 2009; 53: 982–992.
92. Levey AS, Greene T, Kusek J
et al. A simplified equation to predict
glomerular filtration rate from serum creatinine. J Am Soc Nephrol 2000;
11: 155A.
93. Ma YC, Zuo L, Chen JH et al. Modified glomerular filtration rate
estimating equation for Chinese patients with chronic kidney disease.
J Am Soc Nephrol 2006; 17: 2937–2944.
94. Levey AS, Bosch JP, Lewis JB et al. A more accurate method to estimate
glomerular filtration rate from serum creatinine: a new prediction
equation. Modification of Diet in Renal Disease Study Group. Ann Intern
Med 1999; 130: 461–470.
95. Murata K, Baumann NA, Saenger AK et al. Relative performance of the
MDRD and CKD-EPI equations for estimating glomerular filtration rate
among patients with varied clinical presentations. Clin J Am Soc Nephrol
2011; 6: 1963–1972.
96. Lane BR, Demirjian S, Weight CJ et al. Performance of the chronic kidney
disease-epidemiology study equations for estimating glomerular filtra-
tion rate before and after nephrectomy. J Urol 2010; 183: 896–901.
97. Michels WM, Grootendorst DC, Verduijn M et al. Performance of the
Cockcroft-Gault, MDRD, and new CKD-EPI formulas in relation to GFR,
age, and body size. Clin J Am Soc Nephrol 2010; 5: 1003–1009.
98. Tent H, Rook M, Stevens LA et al. Renal function equations before
and after living kidney donation: a within-individual comparison of
performance at different levels of renal function. Clin J Am Soc Nephrol
2010; 5: 1960–1968.
99. Kukla A, El-Shahawi Y, Leister E et al. GFR-estimating models in kidney
transplant recipients on a steroid-free regimen. Nephrol Dial Transplant
2010; 25: 1653–1661.
100. White CA, Akbari A, Doucette S et al. Estimating glomerular filtration
rate in kidney transplantation: is the new chron ic kidney disease
epidemiology collaboration equation any better? Clin Chem 2010; 56:
474–477.
101. Poge U, Gerhardt T, Stoffel-Wagner B et al. Validation of the CKD-EPI
formula in patients after renal transplantation . Nephrol Dial Transplant
2011; 26: 4104–4108.
Kidney International Supplements (2013) 3, 136–150 137
references

102. Jones GR, Imam SK. Validation of the revised MDRD formula and the
original Cockcroft and Gault formula for estimation of the glomerular
filtration rate using Australian data. Pathology 2009; 41: 379–382.
103. Jones GR. Use of the CKD-EPI equation for estimation of GFR in an
Australian cohort. Pathology 2010; 42: 487–488.
104. Cirillo M, Lombardi C, Luciano MG et al. Estimation of GFR: a comparison
of new and established equations. Am J Kidney Dis 2010; 56: 802–804.
105. Eriksen BO, Mathisen UD, Melsom T et al. Cystatin C is not a better
estimator of GFR than plasma creatinine in the general population.
Kidney Int 2010; 78: 1305–1311.
106. Redal-Baigorri B, Stokholm KH, Rasmussen K et al. Estimation of kidney
function in cancer patients. Dan Med Bull 2011; 58: A4236.
107. Matsushita K, Mahmoodi BK, Woodward M et al. Comparison of risk
prediction using the CKD-EPI equation and the MDRD study equation for
estimated glomerular filtration rate. JAMA 2012; 307: 1941–1951.
108. Rule AD, Teo BW. GFR estimation in Japan and China: what accounts for
the difference? Am J Kidney Dis 2009; 53: 932–935.
109. Stevens LA, Claybon MA, Schmid CH et al. Evaluation of the Chronic
Kidney Disease Epidemiology Collaboration equation for estimating the
glomerular filtration rate in multiple ethnicities. Kidney Int 2011; 79:
555–562.
110. Yeo Y, Han DJ, Moon DH et al. Suitability of the IDMS-traceable MDRD
equation method to estimate GFR in early postoperative renal transplant
recipients. Nephron Clin Pract 2010; 114: c108–117.
111. van Deventer HE, George JA, Paiker JE et al. Estimating glomerular filtration
rate in black South Africans by use of the modification of diet in renal
disease and Cockcroft-Gault equations. Clin Chem 2008; 54: 1197–1202.
112. Teo BW, Xu H, Wang D et al. GFR estimating equations in a multiethnic
Asian population. Am J Kidney Dis 2011; 58: 56–63.
113. Inker LA, Schmid CH, Tighiouart H et al. Estimating glomerular filtration
rate from serum creatinine and cystatin C. N Engl J Med 2012; 367: 20–29.
114. Peralta CA, Shlipak MG, Judd S et al. Detection of chronic kidney disease
with creatinine, cystatin C, and urine albumin-to-creatinine ratio and
association with progression to end-stage renal disease and mortality.
JAMA 2011; 305: 1545–1552.
115. Schwartz GJ, Schneider MF, Maier PS et al.
Improved equations estima-
ting GFR in children with chronic kidney disease using an immuno-
nephelometric determination of cystatin C. Kidney Int 2012; 82: 445–453.
116. Inker LA, Eckfeldt J, Levey AS et al. Expressing the CKD-EPI (Chronic
Kidney Disease Epidemiology Collaboration) cystatin C equations for
estimating GFR with standardized serum cystatin C values. Am J Kidney
Dis 2011; 58: 682–684.
117. Stevens LA, Coresh J, Schmid CH et al. Estimating GFR using serum cystatin
C alone and in combination with serum creatinine: a pooled analysis of
3,418 individuals with CKD. Am J Kidney Dis 2008; 51: 395–406.
118. Zappitelli M, Parvex P, Joseph L et al. Derivation and validation of
cystatin C-based prediction equations for GFR in children. Am J Kidney
Dis 2006; 48: 221–230.
119. Filler G, Lepage N. Should the Schwartz formula for estimation of GFR be
replaced by cystatin C formula? Pediatr Nephrol 2003; 18: 981–985.
120. Hoek FJ, Kemperman FA, Krediet RT. A comparison between cystatin C,
plasma creatinine and the Cockcroft and Gault formula for the estimation of
glomerular filtration rate. Nephrol Dial Transplant 2003; 18: 2024–2031.
121. Kwong YT, Stevens LA, Selvin E et al. Imprecision of urinary iothalamate
clearance as a gold-standard measure of GFR decreases the diagnostic
accuracy of kidney function estimating equations. Am J Kidney Dis 2010;
56: 39–49.
122. Lamb EJ, Price CP. Kidney function tests. In: Burtis CA, Ashwood E, (eds.)
Bruns DE. Tietz Textbook of Clinical Chemistry and Molecular Diagnostics,
5
th
edition, Elsevier, 2012, pp 669–708.
123. Ballantyne FC, Gibbons J, O’Reilly DS. Urine albumin should replace total
protein for the assessment of glomerular proteinuria. Ann Clin Biochem
1993; 30 (Pt 1): 101–103.
124. Lamb EJ, MacKenzie F, Stevens PE. How should proteinuria be detected
and measured? Ann Clin Biochem 2009; 46: 205–217.
125. Newman DJ, Thakkar H, Medcalf EA et al. Use of urine albumin
measurement as a replacement for total protein. Clin Nephrol 1995; 43:
104–109.
125a . Hallan SI, Ritz E, Lydersen S et al. Combining GFR and albuminuria to classify
CKD improves prediction of ESRD. JAmSocNephrol2009; 20: 1069–1077.
125b. Brantsma AH, Bakker SJ, Hillege HL et al. Cardiovascular and renal
outcome in subjects with K/DOQI stage 1–3 chronic kidney disease: the
importance of urinary albumin excretion. Nephrol Dial Transplant 2008;
23: 3851–3858.
126. Dawnay A, Wilson AG, Lamb E et al. Microalbuminuria in systemic
sclerosis. Ann Rheum Dis 1992; 51: 384–388.
127. Gross JL, de Azevedo MJ, Silveiro SP et al. Diabetic nephropathy:
diagnosis, prevention, and treatment. Diabetes Care 2005; 28: 164–176.
128. Ninomiya T, Perkovic V, de Galan BE et al. Albuminuria and kidney
function independently predict cardiovascular and renal outcomes in
diabetes. J Am Soc Nephrol 2009; 20: 1813–1821.
129. Shihabi ZK, Konen JC, O’Connor ML. Albuminuria vs urinary total protein
for detecting chronic renal disorders. Clin Chem 1991; 37: 621–624.
130. Martin H. Laboratory measurement of urine albumin and urine
total protein in screening for proteinuria in chronic kidney disease.
Clin Biochem Rev 2011; 32: 97–102.
131. Waugh J, Bell SC, Kilby M et al. Effect of concentration and biochemical
assay on the accuracy of urine dipsticks in hypertensive pregnancies.
Hypertens Pregnancy 2001; 20: 205–217.
132. Waugh J, Bell SC, Kilby MD et al. Urine protein estimation in hypertensive
pregnancy: which thresholds and laboratory assay best predict clinical
outcome? Hypertens Pregnancy 2005; 24: 291–302.
133. McElderry LA, Tarbit IF, Cassells-Smith AJ. Six methods for urinary protein
compared. Clin Chem 1982; 28: 356–360.
134. Nishi HH, Elin RJ. Three turbidimetric methods for determining total
protein compared. Clin Chem 1985;
31: 1377–1380.
135. Sedmak JJ, Grossberg SE. A rapid, sensitive, and versatile assay for protein
using Coomassie brilliant blue G250. Anal Biochem 1977; 79: 544–552.
136. de Keijzer MH, Klasen IS, Branten AJ et al. Infusion of plasma expanders
may lead to unexpected results in urinary protein assays. Scand J Clin Lab
Invest 1999; 59: 133–137.
137. Marshall T, Williams KM. Extent of aminoglycoside interference in the
pyrogallol red-molybdate protein assay depends on the concentration
of sodium oxalate in the dye reagent. Clin Chem 2004; 50: 934–935.
138. Yilmaz FM, Yucel D. Effect of addition of hemolysate on urine and
cerebrospinal fluid assays for protein. Clin Chem 2006; 52: 152–153.
139. Chambers RE, Bullock DG, Whicher JT. External quality assessment of
total urinary protein estimation in the United Kingdom. Ann Clin Biochem
1991; 28 (Pt 5): 467–473.
140. Heick HM, Begin-Heick N, Acharya C et al. Automated determination of
urine and cerebrospinal fluid proteins with Coomassie Brilliant Blue and
the Abbott ABA-100. Clin Biochem 1980; 13: 81–83.
141. Marshall T, Williams KM. Total protein determination in urine: elimination
of a differential response between the coomassie blue and pyrogallol
red protein dye-binding assays. Clin Chem 2000; 46: 392–398.
142. Miller WG. Urine albumin: Recommendations for standardization. Scand J
Clin Lab Invest Suppl 2008; 241: 71–72.
143. Miller WG, Bruns DE, Hortin GL et al. Current issues in measurement and
reporting of urinary albumin excretion. Clin Chem 2009; 55: 24–38.
144. Medicines and Healthcare products Regulatory Agency. MHRA 04086
Point of care devices for the quantitation of microalbuminuria. 2004.
145. Medicines and Healthcare products Regulatory Agency. MHRA 04098.
Point of care devices for the detection and semi-quantitation of
microalbuminuria. 2004.
146. Parsons M, Newman DJ, Pugia M et al. Performance of a reagent strip
device for quantitation of the urine albumin: creatinine ratio in a point of
care setting. Clin Nephrol 1999; 51: 220–227.
147. Parsons MP, Newman DJ, Newall RG et al. Validation of a point-of-care
assay for the urinary albumin:creatinine ratio. Clin Chem 1999; 45: 414–417.
148. Graziani MS, Gambaro G, Mantovani L et al. Diagnostic accuracy of a
reagent strip for assessing urinary albumin excretion in the general
population. Nephrol Dial Transplant 2009; 24: 1490–1494.
149. Guy M, Newall R, Borzomato J et al. Diagnostic accuracy of the urinary
albumin: creatinine ratio determined by the CLINITEK Microalbumin and
DCA 2000 þ for the rule-out of albuminuria in chronic kidney disease.
Clin Chim Acta 2009; 399: 54–58.
150. Waugh JJ, Bell SC, Kilby MD et al. Optimal bedside urinalysis for the
detection of proteinuria in hypertensive pregnancy: a study of
diagnostic accuracy. BJOG 2005; 112: 412–417.
151. Iseki K, Iseki C, Ikemiya Y et al. Risk of developing end-stage renal disease
in a cohort of mass screening. Kidney Int 1996; 49: 800–805.
152. Kaplan RE, Springate JE, Feld LG. Screening dipstick urinalysis: a time to
change. Pediatrics 1997; 100: 919–921.
153. Kitagawa T. Lessons learned from the Japanese nephritis screening
study. Pediatr Nephrol 1988; 2: 256–263.
154. Boulware LE, Jaar BG, Tarver-Carr ME et al. Screening for proteinuria in
US adults: a cost-effectiveness analysis. JAMA 2003; 290: 3101–3114.
155. Bowie L, Smith S, Gochman N. Characteristics of binding between
reagent-strip indicators and urinary proteins. Clin Chem 1977; 23:
128–130.
156. Gyure WL. Comparison of several methods for semiquantitative
determination of urinary protein. Clin Chem 1977; 23: 876–879.
138 Kidney International Supplements (2013) 3, 136–150
references

157. James GP, Bee DE, Fuller JB. Proteinuria: accuracy and precision of
laboratory diagnosis by dip-stick analysis. Clin Chem 1978; 24: 1934–1939.
158. Rumley A. Urine dipstick testing: comparison of results obtained by
visual reading and with the Bayer CLINITEK 50. Ann Clin Biochem 2000;
37 (Pt 2): 220–221.
159. Scotti da Silva-Colombeli A, Falkenberg M. Analytical interferences of
drugs in the chemical examination of urinary protein. Clin Biochem 2007;
40: 1074–1076.
160. Ralston SH, Caine N, Richards I et al. Screening for proteinuria in a
rheumatology clinic: comparison of dipstick testing, 24 h urine
quantitative protein, and protein/creatinine ratio in random urine
samples. Ann Rheum Dis 1988; 47: 759–763.
161. Waugh JJ, Clark TJ, Divakaran TG et al. Accuracy of urinalysis dipstick
techniques in predicting significant proteinuria in pregnancy. Obstet
Gynecol 2004; 103: 769–777.
162. White SL, Yu R, Craig JC et al. Diagnostic accuracy of urine dipsticks for
detection of albuminuria in the general community. Am J Kidney Dis
2011; 58: 19–28.
163. Saudan PJ, Brown MA, Farrell T et al. Improved methods of assessing
proteinuria in hypertensive pregnancy. Br J Obstet Gynaecol 1997; 104:
1159–1164.
164. Beetham R, Cattell WR. Proteinuria: pathophysiology, significance and
recommendations for measurement in clinical practice. Ann Clin Biochem
1993; 30 (Pt 5): 425–434.
165. Keane WF, Eknoyan G. Proteinuria, albuminuria, risk, assessment,
detection, elimination (PARADE): a position paper of the National Kidney
Foundation. Am J Kidney Dis 1999; 33: 1004–1010.
166. Claudi T, Cooper JG. Comparison of urinary albumin excretion rate in
overnight urine and albumin creatinine ratio in spot urine in diabetic
patients in general practice. Scand J Prim Health Care 2001; 19: 247–248.
167. Gatling W, Knight C, Mullee MA et al. Microalbuminuria in diabetes: a
population study of the prevalence and an assessment of three
screening tests. Diabet Med 1988; 5: 343–347.
168. Hutchison AS, O’Reilly DS, MacCuish AC. Albumin excretion rate, albumin
concentration, and albumin/creatinine ratio compared for screening
diabetics for slight albuminuria. Clin Chem 1988; 34: 2019–2021.
169. Marshall SM. Screening for microalbuminuria: which measurement?
Diabet Med 1991; 8: 706–711.
170. Marshall SM, Alberti KG. Screening for early diabetic nephropathy.
Ann Clin Biochem 1986; 23 (Pt 2): 195–197.
171. Chitalia VC, Kothari J, Wells EJ et al. Cost-benefit analysis and prediction
of 24-hour proteinuria from the spot urine protein-creatinine ratio.
Clin Nephrol
2001; 55: 436–447.
172. Cote AM, Brown MA, Lam E et al. Diagnostic accuracy of urinary spot
protein:creatinine ratio for proteinuria in hypertensive pregnant women:
systematic review. BMJ 2008; 336: 1003–1006.
173. Dyson EH, Will EJ, Davison AM et al. Use of the urinary protein creatinine
index to assess proteinuria in renal transplant patients. Nephrol Dial
Transplant 1992; 7: 450–452.
174. Ginsberg JM, Chang BS, Matarese RA et al. Use of single voided urine
samples to estimate quantitative proteinur ia. N Engl J Med 1983; 309:
1543–1546.
175. Leanos-Miranda A, Marquez-Acosta J, Romero-Arauz F et al. Protein:-
creatinine ratio in random urine sampl es is a reliable marker of increased
24-hour protein excretion in hospitalized women with hypertensive
disorders of pregnancy. Clin Chem 2007; 53: 1623–1628.
176. Lemann J, Jr., Doumas BT. Proteinuria in health and disease assessed by
measuring the urinary protein/creatinine ratio. Clin Chem 1987; 33:
297–299.
177. Ruggenenti P, Gaspari F, Perna A et al. Cross sectional longitudinal study
of spot morning urine protein:creatinine ratio, 24 h urine protein excretion
rate, glomerular filtration rate, and end stage renal failure in chronic renal
disease in patients without diabetes. BMJ 1998; 316: 504–509.
178. Pugliese G, Solini A, Fondelli C et al. Reproducibility of albuminuria in
type 2 diabetic subjects. Findings from the Renal Insufficiency And
Cardiovascular Events (RIACE) study. Nephrol Dial Transplant 2011; 26:
3950–3954.
179. Newman DJ, Pugia MJ, Lott JA et al. Urinary protein and albumin
excretion corrected by creatinine and specific gravity. Clin Chim Acta
2000; 294: 139–155.
180. Howey JE, Browning MC, Fraser CG. Selecting the optimum specimen for
assessing slight albuminuria, and a strategy for clinical investigation: novel
uses of data on biological variation. Clin Chem 1987; 33: 2034–2038.
181. Carter JL, Tomson CR, Stevens PE et al. Does urinary tract infection cause
proteinuria or microalbuminuria? A systematic review. Nephrol Dial
Transplant 2006; 21: 3031–3037.
182. Heathcote KL, Wilson MP, Quest DW et al. Prevalence and duration of
exercise induced albuminuri a in healthy people. Clin Invest Med 2009;
32: E261–265.
183. Leung AK, Wong AH. Proteinuria in children. Am Fam Physician 2010; 82:
645–651.
184. Boger CA, Chen MH, Tin A et al. CUBN is a gene locus for albuminuria.
J Am Soc Nephrol 2011;
22: 555–570.
185. Price CP, Newall RG, Boyd JC. Use of protein:creatinine ratio measure-
ments on random urine samples for prediction of significant proteinuria:
a systematic review. Clin Chem 2005; 51: 1577–1586.
186. National Institute for Health and Clinical Excellence. NICE clinical
guideline 73. Chronic kidney disease: early identification and manage-
ment of chronic kidney disease in adults in primary and secondary care.
2008.
187. Montanes Bermudez R, Gracia Garcia S, Perez Surribas D et al. Consensus
document. Recommendations on assessing proteinuria during the
diagnosis and follow-up of chronic kidney disease. Nefrologia 2011;
31: 331–345.
188. Johnson DW, Jones GR, Mathew TH et al. Chronic kidney disease and
measurement of albuminuria or proteinuria: a position statement. Med J
Aust 2012; 197: 224–225.
189. Scottish Intercollegiate Guidelines Network. Guideline 103. Diagnosis
and management of chronic kidney disease. 2008.
190. Caring for Australasians with Renal Impairment. http://www.cari.org.au/
guidelines.php
191. Clarke W, Frost SJ, Kraus E et al. Renal function testing. In: Nichols JH (ed)
Evidence-based Practice for Point-of-Care Testing. National Academy of
Clinical Biochemistry, 2006, pp 126–134.
192. Panek R, Lawen T, Kiberd BA. Screening for proteinuria in kidney
transplant recipients. Nephrol Dial Transplant 2011; 26: 1385–1387.
193. Incerti J, Zelmanovitz T, Camargo JL et al. Evaluation of tests for
microalbuminuria screening in patients with diabetes. Nephrol Dial
Transplant 2005; 20: 2402–2407.
194. Methven S, MacGregor MS, Traynor JP et al. Assessing proteinuria in
chronic kidney disease: protein-creatinine ratio versus albumin-creati-
nine ratio. Nephrol Dial Transplan t 2010; 25: 2991–2996.
195. Methven S, MacGregor MS, Traynor JP et al. Comparison of urinary
albumin and urinary total protein as predictors of patient outcomes in
CKD. Am J Kidney Dis 2011; 57: 21–28.
196. Methven S, Traynor JP, Hair MD et al. Stratifying risk in chronic kidney
disease: an observational study of UK guidelines for measuring total
proteinuria and albuminuria. QJM 2011; 104: 663–670.
197. Nauta FL, Bakker SJ, van Oeveren W et al. Albuminuria, proteinuria, and
novel urine biomarkers as predictors of long-term allograft outcomes in
kidney transplant recipients. Am J Kidney Dis 2011; 57: 733–743.
198. Ellam TJ. Albumin:creatinine ratio–a flawed mea sure? The merits of
estimated albuminuria reporting. Nephron Clin Pract 2011; 118:
c324–330.
199. Comper WD, Osicka TM, Clark M et al. Earlier detection of micro-
albuminuria in diabetic patients using a new urinary albumin assay.
Kidney Int 2004; 65:
1850–1855.
200. Comper WD, Osicka TM, Jerums G. High prevalence of immuno-
unreactive intact albumin in urine of diabetic patients. Am J Kidney Dis
2003; 41: 336–342.
201. Magliano DJ, Polkinghorne KR, Barr EL et al. HPLC-detected albuminuria
predicts mortality. J Am Soc Nephrol 2007; 18: 3171–3176.
202. Osicka TM, Comper WD. Characterization of immunochemically non-
reactive urinary albumin. Clin Chem 2004; 50: 2286–2291.
203. Sviridov D, Drake SK, Hortin GL. Reactivity of urinary albumin
(microalbumin) assays with fragmented or modified albumin. Clin Chem
2008; 54: 61–68.
204. Sviridov D, Meilinger B, Drake SK et al. Coelution of other proteins with
albumin during size-exclusion HPLC: Implications for analysis of urinary
albumin. Clin Chem 2006; 52: 389–397.
205. Tsioufis C, Mazaraki A, Dimitriadis K et al. Microalbuminuria in the
paediatric age: current knowledge and emerging questions. Acta
Paediatr 2011; 100: 1180–1184.
206. Rademacher ER, Sinaiko AR. Albuminuri a in children. Curr Opin Nephrol
Hypertens 2009; 18: 246–251.
207. Wrong OM, Norden AG, Feest TG. Dent’s disease; a familial proximal
renal tubular syndrome with low-molecular-weight proteinuria, hyper-
calciuria, nephrocalcinosis, metabolic bone disease, progressive renal
failure and a marked male predominance. QJM 1994; 87: 473–493.
208. Atkins RC, Briganti EM, Zimmet PZ et al. Association between
albuminuria and proteinuria in the general population: the AusDiab
Study. Nephrol Dial Transplant 2003; 18: 2170–2174.
Kidney International Supplements (2013) 3, 136–150 139
references

209. Gosling P. Proteinuria. In: Marshall WJ, Bangert SK (eds). Clinical
Biochemistry: Metabolic and Clinical Aspects, 2nd Ed. Elsevier, 2008,
pp 156–173.
210. Goren MP, Li JT. The Coomassie Brilliant Blue method underestimates
drug-induced tubular proteinuria. Clin Chem 1986; 32: 386–388.
211. Weber MH, Verwiebe R. Alpha 1-microglobulin (protein HC): features of a
promising indicator of proximal tubular dysfunction. Eur J Clin Chem Clin
Biochem 1992; 30: 683–691.
212. Herget-Rosenthal S, Poppen D, Husing J et al. Prognostic value of tubular
proteinuria and enzymuria in nonoliguric acute tubular necrosis. Clin
Chem 2004; 50: 552–558.
213. Ginevri F, Piccotti E, Alinovi R et al. Reversible tubular proteinuria
precedes microalbuminuria and correlates with the metabolic status in
diabetic children. Pediatr Nephrol 1993; 7: 23–26.
214. Tomlinson PA, Smellie JM, Prescod N et al. Differential excretion of
urinary proteins in children with vesicoureteric reflux and reflux
nephropathy. Pediatr Nephrol 1994; 8: 21–25.
215. Bird JM, Owen RG, D’Sa S et al. Guidelines for the diagnosis and
management of multiple myeloma 2011. Br J Haematol 2011; 154:
32–75.
216. Slack TK, Wilson DM. Normal renal function: CIN and CPAH in healthy
donors before and after nephrectomy. Mayo Clinic Proc 1976; 51: 296–300.
217. Rowe JW, Andres R, Tobin JD et al. The effect of age on creatinine
clearance in men: a cross-sectional and longitudinal study. J Gerontol
1976; 31: 155–163.
218. Lindeman RD, Tobin JD, Shock NW. Association between blood pressure
and the rate of decline in renal function with age. Kidney Int 1984; 26:
861–868.
219. Halbesma N, Kuiken DS, Brantsma AH et al. Macroalbuminuria is a better
risk marker than low estimated GFR to identify individuals at risk for
accelerated GFR loss in population screening. J Am Soc Nephrol 2006; 17:
2582–2590.
220. Imai E, Horio M, Yamagata K et al. Slower decline of glomerular filtration
rate in the Japanese general population: a longitudinal 10-year follow-up
study. Hypertens Res 2008; 31: 433–441.
221. Matsushita K, Selvin E, Bash LD et al. Change in estimated GFR associates
with coronary heart disease and mortality. J Am Soc Nephrol 2009; 20:
2617–2624.
222. Kronborg J, Solbu M, Njolstad I et al. Predictors of change in estimated
GFR: a population-based 7-year follow-up from the Tromso study.
Nephrol Dial Transplant 2008; 23: 2818–2826.
223. Hemmelgarn BR, Zhang J, Manns BJ et al. Progression of kidney
dysfunction in the community-dwelling elderly. Kidney Int 2006; 69:
2155–2161.
224. Keller C, Katz R, Sarnak MJ et al. Inflammatory biomarkers and decline in
kidney function in the elderly: the Cardiovascular Health Study. Nephrol
Dial Transplant 2010; 25: 119–124.
225. John R, Webb M, Young A et al. Unreferred chronic kidney disease: a
longitudinal study. Am J Kidney Dis 2004; 43: 825–835.
226. Levey AS, Gassman JJ, Hall PM et al. Assessing the progression of renal
disease in clinical studies: effects of duration of follow-up and regression
to the mean. Modification of Diet in Renal Disease (MDRD) Study Group.
J Am Soc Nephrol 1991; 1: 1087–1094.
227. Klahr S, Levey AS, Beck GJ et al. The effects of dietary protein restriction
and blood-pressure control on the progression of chronic renal disease.
Modification of Diet in Renal Disease Study Group. N Engl J Med 1994;
330: 877–884.
228. Wright JT, Jr., Bakris G, Greene T et al. Effect of blood pressure
lowering and antihypertensive drug class on progression of hyper-
tensive kidney disease: results from the AASK trial. JAMA 2002; 288:
2421–2431.
229. Eriksen BO, Ingebretsen OC. The progression of chronic kidney disease: a
10-year population-based study of the effects of gender and age. Kidney
Int 2006; 69: 375–382.
230. Jones C, Roderick P, Harris S et al. Decline in kidney function before and
after nephrology referral and the effect on survival in moderate to
advanced chronic kidney disease. Nephrol Dial Transplant 2006; 21:
2133–2143.
231. Levin A, Djurdjev O, Beaulieu M et al. Variability and risk factors for
kidney disease progression and death following attainment of stage 4
CKD in a referred cohort. Am J Kidney Dis 2008; 52: 661–671.
232. Al-Aly Z, Zeringue A, Fu J et al. Rate of kidney function decline associates
with mortality. J Am Soc Nephrol 2010; 21: 1961–1969.
233. Shlipak MG, Katz R, Kestenbaum B et al. Rapid decline of kidney function
increases cardiovascular risk in the elderly. J Am Soc Nephrol 2009; 20:
2625–2630.
234. Cheng TY, Wen SF, Astor BC et al. Mortality risks for all causes and
cardiovascular diseases and reduced GFR in a middle-aged working
population in Taiwan. Am J Kidney Dis
2008; 52: 1051–1060.
235. Rifkin DE, Shlipak MG, Katz R et al. Rapid kidney function decline and
mortality risk in older adults. Arch Intern Med 2008; 168: 2212–2218.
236. Appel LJ, Wright JT, Jr., Greene T et al. Intensive blood-pressure control
in hypertensive chronic kidney disease. N Engl J Med 2010; 363: 918–929.
237. Hunsicker LG, Adler S, Caggiula A et al. Predictors of the progression
of renal disease in the Modification of Diet in Renal Disease Study.
Kidney Int 1997; 51: 1908–1919.
238. Menon V, Wang X, Sarnak MJ et al. Long-term outcomes in nondiabetic
chronic kidney disease. Kidney Int 2008; 73: 1310–1315.
239. Adler AI, Stevens RJ, Manley SE et al. Development and progression of
nephropathy in type 2 diabetes: the United Kingdom Prospective
Diabetes Study (UKPDS 64). Kidney Int 2003; 63: 225–232.
240. Caramori ML, Fiore tto P, Mauer M. Enhancing the predictive value of
urinary albumin for diabetic nephropathy. J Am Soc Nephrol 2006; 17:
339–352.
241. Hoefield RA, Kalra PA, Baker PG et al. The use of eGFR and ACR to predict
decline in renal function in people with diabetes. Nephrol Dial Transplant
2011; 26: 887–892.
242. Rosolowsky ET, Skupien J, Smiles AM et al. Risk for ESRD in type 1
diabetes remains high despite renoprotection. J Am Soc Nephrol 2011;
22: 545–553.
243. Hemmelgarn BR, Clement F, Manns BJ et al. Overview of the Alberta
Kidney Disease Network. BMC Nephrol 2009; 10: 30.
244. Turin TC, Coresh J, Tonelli M et al. One-year change in kidney function is
associated with an increased mortality risk. Am J Nephrol 2012; 36:
41–49.
245. Turin TC, Coresh J, Tonelli M et al. Short-term change in kidney function
and risk of end-stage renal disease. Nephrol Dial Transplant 2012; 27:
3835–3843.
246. Schmieder RE, Mann JF, Schumacher H et al. Changes in albuminuria
predict mortality and morbidity in patients with vascular disease. JAm
Soc Nephrol 2011; 22: 1353–1364.
247. Li L, Astor BC, Lewis J et al.
Longitudinal Progression Trajectory of GFR
Among Patients With CKD. Am J Kidney Dis 2012; 59: 504–512.
248. O’Hare AM, Batten A, Burrows NR et al. Trajectories of Kidney Function
Decline in the 2 Years Before Initiation of Long-term Dialysis. Am J Kidney
Dis 2012; 59: 513–522.
249. Leblanc M, Kellum JA, Gibney RT et al. Risk factors for acute renal failure:
inherent and modifiable risks. Curr Opin Crit Care 2005; 11: 533–536.
250. Naughton CA. Drug-induced nephrotoxicity. Am Fam Physician 2008; 78:
743–750.
251. Pannu N, Nadim MK. An overview of drug-induced acute kidney injury.
Crit Care Med 2008; 36: S216–223.
252. Solomon R, Dauerman HL. Contrast-induced acute kidney injury.
Circulation 2010; 122: 2451–2455.
253. Black C, Sharma P, Scotland G et al. Early referral strategies for
management of people with markers of renal disease: a systematic
review of the evidence of clinical effectiveness, cost-effectiveness and
economic analysis. Health Technol Assess 2010; 14: 1–184.
254. Bang H, Mazumdar M, Newman G et al. Screening for kidney disease in
vascular patients: SCreening for Occult REnal Disease (SCORED)
experience. Nephrol Dial Transplant 2009; 24: 2452–2457.
255. Johnson ES, Smith DH, Thorp ML et al. Predicting the risk of end-stage
renal disease in the population-based setting: a retrospective case-
control study. BMC Nephrol 2011; 12: 17.
256. Wakai K, Kawamura T, Endoh M et al. A scoring system to predict renal
outcome in IgA nephropathy: from a nationwide prospective study.
Nephrol Dial Transplant 2006; 21: 2800–2808.
257. Keane WF, Zhang Z, Lyle PA et al. Risk scores for predicting outcomes in
patients with type 2 diabetes and nephropathy: the RENAAL study. Clin J
Am Soc Nephrol 2006; 1: 761–767.
258. Fine EJ, Blaufox MD. Prediction rule for renal artery stenosis. Ann Intern
Med 1999; 131: 227–228.
259. Kshirsagar AV, Bang H, Bomback AS et al. A simple algorithm to predict
incident kidney disease. Arch Inter n Med 2008; 168: 2466–2473.
260. Tangri N, Stevens LA, Griffith J et al. A predictive model for progres-
sion of chronic kidney disease to kidney failure. JAMA 2011; 305:
1553–1559.
261. Halbesma N, Jansen DF, Heymans MW et al. Development and validation
of a general population renal risk score. Clin J Am Soc Nephrol 2011; 6:
1731–1738.
262. National Kidney Foundation. KDOQI Clinical Practice Guideline for
Diabetes and CKD: 2012 Update. Am J Kidney Dis 2012; 60: 850–886.
140 Kidney International Supplements (2013) 3, 136–150
references

263. Mittalhenkle A, Stehman-Breen CO, Shlipak MG et al. Cardiovascular risk
factors and incident acute renal failure in older adults: the cardiovascular
health study. Clin J Am Soc Nephrol 2008; 3: 450–456.
264. Uchino S, Kellum JA, Bellomo R et al. Acute renal failure in critically ill
patients: a multinational, multicenter study. JAMA 2005; 294: 813–818.
265. Hoste EA, Lameire NH, Vanholder RC et al. Acute renal failure in patients
with sepsis in a surgical ICU: predictive factors, incidence, comorbidity,
and outcome. J Am Soc Nephrol 2003; 14: 1022–1030.
266. McCullough PA, Wolyn R, Rocher LL et al. Acute renal failure after
coronary intervention: incidence, risk factors, and relationship to
mortality. Am J Med 1997; 103: 368–375.
267. Mehran R, Aymong ED, Nikolsky E et al. A simple risk score for prediction
of contrast-induced nephropathy after percutaneous coronary inter-
vention: development and initial validation. J Am Coll Cardiol 2004; 44:
1393–1399.
268. Mehta RH, Grab JD, O’Brien SM et al. Bedside tool for predicting the risk
of postoperative dialysis in patients undergoing cardiac surgery.
Circulation 2006; 114: 2208–2216.
269. Thakar CV, Worley S, Arrigain S et al. Influence of renal dysfunction on
mortality after cardiac surgery: modifying effect of preoperative renal
function. Kidney Int 2005; 67: 1112–1119.
270. Waikar SS, Liu KD, Chertow GM. Diagnosis, epidemiology and outcomes
of acute kidney injury. Clin J Am Soc Nephrol 2008; 3: 844–861.
271. Yegenaga I, Hoste E, Van Biesen W et al. Clinical characteristics of
patients developing ARF due to sepsis/systemic inflammatory response
syndrome: results of a prospective study. Am J Kidney Dis 2004; 43:
817–824.
272. Parfrey PS, Griffiths SM, Barrett BJ et al. Contrast material-induced renal
failure in patients with diabetes mellitus, renal insufficiency, or both.
A prospective controlled study. N Engl J Med 1989; 320: 143–149.
273. Browner WS, Li J, Mangano DT. In-hospital and long-term mortality in
male veterans following noncardiac surgery. The Study of Perioperative
Ischemia Research Group. JAMA 1992; 268: 228–232.
274. Hou SH, Bushinsky DA, Wish JB et al. Hospital-acquired renal
insufficiency: a prospective study. Am J Med 1983; 74: 243–248.
275. Singh P, Rifkin DE, Blantz RC. Chronic kidney disease: an inherent risk
factor for acute kidney injury? Clin J Am Soc Nephrol 2010; 5: 1690–1695.
276. Lafrance JP, Djurdjev O, Levin A. Incidence and outcomes of acute
kidney injury in a referred chronic kidney disease cohort. Nephrol Dial
Transplant 2010; 25: 2203–2209.
277. Chapin E, Zhan M, Hsu VD et al. Adverse safety events in chronic kidney
disease: the frequency of ‘‘multiple hits’’. Clin J Am Soc Nephrol 2010; 5:
95–101.
278. Chertow GM, Christiansen CL, Cleary PD et al. Prognostic stratification in
critically ill patients with acute renal failure requiring dialysis. Arch Intern
Med 1995; 155: 1505–1511.
279. Chertow GM, Soroko SH, Paganini EP et al. Mortality after acute renal
failure: models for prognostic stratification and risk adjustment. Kidney
Int 2006; 70: 1120–1126.
280. Mehta RL, Pascual MT, Gruta CG et al. Refining predictive models in
critically ill patients with acute renal failure. J Am Soc Nephrol 2002; 13:
1350–1357.
281. Paganini EP, Larive B, Kanagasundaram NS. Severity scores and
outcomes with acute renal failure in the ICU setting. Contrib Nephrol
2001: 181–195.
282. Uchino S, Bellomo R, Morimatsu H et al. External validation of severity
scoring systems for acute renal failure using a multinational database.
Crit Care Med 2005; 33: 1961–1967.
283. Waikar SS, Curhan GC, Wald R et al. Declining mortality in patients
with acute renal failure, 1988 to 2002. J Am Soc Nephrol 2006; 17:
1143–1150.
284. Khosla N, Soroko SB, Chertow GM et al. Preexisting chronic kidney
disease: a potential for improved outcomes from acute kidney injury.
Clin J Am Soc Nephrol 2009; 4: 1914–1919.
285. Fouque D, Laville M. Low protein diets for chronic kidney disease in non
diabetic adults. Cochrane Database Syst Rev 2009: CD001892.
286. Fouque D, Laville M, Boissel JP et al. Controlled low protein diets in
chronic renal insufficiency: meta-analysis. BMJ 1992; 304: 216–220.
287. Kasiske BL, Lakatua JD, Ma JZ et al. A meta-analysis of the effects
of dietary protein restriction on the rate of decline in renal function.
Am J Kidney Dis 1998; 31: 954–961.
288. Pedrini MT, Lev ey AS, Lau J et al. The effect of dietary protein restriction
on the progression of diabetic and nondiabetic renal diseases: a meta-
analysis. Ann Intern Med 1996; 124: 627–632.
289. Robertson L, Waugh N, Robertson A. Protein restriction for diabetic renal
disease. Cochra ne Database Syst Rev
2009: CD002181.
290. Menon V, Kopple JD, Wang X et al. Effect of a very low-protein diet on
outcomes: long-term follow-up of the Modification of Diet in Renal
Disease (MDRD) Study. Am J Kidney Dis 2009; 53: 208–217.
291. Knight EL, Stampfer MJ, Hankinson SE et al. The impact of protein intake
on renal function decline in women with normal renal function or mild
renal insufficiency. Ann Intern Med 2003; 138: 460–467.
292. Chaturvedi S, Jones C. Protein restriction for children with chronic renal
failure. Cochrane Database Syst Rev 2007: CD006863.
293. National Kidney Foundation. KDOQI clinical practice guideline for
diabetes and chronic kidney disease: 2012 Update. Am J Kidney Dis
2012; 60: 850–886.
294. Patel A, MacMahon S, Chalmers J et al. Intensive blood glucose control
and vascular outcomes in patients with type 2 diabetes. N Engl J Med
2008; 358: 2560–2572.
295. Ismail-Beigi F, Craven T, Banerji MA et al. Effect of intensive treatment of
hyperglycaemia on microvascular outcomes in type 2 diabetes: an
analysis of the ACCORD randomised trial. Lancet 2010; 376: 419–430.
296. Duckworth W, Abraira C, Moritz T et al. Glucose control and vascular
complications in veterans with type 2 diabetes. N Engl J Med 2009; 360:
129–139.
297. Nathan DM, Zinman B, Cleary PA et al. Modern-day clinical course of type
1 diabetes mellitus after 30 years’ duration: the diabetes control and
complications trial/epidemiology of diabetes interventions and compli-
cations and Pittsburgh epidemiology of diabetes complications experi-
ence (1983–2005). Arch Intern Med 2009; 169: 1307–1316.
298. UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose
control with sulphonylureas or insulin compared with conventional
treatment and risk of complications in patients with type 2 diabetes
(UKPDS 33). Lancet 1998; 352: 837–853.
299. Ichikawa H, Nagake Y, Takahashi M et al. What is the best index of
glycemic control in patients with diabetes mellitus on hemodialysis?
Nihon Jinzo Gakkai Shi 1996; 38: 305–308.
300. Joy MS, Cefalu WT, Hogan SL et al. Long-term glycemic control
measurements in diabetic patients receiving hemodialysis. Am J Kidney
Dis 2002; 39: 297–307.
301. Nakao T, Matsumoto H, Okada T et al. Influence of erythropoietin
treatment on hemoglobin A1c levels in patients with chronic renal
failure on hemodialysis. Intern Med 1998; 37: 826–830.
302. Ng JM, Cooke M, Bhandari S et al. The effect of iron and erythropoietin
treatment on the A1C of patients with diabetes and chronic kidney
disease. Diabetes Care 2010;
33: 2310–2313.
303. Shima K, Chujo K, Yamada M et al. Lower value of glycated haemoglobin
relative to glycaemic control in diabetic patients with end-stage renal
disease not on haemodialysis. Ann Clin Biochem 2012; 49: 68–74.
304. Vos FE, Schollum JB, Coulter CV et al. Assessment of markers of
glycaemic control in diabetic patients with chronic kidney disease using
continuous glucose monitoring. Nephrology (Carlton) 2012; 17: 182–188.
305. American Diabetes Association. Executive summary: Standards of
medical care in diabetes–2012. Diabetes Care 2012; 35 (Suppl 1): S4–S10.
306. Jones-Burton C, Mishra SI, Fink JC et al. An in-depth review of the
evidence linking dietary salt intake and progression of chronic kidney
disease. Am J Nephrol 2006; 26: 268–275.
307. Swift PA, Markandu ND, Sagnella GA et al. Modest salt reduction reduces
blood pressure and urine protein excretion in black hypertensives: a
randomized control trial. Hypertension 2005; 46: 308–312.
308. Hoffmann IS, Cubeddu LX. Increased blood pressure reactivity to dietary
salt in patients with the metabolic syndrome. J Hum Hypertens 2007; 21:
438–444.
309. Bellizzi V, Di Iorio BR, De Nicola L et al. Very low protein diet
supplemented with ketoanalogs improves blood pressure control in
chronic kidney disease. Kidney Int 2007; 71: 245–251.
310. Slagman MC, Waanders F, Hemmelder MH et al. Moderate dietary
sodium restriction added to angiotensin converting enzyme inhibition
compared with dual blockade in lowering proteinuria and blood
pressure: randomised controlled trial. BMJ 2011; 343: d4366.
311. Bellomo G, Venanzi S, Verdura C et al. Association of uric acid with
change in kidney function in healthy normotensive individuals. Am J
Kidney Dis 2010; 56: 264–272.
312. Iseki K, Ikemiya Y, Inoue T et al. Significance of hyperuricemia as a risk
factor for developing ESRD in a screened cohort. Am J Kidney Dis 2004;
44: 642–650.
313. Mok Y, Lee SJ, Kim MS et al. Serum uric acid and chronic kidney disease:
the Severance cohort study. Nephrol Dial Transplant 2012; 27:
1831–1835.
314. Wen CP, David Cheng TY, Chan HT et al. Is high serum uric acid a risk
marker or a target for treatment? Examination of its independent effect
Kidney International Supplements (2013) 3, 136–150 141
references

in a large cohort with low cardiovascular risk. Am J Kidney Dis 2010; 56:
273–288.
315. Yamada T, Fukatsu M, Suzuki S et al. Elevated serum uric acid predicts
chronic kidney disease. Am J Med Sci 2011; 342: 461–466.
316. Goicoechea M, de Vinuesa SG, Verdalles U et al. Effect of allopurinol in
chronic kidney disease progression and cardiovascular risk. Clin J Am Soc
Nephrol 2010; 5: 1388–1393.
317. Siu YP, Leung KT, Tong MK et al. Use of allopurinol in slowing the
progression of renal disease through its ability to lower serum uric acid
level. Am J Kidney Dis 2006; 47: 51–59.
318. Kanbay M, Huddam B, Azak A et al. A randomized study of allopurinol on
endothelial function and estimated glomular filtration rate in asympto-
matic hyperuricemic subjects with normal renal function.
Clin J Am Soc Nephrol 2011; 6: 1887–1894.
319. Kanbay M, Ozkara A, Selcoki Y et al. Effect of treatment of hyperuricemia
with allopurinol on blood pressure, creatinine clearence, and proteinuria
in patients with normal renal functions. Int Urol Nephrol 2007; 39:
1227–1233.
320. Kao MP, Ang DS, Gandy SJ et al. Allopurinol benefits left ventricular ma ss
and endothelial dysfunction in chronic kidney disease. J Am Soc Nephrol
2011; 22: 1382–1389.
321. Saito J, Matsuzawa Y, Ito H et al. The alkalizer citrate reduces serum uric
Acid levels and improves renal function in hyperuricemic patients
treated with the xanthine oxidase inhibitor allopurinol. Endocr Res 2010;
35: 145–154.
322. Malaguarnera M, Vacante M, Russo C et al. Asingledoseofrasburicasein
elderly patients with hyperuricaemia reduces serum uric acid levels and
improves renal function. Expert Opin Pharmacother 2009; 10: 737–742.
323. Miao Y, Ottenbros SA, Laverman GD et al. Effect of a reduction in uric
acid on renal outcomes during losartan treatment: a post hoc analysis of
the reduction of endpoints in non-insulin-dependent diabetes mellitus
with the Angiotensin II Antagonist Losartan Trial. Hypertension 2011; 58:
2–7.
324. Johansen KL. Exercise and chronic kidney disease: current recommenda-
tions. Sports Med 2005; 35: 485–499.
325. Johansen KL, Chertow GM, Ng AV et al. Physical activity levels in patients
on hemodialysis and healthy sedentary controls. Kidney Int 2000; 57:
2564–2570.
326. Padilla J, Krasnoff J, Da Silva M et al. Physical functioning in patients with
chronic kidney disease.
J Nephrol 2008; 21: 550–559.
327. Beddhu S, Baird BC, Zitterkoph J et al. Physical activity and mortality in
chronic kidney disease (NHANES III). Clin J Am Soc Nephrol 2009; 4:
1901–1906.
328. Churchill DN, Torrance GW, Taylor DW et al. Measurement of quality of
life in end-stage renal disease: the time trade-off approach. Clin Invest
Med 1987; 10: 14–20.
329. DeOreo PB. Hemodialysis patient-assessed functional health status
predicts continued survival, hospitalization, and dialysis-attendance
compliance. Am J Kidney Dis 1997; 30: 204–212.
330. Booth FW, Gordon SE, Carlson CJ et al. Waging war on modern chronic
diseases: primary prevention through exercise biology. J Appl Physiol
2000; 88: 774–787.
331. Dickinson HO, Mason JM, Nicolson DJ et al. Lifestyle interventions to
reduce raised blood pressure: a systematic review of randomized
controlled trials. J Hypertens 2006; 24: 215–233.
332. Stewart KJ. Exercise training and the cardiovascular consequences of
type 2 diabetes and hypertension: plausible mechanisms for improving
cardiovascular health. JAMA 2002; 288: 1622–1631.
333. Daul AE, Schafers RF, Daul K et al. Exercise during hemodialysis. Clin
Nephrol 2004; 61 (Suppl 1): S26–30.
334. Deligiannis A. Cardiac adaptations following exercise training in
hemodialysis patients. Clin Nephrol 2004; 61 (Suppl 1): S39–45.
335. Liu SH, C. LC, Yeh SH et al. Effect of exercise training on hemodialysis.
Journal of the Formosan Medical Association 2002; 6: 129–142.
336. Mustata S, Chan C, Lai V et al. Impact of an exercise program on arterial
stiffness and insulin resistance in hemodialysis patients. J Am Soc
Nephrol 2004; 15: 2713–2718.
337. Ouzouni S, Kouidi E, Sioulis A et al. Effects of intradialytic exercise
training on health-related quality of life indices in haemodialysis
patients. Clin Rehabil 2009; 23: 53–63.
338. Storer TW, Casaburi R, Sawelson S et al. Endurance exercise training
during haemodialysis improves strength, power, fatigability and physical
performance in maintenance haemodialysis patients. Nephrol Dial
Transplant 2005; 20: 1429–1437.
339. Vaitkevicius PV, Fleg JL, Engel JH et al. Effects of age and aerobic capacity
on arterial stiffness in healthy adults. Circulation 1993; 88: 1456–1462.
340. Mustata S, Groeneveld S, Davidson W et al. Effects of exercise training on
physical impairment, arterial stiffness and health-related quality of life in
patients with chronic kidney disease: a pilot study. Int Urol Nephrol 2011;
43: 1133–1141.
341. Szromba C, Thies MA, Ossman SS. Advancing chronic kidney disease
care: new imperatives for recognition and intervention. Nephrol Nurs J
2002; 29: 547–559.
342. Chen PY, Huang YC, Kao YH et al. Effects of an exercise program on
blood biochemical values and exercise stage of chronic kidney disease
patients. J Nurs Res 2010; 18: 98–107.
343. Tobita I, Suzuki S, Kobayashi T et al. A programme to encourage
participation of haemodialysis patients in an exercise regimen. J Ren Care
2009; 35: 48–53.
344. van Vilsteren MC, de Greef MH, Huisman RM. The effects of a low-to-
moderate intensity pre-conditioning exercise programme linked with
exercise counselling for sedentary haemodialysis patients in The
Netherlands: results of a randomized clinical trial. Nephrol Dial Transplant
2005; 20: 141–146.
345. Kosmadakis GC, John SG, Clapp EL et al. Benefits of regular walking
exercise in advanced pre-dialysis chronic kidney disease. Nephrol Dial
Transplant 2012; 27: 997–1004.
346. Hall JE, Crook ED, Jones DW et al. Mechanisms of obesity-associated
cardiovascular and renal disease. Am J Med Sci 2002; 324: 127–137.
347. Wahba IM, Mak RH. Obesity and obesity-initiated metabolic syndrome:
mechanistic links to chronic kidney disease. Clin J Am Soc Nephrol 2007;
2: 550–562.
348. Wang Y, Chen X, Song Y et al. Association between obesity and kidney
disease: a systematic review and meta-analysis. Kidney Int 2008; 73:
19–33.
349. Hobbs H, Farmer C, Irving J et al. Is high body mass index independently
associated with diminished glomerular filtration rate? An epidemiologi-
cal study. J Ren Care 2011; 37: 148–154.
350. Mohsen A, Brown R, Hoefield R et al. Body mass index has no effect on
rate of progression of chronic kidney disease in subjects with type 2
diabetes mellitus. J Nephrol 2012: 25: 384–393.
351. Burton JO, Gray LJ, Webb DR et al. Association of anthropometric obesity
measures with chronic kidney disease risk in a non-diabetic patient
population. Nephrol Dial Transplant 2012; 27: 1860–1866.
352. Kasiske BL, Napier J. Glomerular sclerosis in patients with massive
obesity. Am J Nephrol
1985; 5: 45–50.
353. Hsu CY, McCulloch CE, Iribarren C et al. Body mass index and risk for end-
stage renal disease. Ann Intern Med 2006; 144: 21–28.
354. Navaneethan SD, Yehnert H, Moustarah F et al. Weight loss interventions
in chronic kidney disease: a systematic review and meta-analysis. Clin J
Am Soc Nephrol 2009; 4: 1565–1574.
355. Afshinnia F, Wilt TJ, Duval S et al. Weight loss and proteinuria: systematic
review of clinical trials and comparative cohorts. Nephrol Dial Transplant
2010; 25: 1173–1183.
356. Orth SR, Hallan SI. Smoking: a risk factor for progression of chronic
kidney disease and for cardiovascular morbidity and mortality in renal
patients–absence of evidence or evidence of absence? Clin J Am Soc
Nephrol 2008; 3: 226–236.
357. Jungers P, Massy ZA, Nguyen Khoa T et al. Incidence and risk factors of
atherosclerotic cardiovascular accidents in predialysis chronic renal
failure patients: a prospective study. Nephrol Dial Transplant 1997; 12:
2597–2602.
358. Muntner P, He J, Astor BC et al. Traditional and nontraditional risk factors
predict coronary heart disease in chronic kidney disease: results from the
atherosclerosis risk in communities study. J Am Soc Nephrol 2005; 16:
529–538.
359. Myllymaki J, Syrjanen J, Helin H et al. Vascular diseases and their risk
factors in IgA nephropathy. Nephrol Dial Transplant 2006; 21: 1876–1882.
360. Shlipak MG, Fried LF, Cushman M et al. Cardiovascular mortality risk in
chronic kidney disease: comparison of traditional and novel risk factors.
JAMA 2005; 293: 1737–1745.
361. Chase HP, Garg SK, Marshall G et al. Cigarette smoking increases the risk
of albuminuria among subjects with type I diabetes. JAMA 1991; 265:
614–617.
362. Gambaro G, Bax G, Fusaro M et al. Cigarette smoking is a risk factor for
nephropathy and its progression in type 2 diabetes mellitus. Diabetes
Nutr Metab 2001; 14: 337–342.
363. Hallan SI, Orth SR. Smoking is a risk factor in the progression to kidney
failure. Kidney Int 2011; 80: 516–523.
364. Sawicki PT, Didjurgeit U, Muhlhauser I et al. Smoking is associated
with progression of diabetic nephropathy. Diabetes Care 1994; 17:
126–131.
142 Kidney International Supplements (2013) 3, 136–150
references

365. Sung RS, Althoen M, Howell TA et al. Excess risk of renal allograft
loss associated with cigarette smoking. Transplantation 2001; 71:
1752–1757.
366. Inker LA, Coresh J, Levey AS et al. Estimated GFR, albuminuria, and
complications of chronic kidney disease. J Am Soc Nephrol 2011; 22:
2322–2331.
367. Levin A, Bakris GL, Molitch M et al. Prevalence of abnormal serum
vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney
disease: results of the study to evaluate early kidney disease. Kidney Int
2007; 71: 31–38.
368. World Health Organization. Worldwide prevalence of anaemia 1993–2005:
WHO global database on anaemia. de Benoist B, McLean E, Egli I,
and Cogswell M (eds), 2008.
369. Beall CM, Goldstein MC. Hemoglobin concentration of pastoral nomads
permanently resident at 4,850–5,450 meters in Tibet. Am J Phys
Anthropol 1987; 73: 433–438.
370. Cresanta JL, Croft JB, Webber LS et al. Racial difference in hemoglobin
concentration of young adults. Prev Med 1987; 16: 659–669.
371. Meyers LD, Habicht JP, Johnson CL. Components of the difference in
hemoglobin concentrations in blood between black and white women
in the United States. Am J Epidemiol 1979; 109: 539–549.
372. Pan WH, Habicht JP. The non-iron-deficiency-related difference in
hemoglobin concentration distribution between blacks and whites
and between men and women. Am J Epidemiol 1991; 134: 1410–1416.
373. Centers for Disease Control and Prevention. CDC criteria for anemia in
children and childbearing-aged women. MMWR Morb Mortal Wkly Rep
1989; 38: 400–404.
374. Levin A, Djurdjev O, Thompson C et al. Canadian randomized trial of
hemoglobin maintenance to prevent or delay left ventricular mass
growth in patients with CKD. Am J Kidney Dis 2005; 46: 799–811.
375. Pfeffer MA, Burdmann EA, Chen CY et al. A trial of darbepoetin alfa in
type 2 diabetes and chronic kidney disease. N Engl J Med 2009; 361:
2019–2032.
376. Roger SD, McMahon LP, Clarkson A et al. Effects of early and late
intervention with epoetin alpha on left ventricular mass among patients
with chronic kidney disease (stage 3 or 4): results of a randomized
clinical trial. J Am Soc Nephrol 2004; 15: 148–156.
377. Moranne O, Froissart M, Rossert J et al. Timing of onset of CKD-related
metabolic complications. J Am Soc Nephrol 2009; 20: 164–171.
378. Vassalotti JA, Uribarri J, Chen SC et al. Trends in mineral metabolism:
Kidney Early Evaluation Program (KEEP) and the National Health and
Nutrition Examination Survey (NHANES) 1999–2004. Am J Kidney Dis
2008; 51: S56–68.
379. Stevens LA, Djurdjev O, Cardew S et al. Calcium, phosphate, and
parathyroid hormone levels in combination and as a function of dialysis
duration predict mortality: evidence for the complexity of the
association between mineral metabolism and outcomes. J Am Soc
Nephrol 2004; 15: 770–779.
380. Gutierrez OM, Isakova T, Andress DL et al. Prevalence and severity of
disordered mineral metabolism in Blacks with chronic kidney disease.
Kidney Int 2008; 73: 956–962.
381. Gutierrez OM, Farwell WR, Kermah D et al. Racial differences in the
relationship between vitamin D, bone mineral density, and parathyroid
hormone in the National Health and Nutrition Examination Survey.
Osteoporos Int 2011; 22: 1745–1753.
382. National Kidney Foundation. KDOQI clinical practice guideline for
nutrition in children with CKD: 2008 update. Am J Kidney Dis 2009; 53:
S1–124.
383. Nickolas TL, Leonard MB, Shane E. Chronic kidney disease and bone
fracture: a growing concern. Kidney Int 2008; 74: 721–731.
384. Bacchetta J, Boutroy S, Vilayphiou N et al. Early impairment of
trabecular microarchitecture assessed with HR-pQCT in patients
with stage II-IV chronic kidney disease. J Bone Miner Res 2010; 25:
849–857.
385. Nickolas TL, Stein E, Cohen A et al. Bone mass and microarchitecture
in CKD patients with fracture. J Am Soc Nephrol 2010; 21: 1371–1380.
386. Nickolas TL, Cremers S, Zhang A et al. Discriminants of prevalent
fractures in chronic kidney disease. J Am Soc Nephrol 2011; 22:
1560–1572.
387. Palmer SC, Hayen A, Macaskill P et al. Serum levels of phosphorus,
parathyroid hormone, and calcium and risks of death and cardiovascular
disease in individuals with chronic kidney disease: a systematic review
and meta-analysis. JAMA 2011; 305: 1119–1127.
388. Adeney KL, Siscovick DS, Ix JH et al. Association of serum phosphate with
vascular and valvular calcification in moderate CKD. J Am Soc Nephrol
2009; 20: 381–387.
389. Moe SM, Zidehsarai MP, Chambers MA et al. Vegetarian compared with
meat dietary protein source and phosphorus homeostasis in chronic
kidney disease. Clin J Am Soc Nephrol 2011; 6: 257–264.
390. Sigrist MK, Chiarelli G, Lim L et al. Early initiation of phosphate lowering
dietary therapy in non-dialysis chronic kidney disease: a critical review.
J Ren Care 2009; 35 (Suppl 1): 71–78.
391. Lynch KE, Lynch R, Curhan GC et al. Prescribed dietary phosphate
restriction and survival among hemodialysis patients. Clin J Am Soc
Nephrol 2011; 6: 620–629.
392. Navaneethan SD, Palmer SC, Vecchio M et al. Phosphate binders for
preventing and treating bone disease in chronic kidney disease patients.
Cochrane Database Syst Rev 2011: CD006023.
393. Mehrotra R, Kermah D, Budoff M et al. Hypovitaminosis D in chronic
kidney disease. Clin J Am Soc Nephrol 2008; 3: 1144–1151.
394. Navaneethan SD, Schold JD, Arrigain S et al. Low 25-hydroxyvitamin D
levels and mortality in non-dialysis-dependent CKD. Am J Kidney Dis
2011; 58: 536–543.
395. Kandula P, Dobre M, Schold JD et al. Vitamin D supplementation in
chronic kidney disease: a systematic review and meta-analysis of
observational studies and randomized controlled trials. Clin J Am Soc
Nephrol 2011; 6: 50–62.
396. Palmer SC, McGregor DO, Craig JC et al. Vitamin D compounds for
people with chronic kidney disease not requiring dialysis. Cochrane
Database Syst Rev 2009: CD008175.
397. Miller PD. The kidney and bisphosphonates. Bone 2011; 49: 77–81.
398. Torregrosa JV, Ramos AM. [Use of bisphosphonates in chronic kidney
disease]. Nefrologia 2010; 30: 288–296.
399. Bhan I, Dubey A, Wolf M. Diagnosis and management of mineral
metabolism in CKD. J Gen Intern Med 2010; 25: 710–716.
400. Courtney AE, Leonard N, McCloskey MC et al. Bisphosphonate
prescribing in chronic kidney disease. J R Coll Physicians Edinb 2009;
39: 4–9.
401. Ayus JC, Krothapalli RK. Effect of bicarbonate administration on cardiac
function. Am J Med 1989; 87: 5–6.
402. Bailey JL, Wang X, England BK et al. The acidosis of chronic renal failure
activates muscle proteolysis in rats by augmenting transcription of
genes encoding proteins of the ATP-dependent ubiquitin-p roteasome
pathway. J Clin Invest 1996; 97: 1447–1453.
403. Domrongkitchaiporn S, Pongskul C, Sirikulchayanonta V et al. Bone
histology and bone mineral density after correction of acidosis in distal
renal tubular acidosis. Kidney Int 2002; 62: 2160–2166.
404. Kovesdy CP, Anderson JE, Kalantar-Zadeh K. Association of serum
bicarbonate levels with mortality in patients with non-dialysis-depen-
dent CKD. Nephrol Dial Transplant 2009; 24: 1232–1237.
405. Mak RH. Effect of metabolic acidosis on insulin action and secretion in
uremia. Kidney Int 1998; 54: 603–607.
406. Menon V, Tighiouart H, Vaughn NS et al. Serum bicarbonate and
long-term outcomes in CKD. Am J Kidney Dis 2010; 56: 907–914.
407. Mitch WE, Price SR. Mechanisms activated by kidney disease and the loss
of muscle mass. Am J Kidney Dis 2001; 38: 1337–1342.
408. Navaneethan SD, Schold JD, Arrigain S et al. Serum bicarbonate and
mortality in stage 3 and stage 4 chronic kidney disease. Clin J Am Soc
Nephrol 2011; 6: 2395–2402.
409. Raphael KL, Wei G, Baird BC et al. Higher serum bicarbonate levels within
the normal range are associated with better survival and renal outcomes
in African Americans. Kidney Int 2011; 79: 356–362.
410. Shah SN, Abramowitz M, Hostetter TH et al. Serum bicarbonate levels
and the progression of kidney disease: a cohort study. Am J Kidney Dis
2009; 54: 270–277.
411. Bright R. Reports of Medical Cases, Selected with a View of Illustrating the
Symptoms and Cure of Diseases by a Reference to Morbid Anatomy,
Volume 1. London: Brown & Green. 1827.
412. Osman AA. The value of alkalis in the treatment of chronic nephritis.
Lancet 1930; 2: 945–959.
413. Lyon DM, Dunlop DM, Stewart CP. The alkaline treatment of chronic
nephritis. Lancet 1931; 2: 1009–1013.
414. Rustom R, Grime JS, Costigan M et al. Oral sodium bicarbonate reduces
proximal renal tubular peptide catabolism, ammoniogenesis, and
tubular damage in renal patients. Ren Fail 1998; 20: 371–382.
415. Mathur RP, Dash SC, Gupta N et al. Effects of correction of metabolic
acidosis on blood urea and bone metabolism in patients with mild to
moderate chronic kidney disease: a prospective randomized single blind
controlled trial. Ren Fail 2006; 28: 1–5.
416. de Brito-Ashurst I, Varagunam M, Raftery MJ et al. Bicarbonate
supplementation slows progression of CKD and improves nutritional
status. J Am Soc Nephrol 2009; 20: 2075–2084.
Kidney International Supplements (2013) 3, 136–150 143
references

417. Phisitkul S, Khanna A, Simoni J et al. Amelioration of metabolic acidosis
in patients with low GFR reduced kidney endothelin production and
kidney injury, and better preserved GFR. Kidney Int 2010; 77: 617–623.
418. Husted FC, Nolph KD, Maher JF. NaHCO3 and NaCl tolerance in chronic
renal failure. J Clin Invest 1975; 56: 414–419.
419. Husted FC, Nolph KD. NaHCO3 and NaCl tolerance in chronic renal
failure II. Clin Nephrol 1977; 7: 21–25.
420. Hallan SI, Dahl K, Oien CM et al. Screening strategies for chronic kidney
disease in the general population: follow-up of cross sectional health
survey. BMJ 2006; 333: 1047.
421. McCullough PA, Li S, Jurkovitz CT et al. Chronic kidney disease,
prevalence of premature cardiovascular disease, and relationship to
short-term mortality. Am Heart J 2008; 156: 277–283.
422. Anavekar NS, McMurray JJ, Velazquez EJ et al. Relation between renal
dysfunction and cardiovascular outcomes after myocardial infarction.
N Engl J Med 2004; 351: 1285–1295.
423. Shlipak MG, Heidenreich PA, Noguchi H et al. Association of renal
insufficiency with treatment and outcomes after myocardial infarction in
elderly patients. Ann Intern Med 2002; 137: 555–562.
424. Wright RS, Reeder GS, Herzog CA et al. Acute myocardial infarction and
renal dysfunction: a high-risk combination. Ann Intern Med 2002; 137:
563–570.
425. Sarnak MJ. Cardiovascular complications in chronic kidney disease. Am J
Kidney Dis 2003; 41: 11–17.
426. Hemmelgarn BR, Manns BJ, Lloyd A et al. Relation between kidney
function, proteinuria, and adverse outcome s. JAMA 2010; 303: 423–429.
427. Klausen K, Borch-J ohnsen K, Feldt-Rasmussen B et al. Very low levels of
microalbuminuria are associated with increased risk of coronary heart
disease and death independently of renal function, hypertension, and
diabetes. Circulation 2004; 110: 32–35.
428. Gerstein HC, Mann JF, Yi Q et al. A lbuminuria and risk of cardiovascular
events, death, and heart failure in diabetic and nondiabetic individuals.
JAMA 2001; 286: 421–426.
429. Hallan S, Astor B, Romundstad S et al. Association of kidney function and
albuminuria with cardiovascular mortality in older vs younger indivi-
duals: The HUNT II Study. Arch Intern Med 2007; 167: 2490–2496.
430. Ibsen H, Wachtell K, Olsen MH et al. Albuminuria and cardiovascular risk
in hypertensive patients with left ventricular hypertrophy: the LIFE
Study. Kidney Int Suppl 2004; 92: S56–58.
431. Menon V, Shlipak MG, Wang X et al. Cystatin C as a risk factor for
outcomes in chronic kidney disease. Ann Intern Med 2007; 147: 19–27.
432. Peralta CA, Katz R, Sarnak MJ et al. Cystatin C identifies chronic kidney
disease patients at higher risk for complications. J Am Soc Nephrol 2011;
22: 147–155.
433. Phrommintikul A, Haas SJ, Elsik M et al. Mortality and target
haemoglobin concentrations in anaemic patients with chronic kidney
disease treated with erythropoietin: a meta-analysis. Lancet 2007; 369:
381–388.
434. Strippoli GF, Tognoni G, Navaneethan SD et al. Haemoglobin targets: we
were wrong, time to move on. Lancet 2007; 369: 346–350.
435. Eckardt KU, Scherhag A, Macdougall IC et al. Left ventricular geometry
predicts cardiovascular outcomes associated with anemia correction in
CKD. J Am Soc Nephrol 2009; 20: 2651–2660.
436. Kestenbaum B, Sampson JN, Rudser KD et al. Serum phosphate levels
and mortality risk among people with chronic kidney disease. J Am Soc
Nephrol 2005; 16: 520–528.
437. Groothoff JW, Gruppen MP, Offringa M et al. Mortality and causes of
death of end-stage renal disease in children: a Dutch cohort study.
Kidney Int 2002; 61: 621–629.
438. McDonald SP, Craig JC. Long-term survival of children with end-stage
renal disease. N Engl J Med 2004; 350: 2654–2662.
439. Oh J, Wunsch R, Turzer M et al. Advanced coronary and carotid
arteriopathy in young adults with childhood-onset chronic renal failure.
Circulation 2002; 106: 100–105.
440. Parekh RS, Carroll CE, Wolfe RA et al. Cardiovascular mortality in children
and young adults with end-stage kidney disease. J Pediatr 2002; 141:
191–197.
441. Kavey RE, Allada V, Daniels SR et al. Cardiovascular risk reduction in high-
risk pediatric patients: a scientific statement from the American Heart
Association Expert Panel on Population and Prevention Science; the
Councils on Cardiovascular Disease in the Young, Epidemiology and
Prevention, Nutrition, Physical Activity and Metabolism, High Blood
Pressure Research, Cardiovascular Nursing, and the Kidney in Heart
Disease; and the Interdisciplinary Working Group on Quality of Care and
Outcomes Research: endorsed by the American Academy of Pediatrics.
Circulation 2006; 114: 2710–2738.
442. Wilson AC, Schneider MF, Cox C et al.
Prevalence and correlates of
multiple cardiov ascular risk factors in children with chronic kidney
disease. Clin J Am Soc Nephrol 2011; 6: 2759–2765.
443. Litwin M, Wuhl E, Jourdan C et al. Altered morphologic properties of
large arteries in children with chronic renal failure and after renal
transplantation. J Am Soc Nephrol 2005; 16: 1494–1500.
444. Mitsnefes MM, Kimball TR, Kartal J et al. Cardiac and vascular adaptation
in pediatric patients with chronic kidney disease: role of calcium-
phosphorus metabolism. J Am Soc Nephrol 2005; 16: 2796–2803.
445. Mitsnefes MM. Cardiovascular disease in children with chronic kidney
disease. J Am Soc Nephrol 2012; 23: 578–585.
446. Shroff R, Weaver DJ, Jr., Mitsnefes MM. Cardiovascular complications in
children with chronic kidney disease. Nat Rev Nephrol 2011; 7: 642–649.
447. Halimi JM, Giraudeau B, Vol S et al. Effects of current smoking and
smoking discontinuation on renal function and proteinuria in the
general population. Kidney Int 2000; 58: 1285–1292.
448. Boyce ML, Robergs RA, Avasthi PS et al. Exercise training by individuals
with predialysis renal failure: cardiorespiratory endurance, hypertension,
and renal function. Am J Kidney Dis 1997; 30: 180–192.
449. Baigent C, Landray MJ, Reith C et al. The effects of lowering LDL
cholesterol with simvastatin plus ezetimibe in patients with chronic
kidney disease (Study of Heart and Renal Protection): a randomised
placebo-controlled trial. Lancet 2011; 377: 2181–2192.
450. Jardine MJ, Ninomiya T, Perkovic V et al. Aspirin is beneficial in
hypertensive patients with chronic kidney disease: a post-hoc subgroup
analysis of a randomized controlled trial. J Am Coll Cardiol 2010; 56:
956–965.
451. Angiolillo DJ, Bernardo E, Capodanno D et al. Impact of chronic kidney
disease on platelet function profiles in diabetes mellitus patients with
coronary artery disease taking dual antiplatelet therapy. J Am Coll Cardiol
2010; 55: 1139–1146.
452. Best PJ, Steinhubl SR, Berger PB et al. The efficacy and safety of short-
and long-term dual antiplatelet therapy in patients with mild or
moderate chronic kidney disease: results from the Clopidogrel for the
Reduction of Events During Observation (CREDO) trial. Am Heart J 2008;
155: 687–693.
453. Graham I, Atar D, Borch-Johnsen K et al. European guidelines on
cardiovascular disease prevention in clinical practice: executive sum-
mary. Fourth Joint Task Force of the European Society of Cardiology and
other societies on cardiovascular disease prevention in clinical practice
(constituted by representatives of nine societies and by invited experts).
Eur J Cardiovasc Prev Rehabil 2007; 14 (Suppl 2): E1–40.
454. Hippisley-Cox J, Coupland C, Vinogradova Y et al.
Predicting cardiovas-
cular risk in England and Wales: prospective derivation and validation of
QRISK2. BMJ 2008; 336: 1475–1482.
455. Joint British Societies. JBS 2: Joint British Societies’ guidelines on
prevention of cardiovascular disease in clinical practice. Heart 2005; 91
(Suppl 5): v1–52.
456. Wu Y, Liu X, Li X et al. Estimation of 10-year risk of fatal and nonfatal
ischemic cardio vascular diseases in Chinese adults. Circulation 2006; 114:
2217–2225.
457. Liau SY, Izham MI, Hassali MA et al. A literature review of the
cardiovascular risk-assessment tools: applicability among Asian popula-
tion. Heart Asia 2010; 2: 15–18.
458. Nakayama M, Sato T, Sato H et al. Different clinical outcomes for
cardiovascular events and mortality in chronic kidney disease according
to underlying renal disease: the Gonryo study. Clin Exp Nephrol 2010; 14:
333–339.
459. Irie F, Iso H, Sairenchi T et al. The relationships of proteinuria, serum
creatinine, glomerular filtration rate with cardiovascular disease morta-
lity in Japanese general population. Kidney Int 2006; 69: 1264–1271.
460. Yang JG, Li J, Lu C et al. Chronic kidney disease, all-cause mortality and
cardiovascular mortality among Chinese patients with established
cardiovascular disease. J Atheroscler Thromb 2010; 17: 395–401.
461. Zhang L, Zuo L, Wang F et al. Cardiovascular disease in early stages of
chronic kidney disease in a Chinese population. J Am Soc Nephrol 2006;
17: 2617–2621.
462. Banerjee D, Chitalia N, Raja R et al. Metabolic syndrome in chronic kidney
disease and renal transplant patients in North India. Int Urol Nephrol
2012; 44: 937–943.
463. Di Angelantonio E, Chowdhury R, Sarwar N et al. Chronic kidney disease
and risk of major cardiovascular disease and non-vascular mortality:
prospective population based cohort study. BMJ 2010; 341: c4986.
464. Parikh NI, Hwang SJ, Larson MG et al. Chronic kidney disease as a
predictor of cardiovascular disease (from the Framingham Heart Study).
Am J Cardiol 2008; 102: 47–53.
144 Kidney International Supplements (2013) 3, 136–150
references

465. Eiland LS, Luttrell PK. Use of statins for dyslipidemia in the pediatric
population. J Pediatr Pharmacol Ther 2010; 15: 160–172.
466. McAlister FA, Ezekowitz J, Tonelli M et al. Renal insufficiency and heart
failure: prognostic and therapeutic implications from a prospective
cohort study. Circulation 2004; 109: 1004–1009.
467. Smith GL, Lichtman JH, Bracken MB et al. Renal impairment and
outcomes in heart failure: systematic review and meta-analysis. J Am Coll
Cardiol 2006; 47: 1987–1996.
468. Hillege HL, Nitsch D, Pfeffer MA et al. Renal function as a predictor of
outcome in a broad spectrum of patients with heart failure. Circulation
2006; 113: 671–678.
469. Shlipak MG, Smith GL, Rathore SS et al. Renal function, digoxin therapy,
and heart failure outcomes: evidence from the digoxin intervention
group trial. J Am Soc Nephrol 2004; 15: 2195–2203.
470. Smith GL, Shlipak MG, Havranek EP et al. Race and renal impairment in
heart failure: mortality in blacks versus whites. Circulation 2005; 111:
1270–1277.
471. Anand IS, Bishu K, Rector TS et al. Proteinuria, chronic kidney disease,
and the effect of an angiotensin receptor blocker in addition to an
angiotensin-converting enzyme inhibitor in patients with moderate to
severe heart failure. Circulation 2009; 120: 1577–1584.
472. Ezekowitz J, McAlister FA, Humphries KH et al. The association among
renal insufficiency, pharmacotherapy, and outcomes in 6,427 patients
with heart failure and coronary artery disease. J Am Coll Cardiol 2004; 44:
1587–1592.
473. Gottlieb SS, Abraham W, Butler J et al. The prognostic importance of
different definitions of worsening renal function in congestive heart
failure. J Card Fail 2002; 8: 136–141.
474. Middleton RJ, Parfrey PS, Foley RN. Left ventricular hypertrophy in the
renal patient. J Am Soc Nephrol 2001; 12: 1079–1084.
475. Levin A, Singer J, Thompson CR et al. Prevalent left ventricular
hypertrophy in the predialysis population: identifying opportunities for
intervention. Am J Kidney Dis 1996; 27: 347–354.
476. Ha SK, Park HS, Kim SJ et al. Prevalence and patterns of left ventricular
hypertrophy in patients with predialysis chronic renal failure. J Korean
Med Sci 1998; 13: 488–494.
477. Parfrey PS, Foley RN, Harnett JD et al. Outcome and risk factors for left
ventricular disorders in chronic uraemia. Nephrol Dial Transplant
1996;
11: 1277–1285.
478. Cerasola G, Nardi E, Palermo A et al. Epidemiology and pathophysiology
of left ventricular abnormalities in chronic kidney disease: a review.
J Nephrol 2011; 24: 1–10.
479. Ahmed A, Rich MW, Sanders PW et al. Chronic kidney disease associated
mortality in diastolic versus systolic heart failure: a propensity matched
study. Am J Cardiol 2007; 99: 393–398.
480. Lazzeri C, Valente S, Tarquini R et al. Cardiorenal syndrome caused by heart
failure with preserved ejection fraction. Int J Nephrol 2011; 634903.
481. Levin A, Thompson CR, Ethier J et al. Left ventricular mass index increase
in early renal disease: impact of decline in hemoglobin. Am J Kidney Dis
1999; 34: 125–134.
482. Go AS, Yang J, Ackerson LM et al. Hemoglobin level, chronic kidney
disease, and the risks of death and hospitalization in adults with chronic
heart failure: the Anemia in Chronic Heart Failure: Outcomes and
Resource Utilization (ANCHOR) Study. Circulation 2006; 113: 2713–2723.
483. Shlipak MG, Katz R, Kestenbaum B et al. Clinical and subclinical
cardiovascular disease and kidney function decline in the elderly.
Atherosclerosis 2009; 204: 298–303.
484. Balamuthusamy S, Srinivasan L, Verma M et al. Renin angiotensin system
blockade and cardiovascular outcomes in patients with chronic kidney
disease and proteinuria: a meta-analysis. Am Heart J 2008; 155: 791–805.
485. Lewis EJ, Hunsicker LG, Clarke WR et al. Renoprotective effect of the
angiotensin-receptor antagonist irbesartan in patients with nephropathy
due to type 2 diabetes. N Engl J Med 2001; 345: 851–860.
486. Erdmann E, Lechat P, Verkenne P et al. Results from post-hoc analyses of
the CIBIS II trial: effect of bisoprolol in high-risk patient groups with
chronic heart failure. Eur J Heart Fail 2001; 3: 469–479.
487. Wali RK, Iyengar M, Beck GJ et al. Efficacy and safety of carvedilol in
treatment of heart failure with chronic kidney disease: a meta-analysis of
randomized trials. Circ Heart Fail 2011; 4: 18–26.
488. Pitt B, Zannad F, Remme WJ et al. The effect of spironolactone on
morbidity and mortality in patients with severe heart failure. Rando-
mized Aldactone Evaluation Study Investigators. N Engl J Med 1999; 341:
709–717.
489. Juurlink DN, Mamdani MM, Lee DS et al. Rates of hyperkalemia after
publication of the Randomized Aldactone Evaluation Study. N Engl J Med
2004; 351: 543–551.
490. Abdel-Qadir HM, Chugh S, Lee DS. Improving prognosis estimation in
patients with heart failure and the cardiorenal syndrome.
Int J Nephrol
2011; 351672.
491. Levin ER, Gardner DG, Samson WK. Natriuretic peptides. N Engl J Med
1998; 339: 321–328.
492. Wiley CL, Switzer SP, Berg RL et al. Association of B-type natriuretic
Peptide levels with estimated glomerular filtration rate and congestive
heart failure. Clin Med Res 2010; 8: 7–12.
493. Madsen LH, Ladefoged S, Corell P et al. N-terminal pro brain natriuretic
peptide predicts mortality in patients with end-stage renal disease in
hemodialysis. Kidney Int 2007; 71: 548–554.
494. Gustafsson F, Steensgaard-Hansen F, Badskjaer J et al. Diagno stic and
prognostic performance of N-terminal ProBNP in primary care patients
with suspected heart failure. J Card Fail 2005; 11: S15–20.
495. McCullough PA, Omland T, Maisel AS. B-type natriuretic peptides: a
diagnostic breakthrough for clinicians. Rev Cardiovasc Med 2003; 4:
72–80.
496. Maisel A, Mehra MR. Understanding B-type natriuretic peptide and
its role in diagnosing and monitoring congestive heart failure.
Clin Cornerstone 2005; 7 (Suppl 1): S7–17.
497. Wright SP, Doughty RN, Pearl A et al. Plasma amino-terminal pro-brain
natriuretic peptide and accuracy of heart-failure diagnosis in primary
care: a randomized, controlled trial. J Am Coll Cardiol 2003; 42:
1793–1800.
498. Yamamoto K, Burnett JC, Jr., Jougasaki M et al. Superiority of brain
natriuretic peptide as a hormonal marker of ventricular systolic and
diastolic dysfunction and ventricular hypertrophy. Hypertension 1996;
28: 988–994.
499. Burnett JC, Jr., Kao PC, Hu DC et al. Atrial natriuretic peptide elevation in
congestive heart failure in the human. Science 1986; 231: 1145–1147.
500. Mueller C, Laule-Kilian K, Scholer A et al. B-type natriuretic peptide for
acute dyspnea in patients with kidney disease: insights from a
randomized comparison. Kidney Int 2005; 67: 278–284.
501. Vickery S, Price CP, John RI et al. B-type natriuretic peptide (BNP) and
amino-terminal proBNP in patients with CKD: relationship to renal
function and left ventricular hypertrophy. Am J Kidney Dis 2005; 46:
610–620.
502. Ishibe S, Peixoto AJ. Methods of assessment of volume status and
intercompartmental fluid shifts in hemodialysis patients: implications in
clinical practice. Semin Dial 2004; 17: 37–43.
503. Takahashi M, Nagake Y, Ichikawa H et al. Plasma concentrations of
natriuretic peptides in patients on hemodialysis. Res Commun Mol Pathol
Pharmacol 1996; 92:
19–30.
504. Tagore R, Ling LH, Yang H et al. Natriuretic peptides in chronic kidney
disease. Clin J Am Soc Nephrol 2008; 3: 1644–1651.
505. Takami Y, Horio T, Iwashima Y et al. Diagnostic and prognostic value
of plasma brain natriuretic peptide in non-dialysis-de pendent CRF.
Am J Kidney Dis 2004; 44: 420–428.
506. Zoccali C, Mallamaci F, Benedetto FA et al. Cardiac natriuretic peptides
are related to left ventricular mass and function and predict mortality in
dialysis patients. J Am Soc Nephrol 2001; 12: 1508–1515.
507. Suresh M, Farrington K. Natriuretic peptides and the dialysis patient.
Semin Dial 2005; 18: 409–419.
508. Kistorp C, Raymond I, Pedersen F et al. N-terminal pro-brain natriuretic
peptide, C-reactive protei n, and urinary albumin levels as predictors of
mortality and cardiovascular events in older adults. JAMA 2005; 293:
1609–1616.
509. Bettencourt P, Azevedo A, Pimenta J et al. N-terminal-pro-brain
natriuretic peptide predicts outcome after hospital discharge in heart
failure patients. Circulation 2004; 110: 2168–2174.
510. Gardner RS, Ozalp F, Murday AJ et al. N-terminal pro-brain natriuretic
peptide. A new gold standard in predicting mortality in patients with
advanced heart failure. Eur Heart J 2003; 24: 1735–1743.
511. Kragelund CB, Gronning BA, Kober L et al. [Prognostic value of
N-terminal pro-BNP-type natriuretic peptide in patients with stable
coronary heart disease–secondary publication]. Ugeskr Laeger 2006; 168:
697–700.
512. Omland T, Persson A, Ng L et al. N-terminal pro-B-type natriuretic
peptide and long-term mortality in acute coronary syndromes. Circula-
tion 2002; 106: 2913–2918.
513. Olsen MH, Wachtell K, Tuxen C et al. N-terminal pro-brain natriuretic
peptide predicts cardiovascular events in patients with hypertension
and left ventricular hypertrophy: a LIFE study. J Hypertens 2004; 22:
1597–1604.
514. Koch A, Singer H. Normal values of B type natriuretic peptide in infants,
children, and adolescents. Heart 2003; 89: 875–878.
Kidney International Supplements (2013) 3, 136–150 145
references

515. Auerbach SR, Richmond ME, Lamour JM et al. BNP levels predict
outcome in pediatric heart failure patients: post hoc analysis of the
Pediatric Carvedilol Trial. Circ Heart Fail 2010; 3: 606–611.
516. Barret BJ, Culleton B. Reducing the burden of cardiovascular disease in
patients on dialysis. Dial Transplant 2002; 31: 155–163.
517. Beciani M, Tedesco A, Violante A et al. Cardiac troponin I
(2nd generation assay) in chronic haemodialysis patients: prevalence
and prognosti c value. Nephrol Dial Transplant 2003; 18: 942–946.
518. Apple FS, Wu AH. Myocardial infarction redefined: role of cardiac
troponin testing. Clin Chem 2001; 47: 377–379.
519. McLaurin MD, Apple FS, Voss EM et al. Cardiac troponin I, cardiac
troponin T, and creatine kinase MB in dialysis patients without ischemic
heart disease: evidence of cardiac troponin T expression in skeletal
muscle. Clin Chem 1997; 43: 976–982.
520. Musso P, Cox I, Vidano E et al. Cardiac troponin elevations in chronic
renal failure: prevalence and clinical significance. Clin Biochem 1999; 32:
125–130.
521. Alpert JS, Thygesen K, Antman E et al. Myocardial infarction redefined–a
consensus doc ument of The Joint European Society of Cardiology/
American College of Cardiology Committee for the redefinition of
myocardial infarction. J Am Coll Cardiol 2000; 36: 959–969.
522. Collinson PO, Boa FG, Gaze DC. Measurement of cardiac troponins.
Ann Clin Biochem 2001; 38: 423–449.
523. Lindahl B, Venge P, Wallentin L. Troponin T identifies patients with
unstable coronary artery disease who benefit from long-term anti-
thrombotic protection. Fragmin in Unstable Coronary Artery Disease
(FRISC) Study Group. J Am Coll Cardiol 1997; 29: 43–48.
524. Rabbani LE. Acute coronary syndromes–beyond myocyte necrosis.
N Engl J Med 2001; 345: 1057–1059.
525. Tun A, Khan IA, Win MT et al. Specificity of cardiac troponin I and
creatine kinase-MB isoenzyme in asymptomatic long-term hemodialysis
patients and effect of hemodialysis on these cardiac markers. Cardiology
1998; 90: 280–285.
526. Wood GN, Keevil B, Gupta J et al. Serum troponin T measurement in patients
with chronic renal impairment predicts survival and vascular disease: a 2 year
prospective study. Nephrol Dial Transplant 2003; 18: 1610–1615.
527. Abbas NA, John RI, Webb MC et al. Cardiac troponins and renal function
in nondialysis patients with chronic kidney disease. Clin Chem 2005; 51:
2059–2066.
528. Kinnunen P, Vuolteenaho O, Ruskoaho H. Mechanisms of atrial and brain
natriuretic peptide release from rat ventricular myocardium: effect of
stretching. Endocrinology 1993; 132: 1961–1970.
529. deFilippi C, Wasserman S, Rosanio S et al. Cardiac troponin T and
C-reactive protein for predicting prognosis, coronary atherosclerosis,
and cardiomyopathy in patients undergoing long-term hemodialysis.
JAMA 2003; 290: 353–359.
530. Hayashi T, Obi Y, Kimura T et al. Cardiac troponin T predicts occult
coronary artery stenosis in patients with chronic kidney disease at the
start of renal replacement therapy. Nephrol Dial Transplant 2008; 23:
2936–2942.
531. Roongsritong C, Warraich I, Bradley C. Common causes of troponin
elevations in the absence of acute myocardial infarction: incidence and
clinical significance. Chest 2004; 125: 1877–1884.
532. Apple FS, Murakami MM, Pearce LA et al. Multi-biomarker risk
stratification of N-terminal pro-B-type natriuretic peptide, high-sensitiv-
ity C-reactive protein, and cardiac troponin T and I in end-stage renal
disease for all-cause death. Clin Chem 2004; 50: 2279–2285.
533. De Zoysa JR. Cardiac troponins and renal disease. Nephrology (Carlton)
2004; 9: 83–88.
534. Deegan PB, Lafferty ME, Blumsohn A et al. Prognostic value of troponin T
in hemodialysis patients is independent of comorbidity. Kidney Int 2001;
60: 2399–2405.
535. Dierkes J, Domrose U, Westphal S et al. Cardiac troponin T predicts
mortality in patients with end-stage renal disease. Circulation 2000; 102:
1964–1969.
536. Fehr T, Knoflach A, Ammann P et al. Differential use of cardiac troponin T
versus I in hemodialysis patients. Clin Nephrol 2003; 59: 35–39.
537. Francis GS, Tang WH. Cardiac troponins in renal insufficiency and other
non-ischemic cardiac conditions. Prog Cardiovasc Dis 2004; 47: 196–206.
538. Freda BJ, Tang WH, Van Lente F et al. Cardiac troponins in renal
insufficiency: review and clinical implications. J Am Coll Cardiol 2002; 40:
2065–2071.
539. Iliou MC, Fumeron C, Benoit MO et al. Factors associated with increased
serum levels of cardiac troponins T and I in chronic haemodialysis
patients: Chronic Haemodialysis And New Cardiac Markers Evaluation
(CHANCE) study. Nephrol Dial Transplant 2001; 16: 1452–1458.
540. Lamb EJ, Webb MC, Abbas NA. The significance of serum troponin T in
patients with kidney disease: a review of the literature. Ann Clin Biochem
2004; 41: 1–9.
541. Lowbeer C, Ottosson-Seeberger A, Gustafsson SA et al. Increased cardiac
troponin T and endothelin-1 concentrations in dialysis patients may
indicate heart disease. Nephrol Dial Transplant 1999;
14: 1948–1955.
542. Mallamaci F, Zoccali C, Parlongo S et al. Troponin is related to left
ventricular mass and predicts all-cause and cardiovascular mortality in
hemodialysis patients. Am J Kidney Dis 2002; 40: 68–75.
543. Needham DM, Shufelt KA, Tomlinson G et al. Troponin I and T levels in
renal failure patients without acute coronary syndrome: a systematic
review of the literature. Can J Cardiol 2004; 20: 1212–1218.
544. Ooi DS, Zimmerman D, Graham J et al. Cardiac troponin T predicts long-
term outcomes in hemodialysis patients. Clin Chem 2001; 47: 412–417.
545. Roberts MA, Fernando D, Macmillan N et al. Single and serial
measurements of cardiac troponin I in asymptomatic patients on
chronic hemodialysis. Clin Nephrol 2004; 61: 40–46.
546. Collinson PO, Hadcocks L, Foo Y et al. Cardiac troponins in patients with
renal dysfunction. Ann Clin Biochem 1998; 35 (Pt 3): 380–386.
547. Roppolo LP, Fitzgerald R, Dillow J et al. A comparison of troponin T and
troponin I as predictors of cardiac events in patients undergoing chronic
dialysis at a Veteran’s Hospital: a pilot study. J Am Coll Cardiol 1999; 34:
448–454.
548. de Lemos JA, Drazner MH, Omland T et al. Association of troponin
T detected with a highly sensitive assay and cardiac structure and
mortality risk in the general population. JAMA 2010; 304: 2503–2512.
549. Haller C, Zehelein J, Remppis A et al. Cardiac troponin T in patients with
end-stage renal disease: absence of expression in truncal skeletal
muscle. Clin Chem 1998; 44: 930–938.
550. Abaci A, Ekici E, Oguzhan A et al. Cardiac troponins T and I in patients
with end-stage renal disease: the relation with left ventricular mass and
their prognostic value. Clin Cardiol 2004; 27: 704–709.
551. Ooi DS, House AA. Cardiac troponin T in hemodialyzed patients.
Clin Chem 1998; 44: 1410–1416.
552. Parfrey PS, Foley RN. The clinical epidemiology of cardiac disease in
chronic renal failure. J Am Soc Nephrol 1999; 10: 1606–1615.
553. Roberts MA, Hedley AJ, Ierino FL. Understanding cardiac biomarkers in
end-stage kidney disease: Frequently asked questions and the promise
of clinical application. Nephrology (Carlton) 2011; 16: 251–260.
554. Hirsch R, Landt Y, Porter S et al. Cardiac troponin I in pediatrics: normal
values and potential use in the assessment of cardiac injury. J Pediatr
1997;
130: 872–877.
555. Liesemer K, Casper TC, Korgenski K et al. Use and misuse of serum
troponin assays in pediatric practice. Am J Cardiol 2012; 110: 284–289.
556. Lentine KL, Hurst FP, Jindal RM et al. Cardiovascular risk assessment
among potential kidney transplant candidates: approaches and con-
troversies. Am J Kidney Dis 2010; 55: 152–167.
557. McIntyre CW, Odudu A, Eldehni MT. Cardi ac assessment in chronic
kidney disease. Curr Opin Nephrol Hypertens 2009; 18: 501–506.
558. Bennett WM, Kloster F, Rosch J et al. Natural history of asymptomatic
coronary arteriographic lesions in diabetic patients with end-stage renal
disease. Am J Med 1978; 65: 779–784.
559. Braun WE, Phillips DF, Vidt DG et al. Coronary artery disease in 100
diabetics with end-stage renal failure. Transplant Proc 1984; 16: 603–607.
560. Lorber MI, Van Buren CT, Flechner SM et al. Pretransplant coron ary
arteriography for diabetic renal transplant recipients. Transplant Proc
1987; 19: 1539–1541.
561. Manske CL, Wilson RF, Wang Y et al. Prevalence of, and risk factors for,
angiographically determined coronary artery disease in type I-diabetic
patients with nephropathy. Arch Intern Med 1992; 152: 2450–2455.
562. Weinrauch L, D’Elia JA, Healy RW et al. Asymptomatic coronary
artery disease: angiographic assessment of diabetics evaluated for renal
transplantation. Circulation 1978; 58: 1184–1190.
563. Klocke FJ, Baird MG, Lorell BH et al. ACC/AHA/ASNC guidelines for the
clinical use of cardiac radionuclide imaging–executive summary: a report
of the American College of Cardiology/American Heart Association Task
Force on Practice Guidelines (ACC/AHA/ASNC Committee to Revise the
1995 Guidelines for the Clinical Use of Cardiac Radionuclide Imaging).
J Am Coll Cardiol 2003; 42: 1318–1333.
564. Dahan M, Viron BM, Faraggi M et al. Diagnostic accuracy and prognostic
value of combined dipyridamole-exercise thallium imaging in hemo-
dialysis patients. Kidney Int 1998; 54: 255–262.
565. Koistinen MJ, Huikuri HV, Pirttiaho H et al. Evaluation of exercise
electrocardiography and thallium tomographic imaging in detecting
asymptomatic coronary artery disease in diabetic patients. Br Heart J
1990; 63: 7–11.
146 Kidney International Supplements (2013) 3, 136–150
references

566. Marwick TH, Steinmuller DR, Underwood DA et al. Ineffectiveness of
dipyridamole SPECT thallium imaging as a screening technique for
coronary artery disease in patients with end-stage renal failure.
Transplantation 1990; 49: 100–103.
567. Schmidt A, Stefenelli T, Schuster E et al. Informational contribution
of noninvasive screening tests for coronary artery disease in
patients on chronic renal replacement therapy. Am J Kidney Dis 2001;
37: 56–63.
568. Morrow CE, Schwartz JS, Sutherland DE et al. Predictive value of thallium
stress testing for coronary and cardiovascular events in uremic diabetic
patients before renal transplantation. Am J Surg 1983; 146: 331–335.
569. Patel AD, Abo-Auda WS, Davis JM et al. Prognostic value of myocardial
perfusion imaging in predicting outcome after renal transplantation. Am
J Cardiol 2003; 92: 146–151.
570. Rabbat CG, Treleaven DJ, Russell JD et al. Prognostic value of myocardial
perfusion studies in patients with end-stage renal disease assessed for
kidney or kidney-pancreas transplantation: a meta-analysis. J Am Soc
Nephrol 2003; 14: 431–439.
571. Wong CF, Little MA, Vinjamuri S et al. Technetium myocardial perfusion
scanning in prerenal transplant evaluation in the United kingdom.
Transplant Proc 2008; 40: 1324–1328.
572. De Lima JJ, Sabbaga E, Vieira ML et al. Coronary angiography is the best
predictor of events in renal transplant candidates compared with
noninvasive testing. Hypertension 2003; 42: 263–268.
573. Hatta T, Nishimura S, Nishimura T. Prognostic risk stratification of
myocardial ischaemia evaluated by gated myocardial perfusion SPECT in
patients with chronic kidney disease. Eur J Nucl Med Mol Imaging 2009;
36: 1835–1841.
574. Kusuoka H, Nishimura S, Yamashina A et al. Surveillance study for
creating the national clinical database related to ECG-gated myocardial
perfusion SPECT of ischemic heart disease: J-ACCESS study design.
Ann Nucl Med 2006; 20: 195–202.
575. Momose M, Babazono T, Kondo C et al. Prognostic significance of stress
myocardial ECG-gated perfusion imaging in asymptomatic patients with
diabetic chronic kidney disease on initiation of haemodialysis. Eur J Nucl
Med Mol Imaging 2009; 36: 1315–1321.
576. Nakajima K, Nishimura T. Inter-institution preference-based variability of
ejection fraction and volumes using quantitative gated SPECT with
99mTc-tetrofosmin: a multicentre study involving 106 hospitals. Eur J
Nucl Med Mol Imaging 2006; 33: 127–133.
577. Nekolla SG, Reder S, Saraste A et al. Evaluation of the novel myocardial
perfusion positron-emission tomography tracer 18F-BMS-747158-02:
comparison to 13N-ammonia and validation with microspheres in a
pig model. Circulation 2009; 119: 2333–2342.
578. Biomarkers Definitions Working Group. Biomarkers and surrogate
endpoints: preferred definitions and conceptual framework.
Clin
Pharmacol Ther 2001; 69: 89–95.
579. Ix JH, Criqui MH. Epidemiology and diagnosis of peripheral arterial
disease in patients with chronic kidney disease. Adv Chronic Kidney Dis
2008; 15: 378–383.
580. Lau JF, Weinberg MD, Olin JW. Peripheral artery disease. Part 1: clinical
evaluation and noninvasive diagnosis. Nat Rev Cardiol 2011; 8: 405–418.
581. Bello AK, Hemmelgarn B, Lloyd A et al. Associations among estimated
glomerular filtration rate, proteinuria, and adverse cardiovascular out-
comes. Clin J Am Soc Nephrol 2011; 6: 1418–1426.
582. de Vinuesa SG, Ortega M, Martinez P et al. Subclinical peripheral arterial
disease in patients with chronic kidney disease: prevalence and related
risk factors. Kidney Int Suppl 2005; 93: S44–47.
583. Lash JP, Go AS, Appel LJ et al. Chronic Renal Insufficiency Cohort (CRIC)
Study: baseline characteristics and associations with kidney function.
Clin J Am Soc Nephrol 2009; 4: 1302–1311.
584. Liew YP, Bartholomew JR, Demirjian S et al. Combined effect
of chronic kidney disease and peripheral arterial disease on all-cause
mortality in a high-risk population. Clin J Am Soc Nephrol 2008; 3:
1084–1089.
585. O’Hare AM, Glidden DV, Fox CS et al. High prevalence of peripheral
arterial disease in persons with renal insufficiency: results from the
National Health and Nutrition Examination Survey 1999–2000. Circulation
2004; 109: 320–323.
586. O’Hare AM, Newman AB, Katz R et al. Cystatin C and incident peripheral
arterial disease events in the elderly: results from the Cardiovascular
Health Study. Arch Intern Med 2005; 165: 2666–2670.
587. O’Hare AM, Vittinghoff E, Hsia J et al. Renal insufficiency and the risk of
lower extremity peripheral arterial disease: results from the Heart and
Estrogen/Progestin Replacem ent Study (HERS). J Am Soc Nephrol 2004;
15: 1046–1051.
588. Wattanakit K, Folsom AR, Criqui MH et al. Albuminuria and peripheral
arterial disease: results from the multi-ethnic study of atherosclerosis
(MESA). Atherosclerosis 2008; 201: 212–216.
589. Margolis DJ, Hofstad O, Feldman HI. Association between renal failure
and foot ulcer or lower-extremity amputation in patients with diabetes.
Diabetes Care 2008; 31: 1331–1336.
590. Fuster V, Ryden LE, Cannom DS et al. ACC/AHA/ESC 2006 Guidelines for
the Management of Patients with Atrial Fibrillation: a report of the
American College of Cardiology/American Heart Association Task Force
on Practice Guidelines and the European Society of Cardiology
Committee for Practice Guidelines (Writing Committee to Revise the
2001 Guidelines for the Management of Patients With Atrial Fibrillation):
developed in collaboration with the European Heart Rhythm Association
and the Heart Rhythm Society. Circulation 2006; 114: e257–354.
591. Abbas AE, Goodman LM, Timmis R
et al. Predictors of poor outcome in
female patients undergoing endovascular intervention. J Interv Cardiol
2010; 23: 401–410.
592. O’Hare AM, Bertenthal D, Sidawy AN et al. Renal insufficiency and use of
revascularization among a national cohort of men with advanced lower
extremity peripheral arterial disease. Clin J Am Soc Nephrol 2006; 1:
297–304.
593. Goodman and Gilman’s Pharmacological Basis of Therapeutics, 12th Edn.
Brunston LL, Chabner BA, Knollman BA (eds). McGraw-Hill: New York,
2011.
594. Martindale: The Complete Drug Reference, 37th Edn. Sweetman SC (ed).
Royal Pharmaceutical Society, London, 2011.
595. Gokmen MR, Lord GM. Aristolochic acid nephropathy. BMJ 2012; 344:
e4000.
596. Su T, Zhang L, Li X et al. Regular use of nephrotoxic medications
is an independent risk factor for chronic kidney disease–results
from a Chinese population study. Nephrol Dial Transplant 2011; 26:
1916–1923.
597. National Institute for Health and Clinical Excellence. NICE Clinical
Guideline 38: Bipolar Disorder. 2006.
598. Lipska KJ, Bailey CJ, Inzucchi SE. Use of metformin in the setting of mild-
to-moderate renal insufficiency. Diabetes Care 2011; 34: 1431–1437.
599. Rachmani R, Slavachevski I, Levi Z et al. Metformin in patients with type 2
diabetes mellitus: reconsideration of traditional contraindications. Eur J
Intern Med 2002; 13: 428.
600. Salpeter S, Greyber E, Pasternak G et al. Risk of fatal and nonfatal lactic
acidosis with metformin use in type 2 diabetes mellitus. Cochrane
Database Syst Rev 2010: CD002967.
601. Fellstrom BC, Jardine AG, Schmieder RE et al. Rosuvastatin and
cardiovascular events in patients undergoing hemodialysis. NEnglJ
Med 2009; 360: 1395–1407.
602. Ginsberg HN, Elam MB, Lovato LC et al. Effects of combination
lipid therapy in type 2 diabetes mellitus. N Engl J Med 2010; 362:
1563–1574.
603. Keech A, Simes RJ, Barter P et al. Effects of long-term fenofibrate therapy
on cardiovascular events in 9795 people with type 2 diabetes mellitus
(the FIELD study): randomised controlled trial. Lancet 2005; 366:
1849–1861.
604. Wanner C, Krane V, Marz W et al. Atorvastatin in patients with type 2
diabetes mellitus undergoing hemodialysis. NEnglJMed2005; 353:
238–248.
605. Limdi NA, Beasley TM, Baird MF et al. Kidney function influences warfarin
responsiveness and hemorrhagic complications. J Am Soc Nephrol
2009;
20: 912–921.
606. Wahba IM, Olyaei AJ, Rozansky D et al. Handling of drugs in children with
abnormal renal function, in Pediatric Nephrology, eds. Avner ED, Harmon
WE, Niaudet P, Yoshikawa, N. Springer-Verlag: 2009, pp 1693–1711.
607. Blowey DL. Chapter 64: Drug Use and Dosage in Renal Failure, in
Comprehensive Pediatric Nephrology, eds. Geary DF, Schaefer F. Mosby
Elsevier: Philadelphia, 2008, pp 991–1002.
608. Drug prescribing in renal failure. http://www.kdp-baptist.louisville.edu/
renalbook/.
609. Goldfarb S, McCullough PA, McDermott J et al. Contrast-induced acute
kidney injury: specialty-specific protocols for interventional radiology,
diagnostic computed tomography radiology, and interventional cardio-
logy. Mayo Clin Proc 2009; 84: 170–179.
610. American College Radiology Guidelines. Manual on Contrast Media
Version 8. http://www.acr.org/B/media/ACR/Documents/PDF/Quality
Safety/Resources/Contrast%20Manual/Contrast%20Nephrotoxicity.pdf.
Accessed October 12, 2012.
611. European Society for Urological Radiology (ESUR). http:/ /www.esur.org/
Contrast-media.51.0.html, Accessed October 9, 2012.
Kidney International Supplements (2013) 3, 136–150 147
references

612. Heinrich MC, Haberle L, Muller V et al. Nephrotoxicity of iso-osmolar
iodixanol compared with nonionic low-osmolar contrast media: meta-
analysis of randomized controlled trials. Radiology 2009; 250: 68–86.
613. Perazella MA. Current status of gadolinium toxicity in patients with
kidney disease. Clin J Am Soc Nephrol 2009; 4: 461–469.
614. Agarwal R, Brunelli SM, Williams K et al. Gadolinium-based contrast
agents and nephrogenic systemic fibrosis: a systematic review and meta-
analysis. Nephrol Dial Transplant 2009; 24: 856–863.
615. American College Radiology Guidelines. Manual on Contrast Media
Version 8. http://www.acr.org/B/media/ACR/Documents/PDF/Quality-
Safety/Resources/Contrast%20Manual/Nephrogenic%20Systemic%20Fi-
brosis.pdf. Accessed October 12, 2012.
616. Saitoh T, Hayasaka K, Tanaka Y et al. Dialyzability of gadodiamide in
hemodialysis patients. Radiat Med 2006; 24: 445–451.
617. Meng H, Grosse-Wortmann L. Gadolinium in pediatric cardiovascular
magnetic resonance: what we know and how we practice. J Cardiovasc
Magn Reson 2012; 14: 56.
618. Lien YH. Is bowel preparation before colonoscopy a risky business for
the kidney? Nat Clin Pract Nephrol 2008; 4: 606–614.
619. Markowitz GS, Perazella MA. Acute phosphate nephropathy. Kidney Int
2009; 76: 1027–1034.
620. Heher EC, Thier SO, Rennke H et al. Adverse renal and metabolic effects
associated with oral sodium phosphate bowel preparation. Clin J Am Soc
Nephrol 2008; 3: 1494–1503.
621. Palmadottir VK, Gudmundsson H, Hardarson S et al. Incidence and
outcome of acute phosphate nephropathy in Iceland. PLoS One 2010; 5:
e13484.
622. Wexner SD, Beck DE, Baron TH et al. A consensus document on bowel
preparation before colonoscopy: prepared by a task force from the
American Society of Colon and Rectal Surgeons (ASCRS), the American
Society for Gastrointestinal Endoscopy (ASGE), and the Society of
American Gastrointestinal and Endoscopic Surgeons (SAGES). Gastro-
intest Endosc 2006; 63: 894–909.
623. Brunelli SM. Association between oral sodium phosphate bowel
preparations and kidney injury: a systematic review and meta-analysis.
Am J Kidney Dis 2009; 53: 448–456.
624. Johanson JF, Popp JW, Jr., Cohen LB et al. A randomized, multicenter
study comparing the safety and efficacy of sodium phosphate tablets
with 2 L polyethylene glycol solution plus bisacodyl tablets for colon
cleansing. Am J Gastroenterol 2007; 102: 2238–2246.
625. Foley RN, Guo H, Snyder JJ et al.
Septicemia in the United States dialysis
population, 1991 to 1999. J Am Soc Nephrol 2004; 15: 1038–1045.
626. Hoen B, Paul-Dauphin A, Hestin D et al. EPIBACDIAL: a multicenter
prospective study of risk factors for bacteremia in chronic hemodialysis
patients. J Am Soc Nephrol 1998; 9: 869–876.
627. Ishani A, Collins AJ, Herzog CA et al. Septicemia, access and
cardiovascular disease in dialysis patients: the USRDS Wave 2 study.
Kidney Int 2005; 68: 311–318.
628. Sarnak MJ, Jaber BL. Mortality caused by sepsis in patients with end-
stage renal disease compared with the general population. Kidney Int
2000; 58: 1758–1764.
629. Fried LF, Katz R, Sarnak MJ et al. Kidney function as a predictor of
noncardiovascular mortality. J Am Soc Nephrol 2005; 16: 3728–3735.
630. Nissenson AR, Collins AJ, Hurley J et al. Opportunities for improving the
care of patients with chronic renal insufficiency: current practice
patterns. J Am Soc Nephrol 2001; 12: 1713–1720.
631. US Renal Data System. Atlas of Chronic Kidney Disease in the United
States. Bethesda, MD: National Institutes of Diabetes and Digestive and
Kidney Diseases, National Institutes of Health. 2007.
632. Naqvi SB, Collins AJ. Infectious complications in chronic kidney disease.
Adv Chronic Kidney Dis 2006; 13: 199–204.
633. US Renal Data System. Atlas of Chronic Kidney Disease in the United
States. Bethesda, MD: National Institutes of Diabetes and Digestive and
Kidney Diseases, National Institutes of Health. 2010.
634. McIntyre P, Craig JC. Prevention of serious bacterial infection in
children with nephrotic syndrome. J Paediatr Child Health 1998; 34:
314–317.
635. Vanholder R, Ringoir S. Infectious morbidity and defects of phagocytic
function in end-stage renal disease: a review. J Am Soc Nephrol 1993; 3:
1541–1554.
636. Cohen G, Haag-Weber M, Horl WH. Immune dysfunction in uremia.
Kidney Int Suppl 1997; 62: S79–82.
637. Minnaganti VR, Cunha BA. Infections associated with uremia and dialysis.
Infect Dis Clin North Am 2001; 15: 385–406.
638. Pesanti EL. Immunologic defects and vaccination in patients with
chronic renal failure. Infect Dis Clin North Am 2001; 15: 813–832.
639. Kausz AT, Guo H, Pereira BJ et al. General medical care among patients
with chronic kidney disease: opportuni ties for improving outcomes.
J Am Soc Nephrol 2005; 16: 3092–3101.
640. DaRoza G, Loewen A, Djurdjev O et al. Stage of chronic kidney disease
predicts seroconversion after hepatitis B immunization: earlier is better.
Am J Kidney Dis 2003;
42: 1184–1192.
641. Kausz AT, Gilbertson DT. Overview of vaccination in chronic kidney
disease. Adv Chronic Kidney Dis 2006; 13: 209–214.
642. McNulty CA, Bowen JK, Williams AJ. Hepatitis B vaccination in predialysis
chronic renal failure patients a comparison of two vaccination schedules.
Vaccine 2005; 23: 4142–4147.
643. Robinson J. Efficacy of pneumococcal immunization in patients with
renal disease–what is the data? Am J Nephrol 2004; 24: 402–409.
644. Gilbertson DT, Unruh M, McBean AM et al. Influenza vaccine delivery
and effectiveness in end-stage renal disease. Kidney Int 2003; 63:
738–743.
645. Dinits-Pensy M, Forrest GN, Cross AS et al. The use of vaccines in adult
patients with renal disease. Am J Kidney Dis 2005; 46: 997–1011.
646. Nikoskelainen J, Koskela M, Forsstrom J et al. Persistence of antibodies to
pneumococcal vaccine in patients with chronic renal failure. Kidney Int
1985; 28: 672–677.
647. Miller ER, Alter MJ, Tokars JI. Protective effect of hepatitis B vaccine in
chronic hemodialysis patients. Am J Kidney Dis 1999; 33: 356–360.
648. Fattom A, Fuller S, Propst M et al. Safety and immunogenicity of a
booster dose of Staphylococcus aureus types 5 and 8 capsular
polysaccharide conjugate vaccine (StaphVAX) in hemodialysis patients.
Vaccine 2004; 23: 656–663.
649. Shinefield H, Black S, Fattom A et al. Use of a Staphylococcus aureus
conjugate vaccine in patients receiving hemodialysis. N Engl J Med 2002;
346: 491–496.
650. CDC immunization schedules. http://www.cdc.gov/vaccines/schedules/
index.html.
651. American Academy of Pediatrics. Immunization: http://www2.aap.org/
immunization/.
652. Neu AM. Immunizations in children with chronic kidney disease. Pediatr
Nephrol 2012; 27: 1257–1263.
653. Goldstein SL. Acute kidney injury in children: prevention, treatment and
rehabilitation. Contrib Nephrol 2011; 174: 163–172.
654. Goldstein SL. Acute kidney injury in children and its potential
consequences in adulthood. Blood Purif 2012; 33: 131–137.
655. Zappitelli M, Goldstein SL. Acute kidney failure in children, In: Jorres A,
Ronco C, Kellum JA (eds.). Management of Acute Kidney Problems,
Springer, 2010, pp 459–467.
656. Andreoli SP. Clinical evaluation of acute kidney injury in children. In:
Avner ED, Harmon WE, Niaudet P, Yoshikawa N (eds.). Pediatric
Nephrology, (6th edn.). Springer, 2009, pp 1603–1618.
657. Zappitelli M, Goldstein SL. Management of acute kidney failure, In: Avner
ED, Harmon WE, Niaudet P, Yoshikawa N (eds.). Pediatric Nephrology
(6th edn), Springer, 2009, pp 1619–1628.
658. Zappitelli M, Goldstein SL. Acute kidney injury: General aspects. In:
Kiessling SG, Goebel J, Somers MJG (eds.). Pediatric Nephrology in the ICU,
Springer, 2009, pp 85–97.
659. Arora P, Kausz AT, Obrador GT et al. Hospital utilization am ong chronic
dialysis patients. J Am Soc Nephrol 2000; 11: 740–746.
660. Khan SS, Kazmi WH, Abichandani R et al. Health care utilization among
patients with chronic kidney disease. Kidney Int 2002; 62: 229–236.
661. Becker BN, Coomer RW, Fotiadis C et al. Risk factors for hospitalization in
well-dialyzed chronic hemodialysis patients. Am J Nephrol 1999; 19:
565–570.
662. Popovic JR, Kozak LJ. National hospital discharge survey: annual
summary, 1998. Vital Health Stat 13 2000: 1–194.
663. Rocco MV, Soucie JM, Reboussin DM et al. Risk factors for hospital
utilization in chronic dialysis patients. Southeastern Kidney Council
(Network 6). J Am Soc Nephrol 1996; 7: 889–896.
664. Thamer M, Ray NF, Fehrenbach SN et al. Relative risk and economic
consequences of inpatient care among patients with renal failure. JAm
Soc Nephrol 1996; 7: 751–762.
665. Holland DC, Lam M. Predictors of hospitalization and death among pre-
dialysis patients: a retrospective cohort study. Nephrol Dial Transplant
2000; 15: 650–658.
666. Culleton BF, Larson MG, Wilson PW et al. Cardiovascular disease and
mortality in a community-based cohort with mild renal insufficiency.
Kidney Int 1999; 56: 2214–2219.
667. Meyer KB, Levey AS. Controlling the epidemic of cardiovascular disease
in chronic renal disease: report from the National Kidney Foundation
Task Force on cardiovascular disease. J Am Soc Nephrol 1998; 9: S31–42.
148 Kidney International Supplements (2013) 3, 136–150
references

668. Dru
¨
eke TB, Locatelli F, Clyne N et al. Normalization of hemoglobin level
in patients with chronic kidney disease and anemia. N Engl J Med 2006;
355: 2071–2084.
669. Singh AK, Szczech L, Tang KL et al. Correction of anemia with
epoetin alfa in chronic kidney disease. N Engl J Med 2006; 355:
2085–2098.
670. De Coster C, McLaughlin K, Noseworthy TW. Criteria for referring patients
with renal disease for nephrology consultation: a review of the literature.
J Nephrol 2010; 23: 399–407.
671. Navaneethan SD, Kandula P, Jeevanantham V et al. Referral patterns of
primary care physicians for chronic kidney disease in general population
and geriatric patients. Clin Nephrol 2010; 73: 260–267.
672. Navaneethan SD, Nigwekar S, Sengodan M et al. Referral to nephrolo-
gists for chronic kidney disease care: is non-diabetic kidney disease
ignored? Neph ron Clin Pract 2007; 106: c113–118.
673. Chan MR, Dall AT, Fletcher KE et al. Outcomes in patients with chronic
kidney disease referred late to nephrologists: a meta-analysis. Am J Med
2007; 120: 1063–1070.
674. Smart NA, Titus TT. Outcomes of early versus late nephrology referral in
chronic kidney disease: a systematic review. Am J Med 2011; 124:
1073–1080.
675. McLaughlin K, Manns B, Culleton B et al. An economic evaluation of early
versus late referral of patients with progressive renal insufficiency. Am J
Kidney Dis 2001; 38: 1122–1128.
676. Klebe B, Irving J, Stevens PE et al. The cost of implementing UK
guidelines for the management of chronic kidney disease. Nephrol Dial
Transplant 2007; 22: 2504–2512.
677. Barakat AJ. Presentation of the child with renal disease and guidelines
for referral to the pediatric nephrologist. Int J Pediatr 2012; 978673.
678. Barakat AJ, Chesney RW (eds.). Pediatric Nephrology for Primary Care.
American Academy of Pediatrics: Illinois, 2009.
679. Kennedy SE, Bailey R, Kainer G. Causes and outcome of late referral of
children who develop end-stage kidney disease. J Paediatr Child Health
2012; 48: 253–258.
680. Boehm M, Winkelmayer WC, Arbeiter K et al. Late referral to paediatric
renal failure service impairs access to pre-emptive kidney transplantation
in children. Arch Dis Child 2010; 95: 634–638.
681. Epping-Jordan JE, Pruitt SD, Bengoa R et al. Improving the quality of
health care for chronic conditions. Qual Saf Health Care 2004; 13:
299–305.
682. Ajarmeh S, Er L, Brin G et al. The effect of a multidisciplinary care clinic on
the outcomes in pediatric chronic kidney disease. Pediatr Nephrol 2012;
27: 1921–1927.
683. Menon S, Valentini RP, Kapur G et al. Effectiveness of a multidisciplinary
clinic in managing children with chronic kidney disease. Clin J Am Soc
Nephrol 2009; 4: 1170–1175.
684. Korevaar JC, Jansen MA, Dekker FW et al. When to initiate dialysis:
effect of proposed US guidelines on survival. Lancet 2001; 358:
1046–1050.
685. Traynor JP, Simpson K, Geddes CC et al. Early initiation of dialysis fails to
prolong survival in patients with end-stage renal failure. J Am Soc
Nephrol 2002; 13: 2125–2132.
686. Beddhu S, Samore MH, Roberts MS et al. Impact of timing of initiation of
dialysis on mortality. J Am Soc Nephrol 2003; 14: 2305–2312.
687. Clark WF, Na Y, Rosansky SJ et al. Association between estimated
glomerular filtration rate at initiation of dialysis and mortality. CMAJ
2011; 183: 47–53.
688. Hwang SJ, Yang WC, Lin MY et al. Impact of the clinical conditions at
dialysis initiation on mortality in incident haemodialysis patients: a
national cohort study in Taiwan. Nephrol Dial Transplant 2010; 25:
2616–2624.
689. Kazmi WH, Gilbertson DT, Obrador GT et al. Effect of comorbidity on the
increased mortality associated with early initiation of dialysis. Am J
Kidney Dis 2005; 46: 887–896.
690. Lassalle M, Labeeuw M, Frimat L et al. Age and comorbidity may explain
the paradoxical association of an early dialysis start with poor survival.
Kidney Int 2010; 77: 700–707.
691. Sawhney S, Djurdjev O, Simpson K et al. Survival and dialysis initiation:
comparing British Columbia and Scotland registries. Nephrol Dial
Transplant 2009; 24: 3186–3192.
692. Shiao CC, Huang JW, Chien KL et al. Early initiation of dialysis and late
implantation of catheters adversely affect outcomes of patients on
chronic peritoneal dialysis. Perit Dial Int 2008; 28: 73–81.
693. Stel VS, Dekker FW, Ansell D et al. Residual renal function at the start
of dialysis and clinical outcomes. Nephrol Dial Transplant 2009; 24:
3175–3182.
694. Tang SC, Ho YW, Tang AW et al. Delaying initiation of dialysis till
symptomatic uraemia–is it too late? Nephrol Dial Transplant 2007; 22:
1926–1932.
695. Wilson B, Harwood L, Locking-Cusolito H et al. Optimal timing of
initiation of chronic hemodialysis? Hemodial Int 2007; 11: 263–269.
696. Wright S, Klausner D, Baird B et al. Timing of dialysis initiation and
survival in ESRD. Clin J Am Soc Nephrol 2010; 5: 1828–1835.
697. Cooper BA, Branley P, Bulfone L et al. A randomized, controlled
trial of early versus late initiation of dialysis. N Engl J Med 2010; 363:
609–619.
698. Rosansky SJ, Eggers P, Jackson K et al. Early start of hemodialysis may be
harmful. Arch Intern Med 2011; 171: 396–403.
699. Harris A, Cooper BA, Li JJ et al. Cost-effectiveness of initiating
dialysis early: a randomized controlled trial. Am J Kidney Dis 2011; 57:
707–715.
700. Greenbaum LA, Schaefer F. The decision to initiate dialysis in a pediatric
patient. In: Warady BA, Schaefer F, Alexander SR (eds). Pediatric Dialysis,
Springer: New York, 2012, pp 85–100.
701. Kramer A, Stel VS, Tizard J et al. Characteristics and survival of young
adults who started renal replacement therapy during childhood. Nephrol
Dial Transplant 2009; 24: 926–933.
702. Davison SN. Pain in hemodialysis patients: prevalence, cause, severity,
and management. Am J Kidney Dis 2003; 42: 1239–1247.
703. Davison SN, Jhangri GS, Johnson JA. Cross-sectional validity of a
modified Edmonton symptom assessment system in dialysis patients: a
simple assessment of symptom burden. Kidney Int 2006; 69: 1621–1625.
704. Davison SN, Jhangri GS, Johnson JA. Longitudinal validation of a
modified Edmonton symptom assessment system (ESAS) in haemo-
dialysis patients. Nephrol Dial Transplant 2006; 21: 3189–3195.
705. Murphy EL, Murtagh FE, Carey I et al. Understanding symptoms in
patients with advanced chronic kidney disease managed without
dialysis: use of a short patient-completed assessment tool. Nephron Clin
Pract 200 9; 111: c74–80.
706. Murtagh FE, Addington-Hall JM, Donohoe P et al. Symptom manage-
ment in patients with established renal failure managed without dialysis.
EDTNA ERCA J 2006; 32: 93–98.
707. Murtagh FE, Addington-Hall JM, Edmonds PM et al.
Symptoms in
advanced renal disease: a cross-sectional survey of symptom prevalence
in stage 5 chronic kidney disease managed without dialysis. J Palliat Med
2007; 10: 1266–1276.
708. Saini T, Murtagh FE, Dupont PJ et al. Comparative pilot study of
symptoms and quality of life in cancer patients and patients with end
stage renal disease. Palliat Med 2006; 20: 631–636.
709. Temel JS, Greer JA, Muzikansky A et al. Early palliative care for patients
with metastatic non-small-cell lung cancer. N Engl J Med 2010; 363:
733–742.
710. Chandna SM, Da Silva-Gane M, Marshall C et al. Survival of elderly
patients with stage 5 CKD: comparison of conservative management
and renal replacement therapy. Nephrol Dial Transplant 2011; 26:
1608–1614.
711. Davison SN. End-of-life care preferences and needs: perceptions of
patients with chronic kidney disease. Clin J Am Soc Nephrol 2010; 5:
195–204.
712. Davison SN, Murtagh FE, Higginson IJ. Methodological considerations for
end-of-life research in patients with chronic kidney disease. J Nephrol
2008; 21: 268–282.
713. De Biase V, Tobaldini O, Boaretti C et al. Prolonged conservative
treatment for frail elderly patients with end-stage renal disease: the
Verona experience. Nephrol Dial Transplant 2008; 23: 1313–1317.
714. Ellam T, El-Kossi M, Prasanth KC et al. Conservatively managed patients
with stage 5 chronic kidney disease–outcomes from a single center
experience. QJM 2009; 102: 547–554.
715. Germain MJ, Kurella Tamura M, Davison SN. Palliative care in CKD: the
earlier the better. Am J Kidney Dis 2011; 57: 378–380.
716. Murray AM, Arko C, Chen SC et al. Use of hospice in the United States
dialysis population. Clin J Am Soc Nephrol 2006; 1: 1248–1255.
717. Hearn J, Higginson IJ. Do specialist palliative care teams improve
outcomes for cancer patients? A systematic literature review. Palliat Med
1998; 12: 317–332.
718. Higginson IJ, Wade AM, McCarthy M. Effectiveness of two palliative
support teams. J Public Health Med 1992; 14: 50–56.
719. Ventafridda V, De Conno F, Vigano A et al. Comparison of home and
hospital care of advanced cancer patients. Tumori 1989; 75: 619–625.
720. Wallston KA, Burger C, Smith RA et al. Comparing the quality of
death for hospice and non-hospice cancer patients. Med Care 1988; 26:
177–182.
Kidney International Supplements (2013) 3, 136–150 149
references

721. Fassbender K, Smythe JG, Carson M, et al. Report of the Institute for Public
Economics Health Research Group to Alberta Health and Wellness: cost and
utilization of health care services at end of life in Alberta, 1999–2002.
Edmonton, AB: University of Alberta. 2006.
722. Owens DK, Lohr KN, Atkins D et al. AHRQ series paper 5: grading the
strength of a body of evidence when comparing medical interventions–
agency for healthcare research and quality and the effective health-care
program. J Clin Epidemiol 2010; 63: 513–523.
723. Atkins D, Best D, Briss PA et al. Grading quality of evidence and strength
of recommendations. BMJ 2004; 328: 1490.
724. Guyatt GH, Oxman AD, Kunz R et al. Going from evidence to
recommendations. BMJ 2008; 336: 1049–1051.
725. Uhlig K, Macleod A, Craig J et al. Grading evidence and recommenda-
tions for clinical practice guidelines in nephrology. A position statement
from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int
2006; 70: 2058–2065.
726. The AGREE Collaboration. Development and validation of an interna-
tional appraisal instrument for assessing the quality of clinical
practice guidelines: the AGREE project. Qual Saf Health Care 2003; 12:
18–23.
727. Shiffman RN, Shekelle P, Overhage JM et al. Standardized reporting of
clinical practice guidelines: a proposal from the Conference on Guideline
Standardization. Ann Intern Med 2003; 139: 493–498.
728. Institute of Medicine. Finding What Works in Health Care: Standards
for Systematic Reviews. The National Academies Press: Washington,
DC, 2011.
729. Institute of Medicine. Clinical Practice Guidelines We Can Trust.The
National Academies Press: Washington, DC, 2011.
150 Kidney International Supplements (2013) 3, 136–150
references
